This application brief demonstrates the simultaneous enantiomeric separation of beta blockers using UltraPerformance Convergence Chromatography (UPC2), resulting in time and cost savings compared to traditional normal phase techniques.
Beta blockers, one of the most widely prescribed classes of pharmaceuticals, are present in racemic mixtures. Simultaneous enantiomeric separation of this compound class has applicability for a wide range of samples.
Beta blockers, a class of pharmaceuticals, are some of the most widely prescribed drugs. While this class of pharmaceutical compounds is typically present as a racemic mixture, differences in pharmacological activity and potency have been observed for the enantiomers; for example, the S- enantiomer of propanolol is more potent than the R- isomer. These differences may not only affect patient efficacy but may also have an impact on the environment as biodegradation pathways are often enantioselective. This phenomenon has led to the development of a number of simultaneous enantiomeric screening methods for multiple beta blockers under normal phase conditions.1 Method development for these separations requires the screening of multiple chiral columns because selectivity is most easily manipulated by the choice of the stationary phase. This type of approach is both time-consuming and costly.
The Waters ACQUITY UPC2 System utilizes compressed CO2 mobile phases to allow for the analysis of chiral pharmaceutical compounds in less time with reduced solvent consumption than traditional normal phase separations.
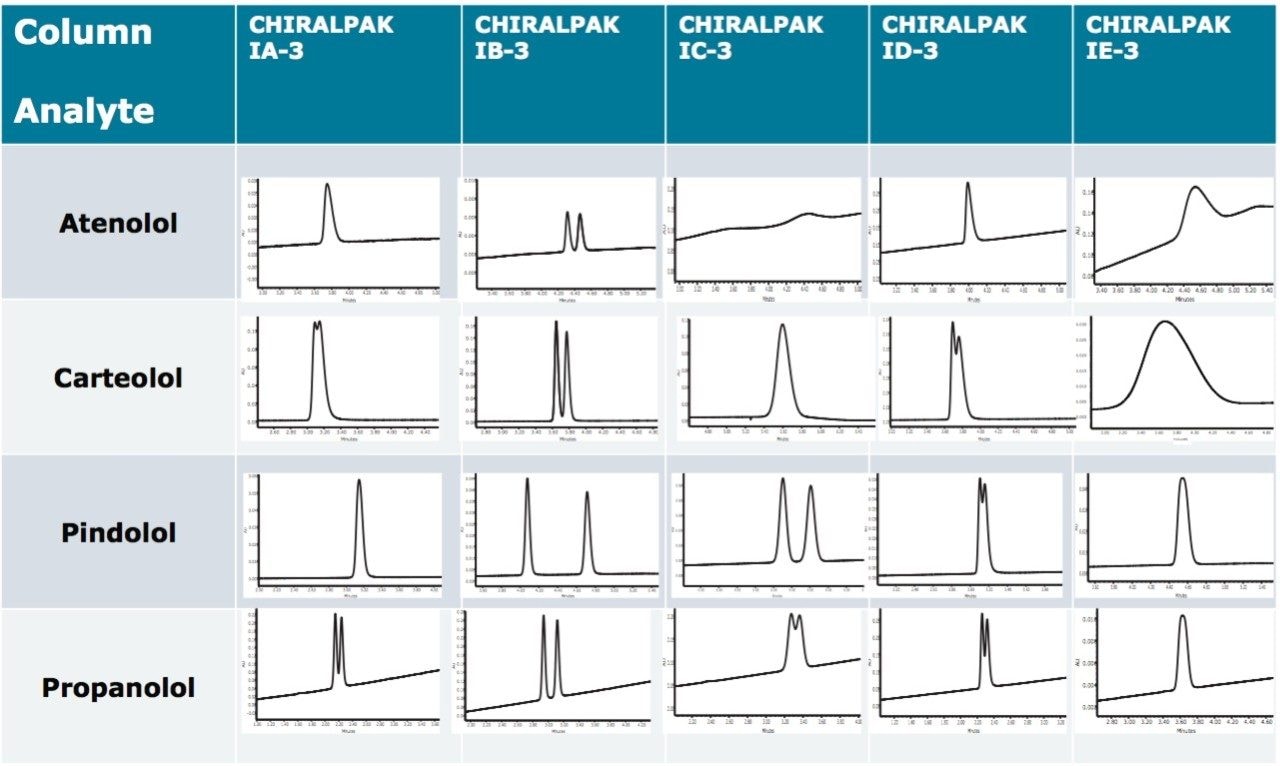
Beta blockers, a class of drugs that are typically basic, were screened across multiple columns. The use of CO2 mobile phase allowed for higher linear velocities than those used in traditional normal phase separations. Coupled with a fast gradient and 50-mm columns, the conditions facilitated screening of six columns in under 30 minutes. Methanol was used as the modifier (co-solvent) for the desired retention, and ammonium acetate was used as the basic additive to achieve the required peak shape. Evaluation of a wide range of chiral columns, as shown in Figure 1, demonstrated that the CHIRALPAK IB-3, 2.1 x 50 mm column provided the highest resolution for the beta blockers enantiomers. All other columns exhibited either a single peak or a partially resolved pair for the majority of the enantiomers.
The selected column stationary phase (CHIRALPAK IB, 2.1 x 100 mm) was then used to develop a single assay for multiple beta blockers. A mixture of multiple beta blockers was evaluated under gradient conditions using 20 mM ammonium acetate in methanol as a modifier. These conditions provided no co-elutions of the four beta blockers tested while allowing for retention times of less than seven minutes for all solutes. To improve the selectivity of the separation, the effect of column temperature was evaluated. Based on these observations, a temperature of 35 °C provided the highest resolution for the four beta blockers tested. The resulting separation provided baseline resolution for all of the enantiomers in a single run of less than six minutes.
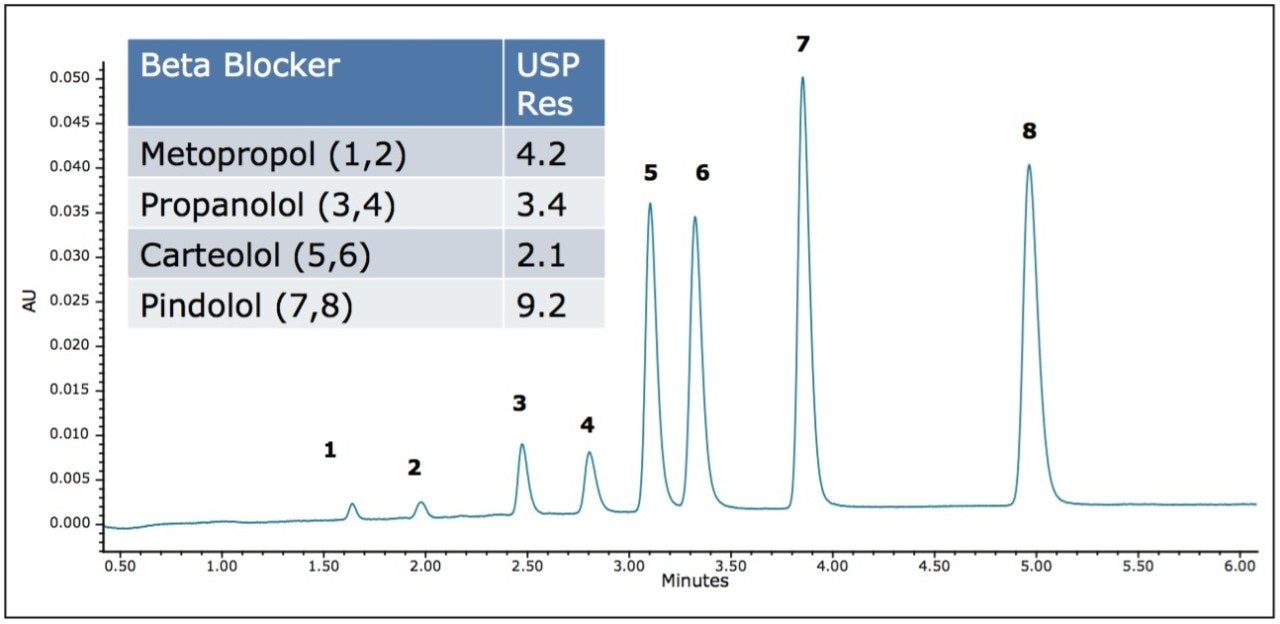
Enantiomers of pharmaceutical compounds, including beta blockers, have traditionally been analyzed under normal phase conditions. Consequently, analysis of multiple chiral compounds can be time-consuming and require extensive method development. By utilizing the ACQUITY UPC2 System in combination with a CHIRALPAK IB-3 2.1 x 100 mm column, analyses consisting of the simultaneous separation of multiple beta blockers can be completed in less than six minutes, resulting in 2x increase in throughput versus comparable normal phase separations.1
720004526, December 2012