
This application note highlights the utility of the ACQUITY UPLC system when used with an ACQUITY UPLC BEH column. The combined system provides accurate, reproducible, and rapid separation of oligonucleotides. It also illustrates that analysis times can be dramatically reduced by scaling both the gradient slope and flow rate proportionately, thus yielding constant gradient slope per volume of mobile phase.
We highlight the utility of the ACQUITY UPLC System when used with a 1.7-μm column. The platform provides accurate, reproducible, and rapid separation of oligonucleotides. The method also illustrates that analysis times can be dramatically reduced by scaling both the gradient slope and flow rate proportionately, thus yielding constant gradient slope per volume of mobile phase.
There is an increased desire to perform rapid and accurate analysis of oligonucleotides. The ACQUITY Ultra Performance LC (UPLC) System combined with Waters Oligonucleotide Separation Technology (OST) Columns packed with 1.7-μm sorbent as shown here allows for both rapid and accurate analysis that is not possible using conventional HPLC.
Conventional HPLC methods for the analysis of oligonucleotides generally suffer from low resolution of oligonucleotides, particularly when analysis times are reduced. The UPLC method presented here expands on the topic discussed previously in application note, “UPLC-MS Separation of Oligonucleotides in Less Than Five Minutes: Method Development,”1 which provides guidelines for the rapid analysis of RNAi and DNA in less than 2.5 minutes in most cases.
OST MassPREP Standard was dissolved in 100 mM TEAA. RNAi and DNA samples (IDT Technologies) were prepared by partial digestion of purified samples with phosphodiesterase II (Sigma).
|
LC System: |
Waters ACQUITY UPLC System with ACQUITY UPLC PDA Detector |
|
Column: |
ACQUITY UPLC OST C18, 2.1 x 50 mm, 1.7 μm |
|
Column temp.: |
60 °C |
|
Flow rate: |
0.2 to 0.8 mL/min |
|
Mobile phase A: |
100 mM hexylammonium acetate, pH 7.0 |
|
Mobile phase B: |
Acetonitrile |
|
Gradient: |
28 to 39 %B |
|
Detection: |
PDA UV 260 nm |
Ion-pairing reversed phase (IP-RP) liquid chromatography is a commonly accepted separation strategy for the analysis of oligonucleotides. Of the known ion-pairing systems, triethylamine hexafluoroisopropanol (TEA/HFIP) is commonly used due to its MS compatibility and impressive resolving power. Additionally, we utilized hexylammonium acetate (HAA), which also offers exceptional resolution.
We developed a method for the analysis of up to 35-mer oligonucleotides in under 2.5 minutes and oligonucleotides up to 25 mer in under 2 minutes with impressive resolution. This was accomplished by establishing appropriate UPLC conditions to successfully elute all species in under 10 minutes at a flow rate of 0.2 mL/min, which corresponds to a total flow of 2 mL. We then increased the flow rate incrementally while maintaining a constant gradient slope. By scaling the overall analysis time so that the gradient was completed within the same pumped volume, we achieved a constant gradient slope per column volume. As shown in Figure 1, the ACQUITY UPLC coupled with an ACQUITY UPLC OST column offers exceptional resolution at all flow rates tested.
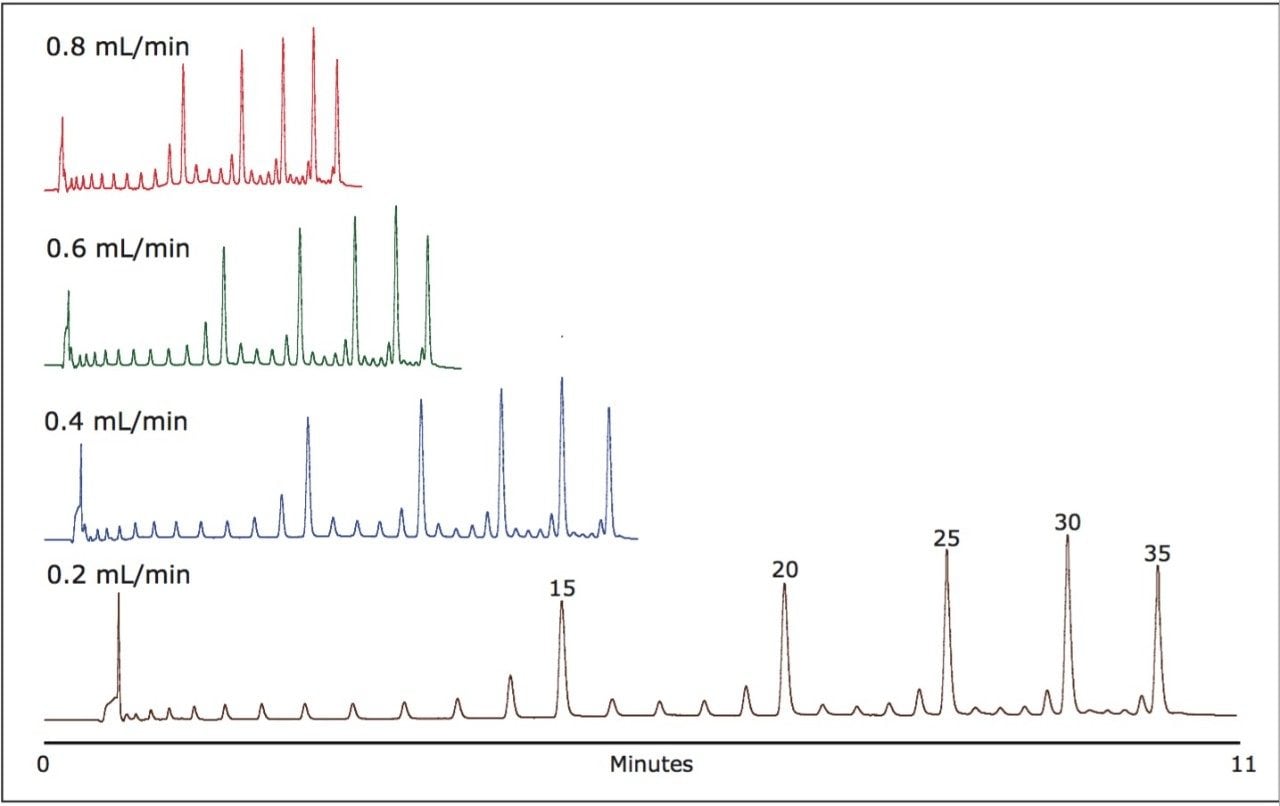
Additionally, system pressure increased significantly as the flow rate increased, ranging from 3200 psi at 0.2 mL/min to 12,000 at 0.8 mL/min. These pressures far exceed those possible with conventional HPLC but are accomplished routinely using the ACQUITY UPLC System.
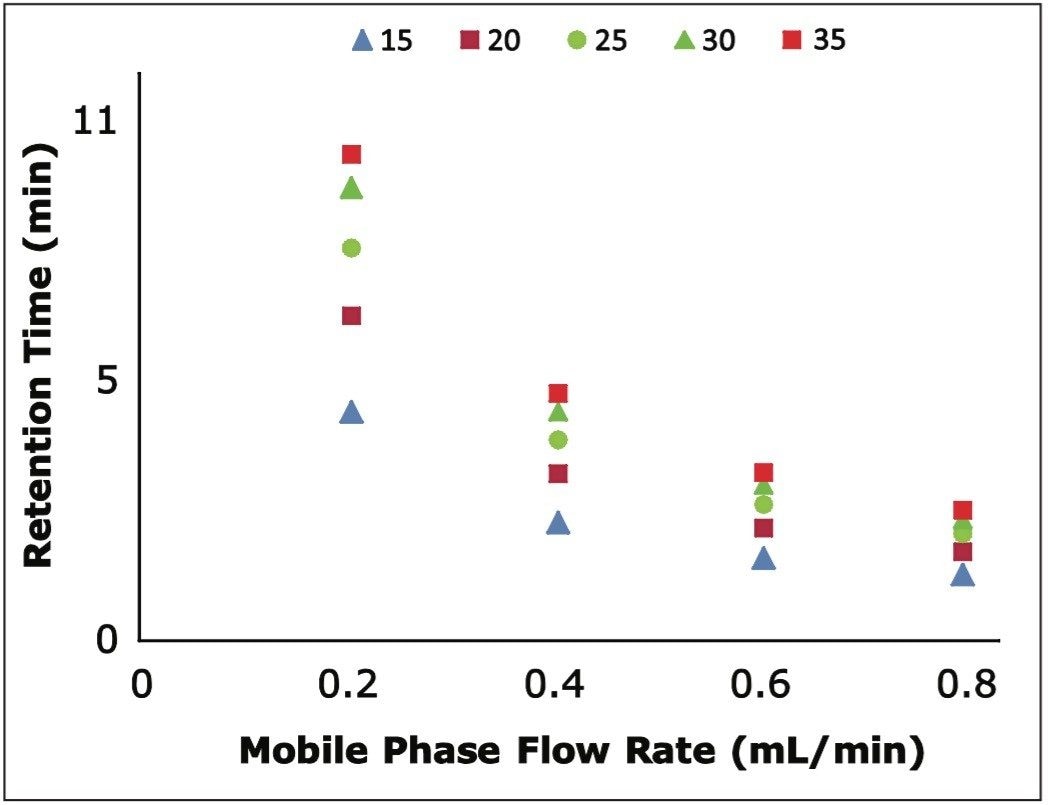
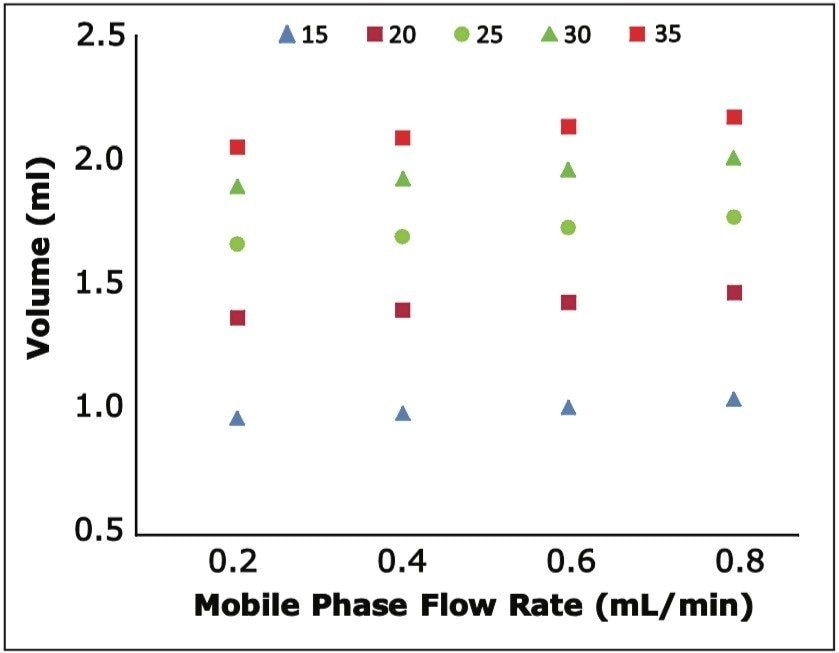
To determine the reproducibility of retention time as a function of gradient slope, we determined the volume of eluent necessary to elute each oligonucleotide at the flow rates tested. We found exceptional reproducibility of delivered volume to oligonucleotide elution as shown in Figure 3. This highlights the selectivity of our system for oligonucleotides, regardless of flow rate.
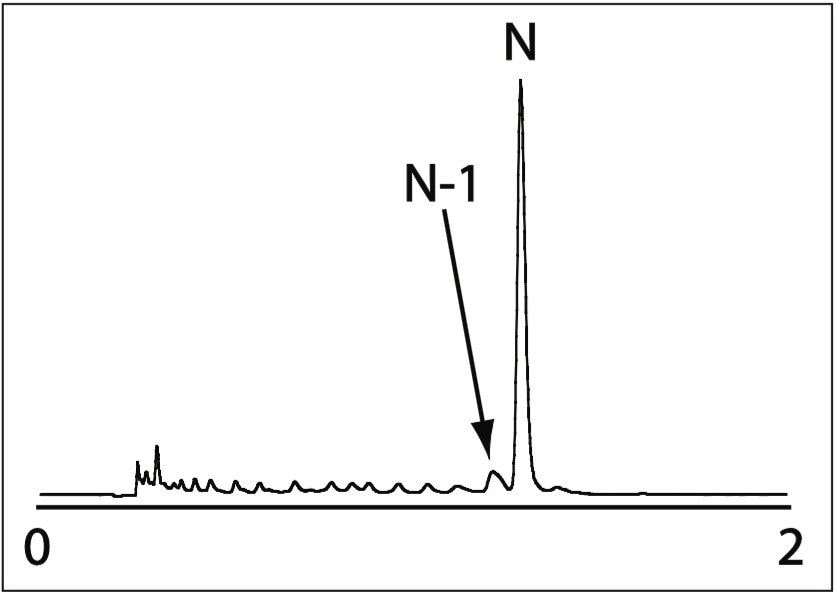
We tested our method with a more challenging sample, a heteromolecular DNA sequence, Figure 4. Our rapid separation method provided regular retention based on oligo length with impressive resolution from failure sequences.
The method presented in highlights the utility of the ACQUITY UPLC system when used with an ACQUITY UPLC BEH column. The combined system provides accurate, reproducible, and rapid separation of oligonucleotides. The method also illustrates that analysis times can be dramatically reduced by scaling both the gradient slope and flow rate proportionately, thus yielding constant gradient slope per volume of mobile phase. This is accomplished by using sub-2-micron particles and the high pressure capabilities of the ACQUITY UPLC. The method provides accurate and predictable retention of both homo- and hetero-molecular oligonucleotides.
720003386, March 2010