
Xevo G2 QTof is a valuable tool for the identification and structural elucidation of extractables. MSE functionality allows simultaneous acquisition of precursor and fragment ions. Accurate mass and fragmentation information assists in the assignment of structures for many unknown compounds.
The workflow described in this application note can facilitate the daunting task of identifying the unknowns in any field that deals with structural elucidation, such as Pharmaceutical, Chemical Materials, and/or Food industries.
The Pharmaceutical industry is required by the U.S. FDA to demonstrate that no toxic or harmful substances migrate from packaging materials into a drug during its expected product shelf life.1-5 Similarly, in the Food and Cosmetics industries, there is significant interest in the investigation of packaging leachables present in their products. By definition, extractables are compounds that are extracted from packaging or device components under controlled extraction conditions. Leachables are compounds that migrate from the packaging into the product during its normal shelf life. In the ideal case, leachables are a subset of extractables. If a thorough and accurate identification – or at least compound class identification of all potential contaminants is not performed, it can lead to product recall, financial losses, and/or brand alienation for the company.6
The initial investigation, called a controlled extraction study, involves some type of solvent extraction, typically a reflux, microwave, or supercritical fluid extraction.7 The solvents chosen must cover a wide range of polarities to ensure that non-polar and polar analytes are extracted. The analytical techniques employed for analyzing extracts must be comprehensive to cover as many analytes as possible including GC-FID-MS (volatiles) and LC-UV-MS (non-volatiles).5
The challenge with the compounds observed in a controlled extraction study is their identification. Resin manufacturers rarely provide a complete list of all the additives in polymers used for packaging. The original ingredients can degrade or undergo chemical changes during the manufacturing process. Also, the resin manufacturer may not be aware of possible contaminants present within the compounds. Typical extractables include monomers and oligomers from incomplete polymerization reactions; plasticizers, stabilizers, fillers, coloring agents, antioxidants, and antistatic agents, as well as their degradants. Additionally, residues from detergents and mold release agents that can be present on the resin after the molding process.
Many of the analytes obtained from single quadrupole GC-MS data can be identified using commercially available libraries, such as NIST. However, a difficulty arises for volatiles analysis when the compound of interest is not listed in the library, or when the sensitivity of a single quadrupole MS is not sufficient for a positive identification. Therefore, additional techniques, such as Atmospheric Pressure Gas Chromatography (APGC) and Quadrupole Time-of-Flight (QTof) described in this application note, are beneficial.8 Due to the absence of libraries for LC-MS data accurate mass data would vastly facilitate the non-volatile analysis. For both volatile and semi-volatile analysis performed here, MSE data, acquisition on a quadrupole time of flight mass spectrometer, with commercially available structural elucidation tools proves to be valuable for identification of the unknown compounds.
Samples were prepared by microwave extraction. The samples of polypropylene and nylon (2 g) were extracted in 10 mL of isopropanol for 3 h at 70˚C. After the extraction the supernatant was transferred to the GC vials.
|
MS System: |
Xevo G2 QTof with 7890A GC |
|
Column: |
HP1-MS, 30 m x 0.32 mm, 1.0 μm film |
|
Carrier gas: |
He at 2 mL/min |
|
Temp.: |
35 °C for 5 min, 20 °C/min to 320 °C, hold 20.75 min |
|
Injection port: |
300 °C |
|
Injection type: |
1 μL splitless, 1 min purge |
|
Makeup gas: |
N2 at 500 mL/min |
|
Scan range: |
50 to 1,000 Da |
|
Collision ramp for MSE: |
15 to 25 eV |
|
Data management: |
MassLynx v4.1 |
Two widely available polymer materials were chosen for this study: polypropylene and nylon. In this application note, the identification of three different types of extractables is shown: an antioxidant, a monomer and a degradant of a monomer.
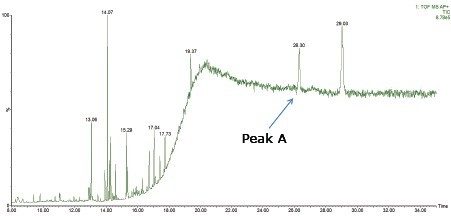
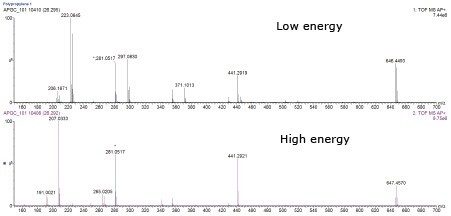
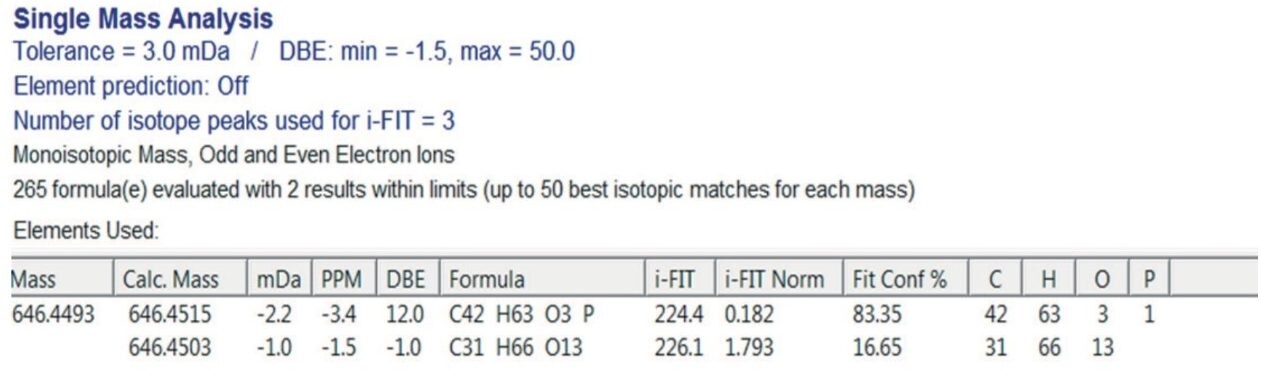
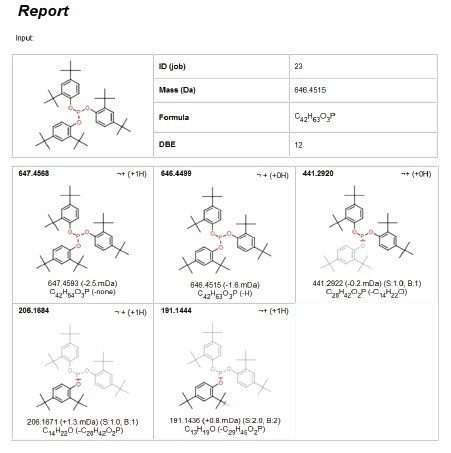
In the polypropylene sample, a peak (Peak A) was observed at a retention time of 26.3 min, as shown in Figure 1. Performing elemental composition analysis on the accurate mass APGC spectrum, shown in Figure 2, suggested a molecular formula of C43H63O3P, as shown in Figure 3. The elemental composition software calculates the possible molecular formulas for the observed mass and also uses the isotope pattern algorithm to match the observed pattern with the theoretical one for each candidate molecular formula. In this case, there are two choices shown for the ion with the second being a closer match if only mass difference is considered. However, the combination of mass difference and isotope fit brings the correct one to the top of the list.
The APGC analysis was performed under dry source conditions,9 which promotes molecular ion (M.+) formation ahead of the protonated adduct ([M+H]+). It is interesting to note that under high energy collision conditions the molecular ion fragments more easily than the protonated adduct; therefore the difference in the base peak was observed (646.4 versus 647.4) between the two channels, shown in Figure 2.
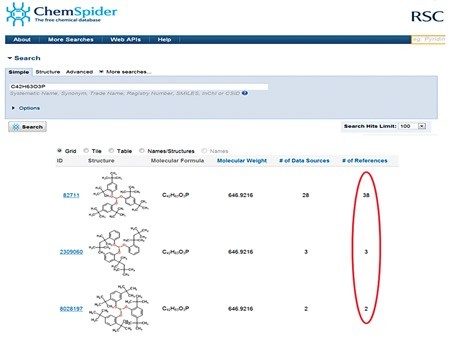
Performing a search of the proposed elemental composition formula in ChemSpider gave Irgafos 168, shown in Figure 4, as the top answer when sorted by “# of References”, as described by Little, et al.10 Irgafos 168 is a trisarylphosphite processing stabilizer and protects the resin polymer, such as polypropylene, against oxidation during resin synthesis.
Confidence in the identification was increased when another structural elucidation tool, Waters MassFragment Software, was able to match several fragments observed in the high and low energy spectra to major fragment ions of Irgafos 168, as shown in Figure 5. MassFragment identifies bonds in precursor structure and then assigns a score based on the type and likelihood of the bond breakage. In addition, the number of bonds broken is listed. The lower the score (e.g. S:1.0, B:1.0 vs. S:4.5, B:2.0) the more probable the appearance of the fragment substructure.
The next step in this workflow is to purchase a standard and compare the retention time and fragmentation pattern with the sample.
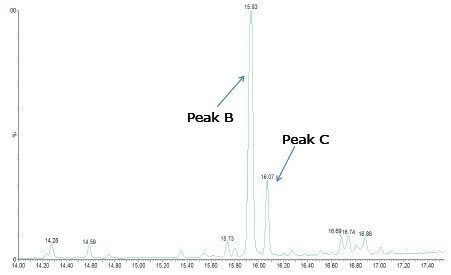
Laurin lactam is a known starting material for the manufacturing of nylon. In the nylon extract the laurin lactam monomer (Peak B) is observed at a retention time of 15.93 minutes, as shown in Figure 6. The identity of the peak was confirmed by molecular formula and MassFragment following the workflow described in the previous example. A smaller peak is observed at a retention time of 16.07 minutes (Peak C). The measured mass is consistent with a molecular formula of C12H21NO, shown in Figure 7, which indicated that the peak was likely a laurin lactam degradant with an extra double bond in the molecule (laurin lactam monomer is C12H23NO). The parent ions in each spectra were confirmed by the presence of the in-source dimers (2M+H). For laurin lactam the observed dimer has m/z 395.3652 and for the degradant it is m/z 391.3324.
![Spectra and molecular formula [M+H]+ for Peaks B and C.](/content/dam/waters/en/app-notes/2012/720004391/720004391en-f7-spectra-and-molecular-formula-b-and-c.jpg.82.resize/img.jpg)
The ChemSpider search for C12H23NO showed laurin lactam as the second top choice. The search of C12H21NO did not provide any appropriate match based on the known compounds in the polymer.
Since a standard of this degradant is not likely to be available, the Xevo G2 QTof data allowed the assignment of a structure to this compound. It is not possible to determine the exact location of the double bond on the laurin lactam ring. However, in these types of studies it is not always necessary to determine an exact structure. It is sufficient if the compound’s class has been identified. It was clear that the degradant is related to laurin lactam, therefore its toxicological profile was expected to be similar.
720004391, June 2012