
This application note describes an optimized method for determination of paraquat and diquat in drinking water using an Oasis WCX cartridge for SPE prior to fast and sensitive UPLC-MS/MS analysis.
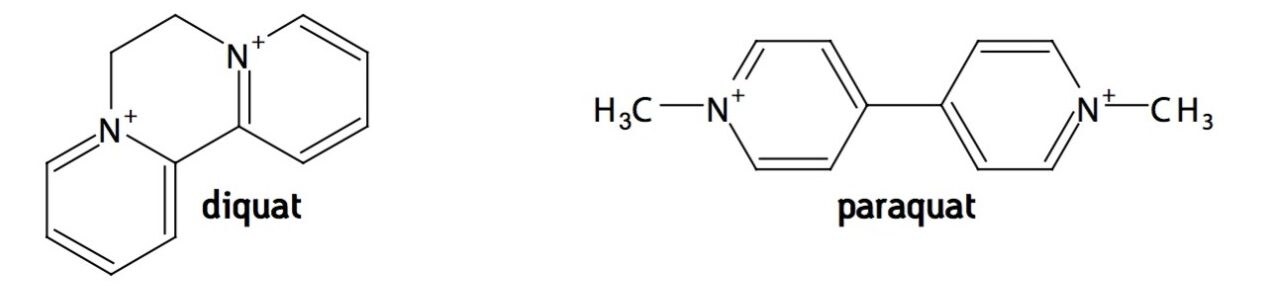
Paraquat and diquat are quaternary ammonium herbicides used in great quantities worldwide for defoliation and weed control (see Figure 1). In the United States alone, paraquat use was 4 million pounds and diquat use was 20,000 pounds in 20021. Because these compounds are potentially toxic to wildlife and to humans, groundwater and surface water samples are monitored to assure that residue levels meet safety standards. These limits range from 100 ng/L in Europe to 20 μg/L in the United States. This application note describes an optimized method for determination of paraquat and diquat in drinking water using an Oasis WCX cartridge for SPE prior to fast and sensitive UPLC-MS/MS analysis. Oasis WCX is a mixed-mode weak cation exchange sorbent that is effective for isolation of paraquat and diquat from aqueous samples. A prior publication discussed HPLC-MS determination of quats after SPE with Oasis WCX2. The new UPLC method requires half the analysis time compared with the HPLC method. Also, the SPE has been improved; the earlier protocol utilized trifluoroacetic acid, a toxic perfluorinated compound and persistant organic pollutant. In the new SPE protocol, formic acid is used as an acidic modifier for the SPE eluent.
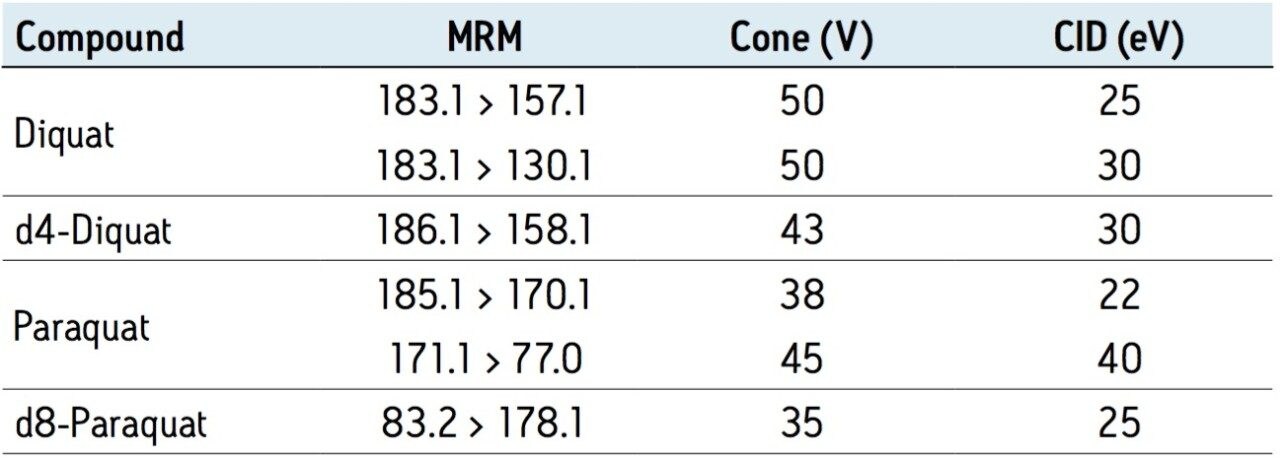
|
LC System: |
ACQUITY UPLC H-Class system |
|
Column: |
ACQUITY BEH HILIC |
|
Mobile Phase (Isocratic): |
40:60 A/B |
|
Mobile Phase A: |
150 mM ammonium formate buffer (pH 3.7) |
|
Mobile Phase B: |
Acetonitrile |
|
Injection Volume: |
10 μL |
|
Column Temperature: |
30 °C |
|
Weak Needle Wash: |
50:50 methanol/water (600 μL) |
|
Strong Needle Wash: |
50:50 methanol/water (600 μL) |
|
Seal Wash: |
10:90 acetonitrile/water |
|
Flow Rate: |
0.50 mL/min |
|
MS System: |
TQD |
|
Ionization Mode: |
Positive Electrospray |
|
Source Temperature: |
150 °C |
|
Desolvation Temperature: |
350 °C |
|
Desolvation Gas Flow: |
800 L/Hr |
|
Cone Gas Flow: |
30 L/Hr |
|
Collision Gas Flow: |
0.20 mL/Min |
|
Data Management: |
MassLynx v4.1 |
Three types of water samples were chosen to demonstrate this new method. Chlorinated tap water was obtained from a local municipal source, groundwater was obtained from a commercial bottled source, and untreated surface water was obtained from a local river. All samples were adjusted with phosphate buffer to pH 7 prior to SPE. Municipal drinking water samples were dechlorinated with sodium thiosulfate prior to SPE. The Oasis WCX cartridge was then used for effective enrichment and cleanup prior to UPLC/MS(MS) analysis.
Accurately weigh 20.9 g of potassium phosphate dibasic and 10.9 g of potassium phosphate monobasic into a 500 mL volumetric flask. Add sufficient reagent water (Milli-Q or equivalent) to completely dissolve and then dilute to the mark reagent water. Check pH—if necessary, adjust to 7.0 using very dilute KOH or H3PO4 aqueous solution.
Mix 200 mL acetonitrile, 200 mL isopropanol, and 100 mL formic acid (caution—use only with appropriate personal protective clothing and eyewear).
Take 25 mL of the 400 mM pH 7 buffer and dilute to 400 mL with reagent water.
Accurately weigh 1.6 g of ammonium formate into a 1 L volumetric flask. Add sufficient reagent water to completely dissolve and dilute to approximately 900 mL. Add 1.4 mL of 30 % ammonium hydroxide and mix well. Adjust to exactly pH 8 with dilute formic acid or ammonium hydroxide as needed. Dilute to mark with reagent water.
Note: Polypropylene containers should be used for sample collection and for all sample preparation steps. Polypropylene autosampler vials (part number 186002642) are recommended for UPLC analysis.
Transfer a 10 mL sample to an appropriate polypropylene container (15 mL centrifuge tubes were used for this study). For chlorinated samples add 10 mg of sodium thiosulfate and mix well. For all samples, adjust pH by addition of 25 µL of 400 mM pH 7 phosphate buffer.
Perform SPE enrichment and cleanup using Oasis WCX cartridges (see SPE details in Figure 2). To allow convenient loading of the 10 mL sample, attach a 30 cc polypropylene reservoir (part number WAT011390) to each cartridge.

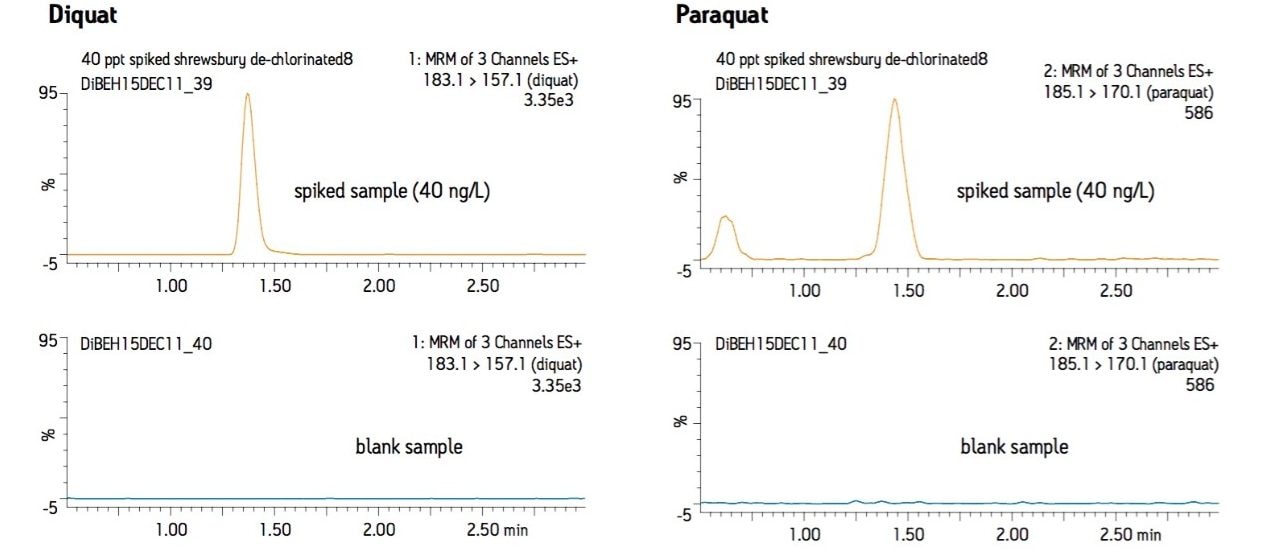
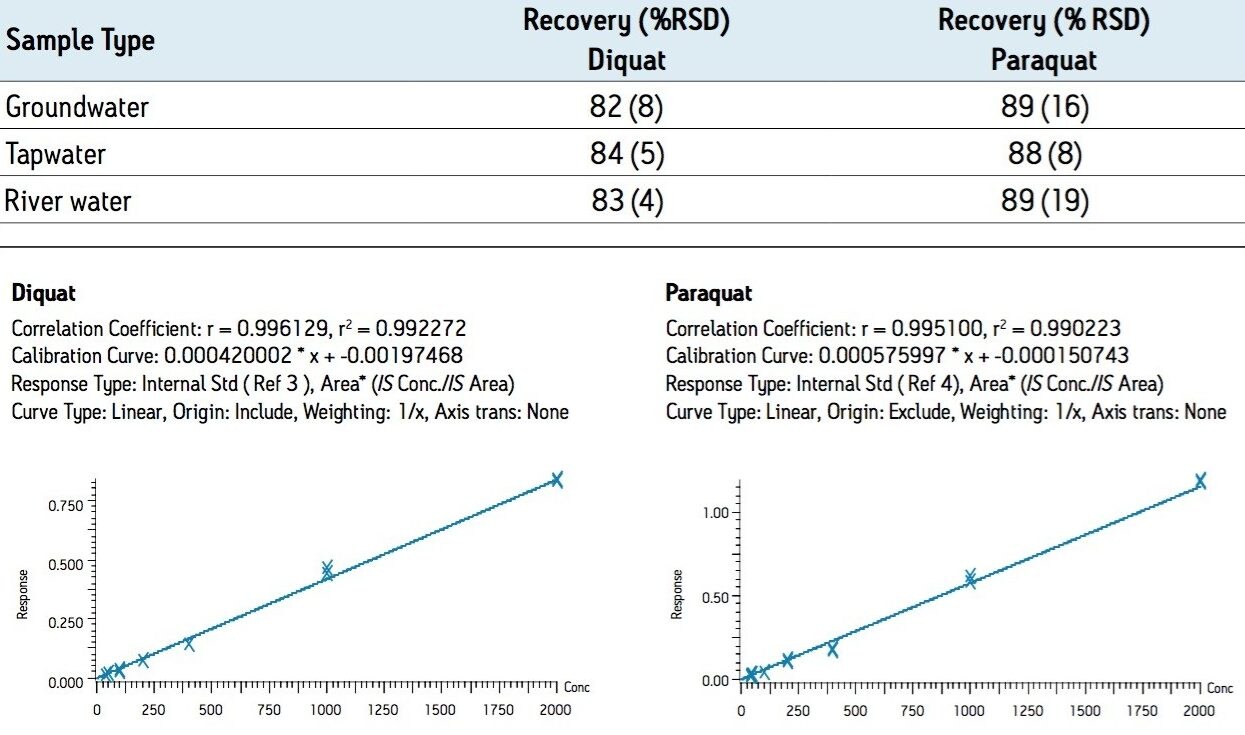
Figure 3 shows typical LC-MS chromatograms obtained from analysis of a matrix-matched standard of diquat and paraquat at 40 ng/L prepared in dechlorinated municipal water. Performance was similar for the other water types. Table 2 shows the recovery data obtained from replicate analysis of water samples spiked at 40 ng/L. Typical matrix-matched calibration curves were linear in the range from 40–2000 ng/L for all water types. Representative calibration curves are presented in Figure 4.
720004220, January 2012