
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the importance of sample preparation in complex matrices using Oasis HLB and the Xevo TQD with RADAR functionality.
Oasis HLB is a unique sorbent that enables simpler methods for SPE with proven results.
Development of analytical methods for the analysis of contaminants in food is often complicated due to co-extracted interferences arising from the complex matrices. To prevent or minimize matrix effects, sample preparation prior to LC-MS/MS analysis is critical. For low levels of detection in challenging matrices such as animal tissue, multiple steps are usually required for the sample preparation protocol. Liquid-liquid extraction (LLE) and solid phase extraction (SPE) are frequently combined to remove co-eluting matrix components that can compete with the analyte of interest during the ionization process. Waters Oasis HLB is an SPE sorbent with unique properties derived from its copolymer structure. Because of its excellent selectivity, high capacity, reproducibility, and ease-of-use, Oasis is the SPE sorbent of choice for many scientists when extracting analytes from complex matrices.
The Xevo TQD with RADAR provides an innovative solution to assist in the method development process because it simultaneously monitors MRMs (multiple reaction monitoring) and full scan background data. This acquisition mode offers the ability to monitor not only the quantification trace, but also the background interference simultaneously. With this information, rapid decisions can be made about the optimization of the sample preparation protocol and method development can progress more quickly.

The sample preparation protocol for chloramphenicol analysis in chicken muscle involves a series of steps. Homogenized chicken tissue was extracted with ethyl acetate followed by centrifugation. The supernatant was evaporated to dryness under nitrogen at 40 °C. The residue was re-dissolved in methanol and mixed with sodium chloride solution, and then de-fatted using hexane. The hexane layer was discarded and the aqueous portion was subjected to SPE cleanup using Oasis HLB. The SPE eluate was evaporated to dryness, reconstituted in water, and filtered prior to injection onto an ACQUITY UPLC System, coupled to the Xevo TQD. The data were acquired using RADAR, which enables the simultaneous acquisition of MRMs and full scan data.
The background matrix was monitored to assess the sample preparation process and observe any co-extracted interferences that could impact the detection and quantification of chloramphenicol. The extract before and after the Oasis HLB SPE step was analyzed by UPLC-MS and UPLC-MS/MS simultaneously using the RADAR acquisition mode. Figure 2A shows an MRM chromatogram of chloramphenicol in solvent, with a retention time of 0.82 min. Base peak intensity (BPI) chromatograms from RADAR acquisitions are shown for a blank chicken muscle extract following LLE with and without Oasis HLB cleanup, as shown in Figures 2B and 2C. Figure 2B reveals a co-extracted matrix interference at a retention time of 0.82 min. The mass spectrum of this peak is shown in the inset. Following Oasis HLB cleanup, this interfering compound is no longer detected, as shown in Figure 2C, indicating that the matrix component that co-eluted with the analyte was successfully removed.
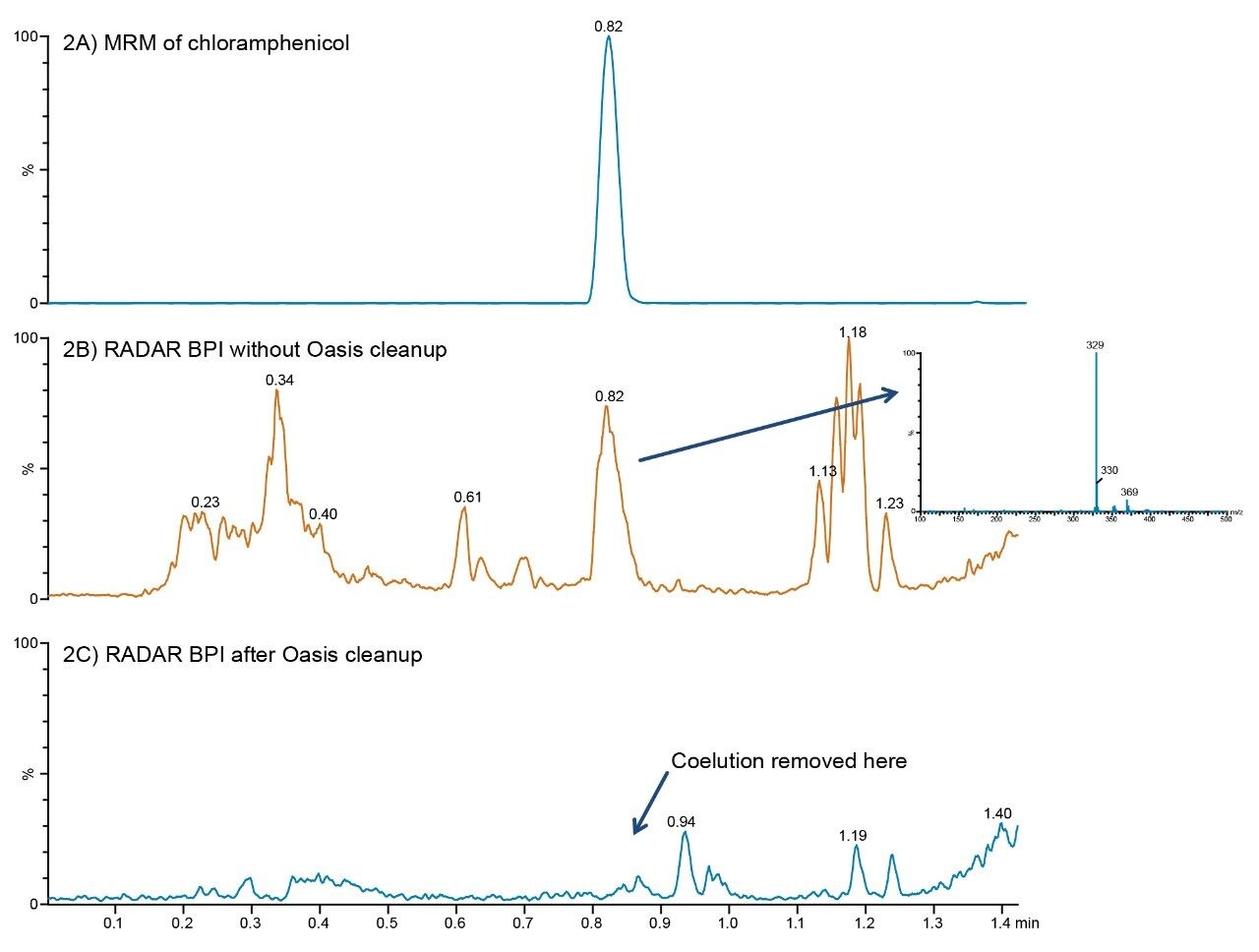
The ability to constantly acquire full scan background data while developing quantitative studies means that any changes in the matrix can be monitored and assessed. With complex matrices, such as the animal tissue in this study, the ability to screen the background for any variation is invaluable.
Sample preparation for removing contaminants in complex matrices is one of the most important but time-consuming steps. In this technical brief, tools have been demonstrated that help minimize method development time for this crucial step and at the same time, ensure the best possible method performance. Oasis HLB is a unique sorbent that enables simpler methods for SPE with proven results. The combination of the ACQUITY UPLC System with the Xevo TQD offers an innovative solution for the quantification of analytes of interest, while monitoring background interferences. This enables informative decisions to be made during method development and continuous monitoring of the matrix background to ensure a robust and reliable method.
720003979, May 2011