
This application note demonstrates how the use of Ultra Performance LC (UPLC) delivers better resolving power than HPLC for the same run times, resulting in resolution of matrix from analyte and retaining productivity and also greater sensitivity in the same analysis time.
LC-MS/MS in bioanalytical assays is widely accepted as the technique for highly sensitive and selective analysis to monitor and quantify pharmaceutical compounds in biological matrices. Using MS/MS multiple reaction monitoring (MRM) transitions for specific analytes results in very fast chromatography or ballistic gradients, making this a popular choice. The analyst is dependent on the MS technology for specificity on the assay rather than the chromatographic process. The separation and the fast elution profiles mean higher throughput for the assay. However, the possibility of ion suppression or enhancement from other matrix components means matrix effects must be removed by sample preparation or minimized by managing the chromatographic separation.
In this application note, we discuss how the use of Ultra Performance LC (UPLC) delivers:
Using the unique functionality of the Xevo TQ mass spectrometer, RADAR, which provides the ability to capture both full-scan and MRM data at the same time, we were able to observe potential matrix coelutions. This allowed us to monitor the matrix and analyte during the course of the runs without compromising MRM sensitivity.
Protein precipitation is a common method of sample preparation for biological matrices because it is relatively cheap and fast. However, it only removes proteinaceous material. Phospholipids as well as other interferences are still present in the sample, which may be the result of lifestyle, genotype, or phenotype, factors such as formulation agents, or other medications. These matrix components may be unpredictable and can affect the ruggedness and sensitivity of an assay.
Chromatographic resolution can help mitigate the impact of matrix. However, when using conventional HPLC, greater resolution from other analytes and interferences can often only be gained through use of longer, more shallow gradients or longer columns, which will compromise productivity goals. UPLC utilizes sub-2-μm particles that enable higher chromatographic efficiency for greater matrix interferences resolution without compromising productivity.
These experiments were designed to compare fast HPLC chromatography using 3.5-μm column particle packing against 1.7-μm UPLC. The chromatographic conditions for both experiments were identical in terms of sample turnaround, resulting in the same analytical run time and therefore the same productivity.
The samples, propranolol spiked into rat plasma, were treated with acetonitrile (2:1) to precipitate proteins, centrifuged, and the supernatant injected on to each column configuration. Both the 3.5-μm column and the 1.7-μm column were run on an ACQUITY UPLC System, which, because of its low system volume, enhanced performance of each analytical column.
Mass spectrometry was carried out on a Xevo TQ MS using the MRM transition 260 > 183 to monitor propranolol. The matrix was monitored at the same time by acquiring full-scan data over the mass range m/z 50 to 500 using the RADAR functionality.
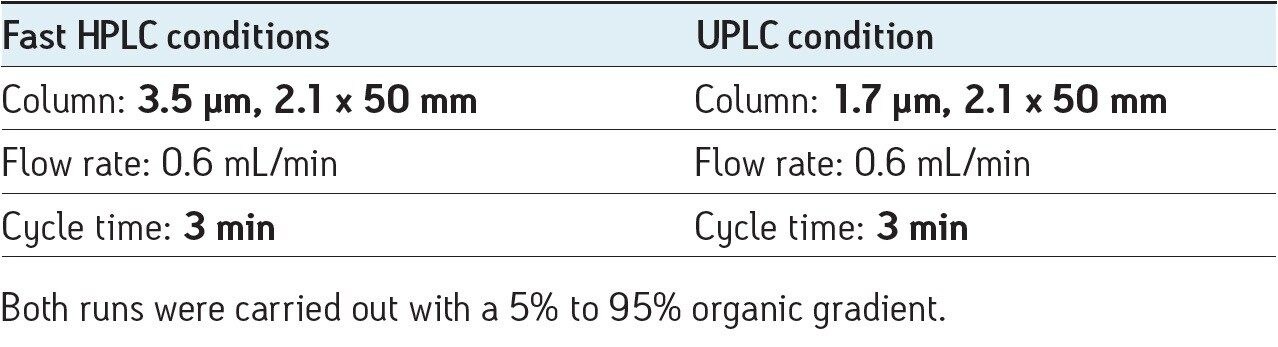
When the runs were compared (Figure 1), the retention time for propranolol and cycle times for fast HPLC and UPLC were very similar. In terms of productivity there was no difference between the two techniques. A comparison of peak height showed a better (~3X) response for the sharper UPLC propranolol peak.
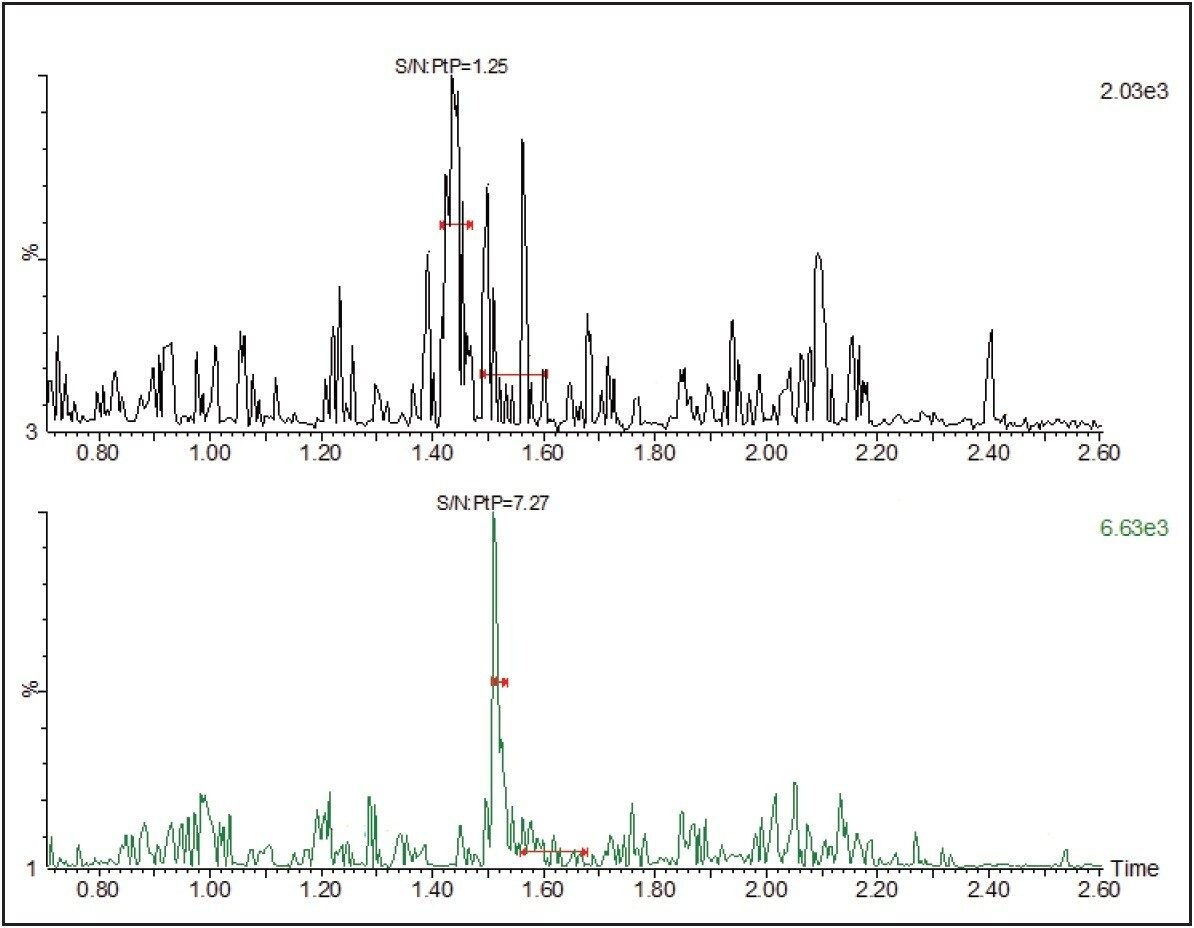
Furthermore, when signal/noise was compared, the UPLC had a 5X improvement over the HPLC peak. Such improvements in signal/noise will significantly improve the LLOQ of a bioanalytical assay. Additional benefits of such sharp peaks include the ease and consistency of detecting the beginning, apex, and end of a peak for integration purposes, leading to greater accuracy and precision for quantitation.
In each case, propranolol elutes in the middle of the chromatogram, well away from the void. It is also likely that the analyte is resolved from the phospholipids that are not removed through protein precipitation and tend to elute in high organic concentrations at the end of the chromatogram. The matrix also contains many other endogenous components. Any of these may interfere with the ionization of the analyte in the source and result in inaccurate quantitation. Detecting these interferences with conventional mass spectrometry when using MRM transitions for the detection of a specific analyte is time consuming.
The Xevo TQ MS employs a unique collision cell design that addresses the problem of observing matrix interferences while carrying out MRM quantitation of analytes. The collision cell is constantly filled with gas, which allows the mass spectrometer to quickly switch between full-scan and MRM modes. By making use of this extended functionality of the Xevo TQ MS, further observations and comparisons can be made for each run, allowing faster and more effective method development. In each case, a full scan was also carried out while the MS collected MRM data. The results are shown in Figures 2 and 3.
Figure 2 shows the MRM transition for propranolol and full-scan data from the HPLC analysis.
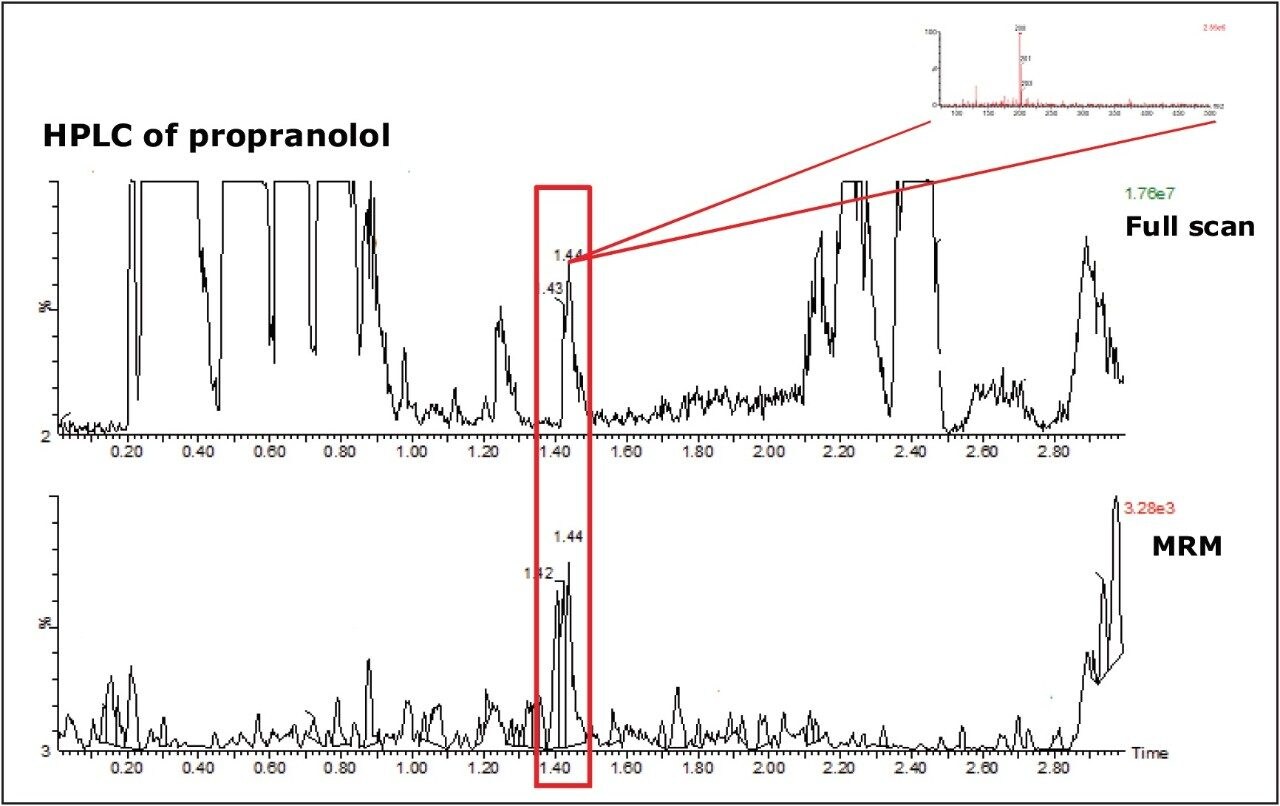
The full-scan data from the run shows that an interference coelutes with propranolol under fast HPLC conditions. We are able to mine the data further to retrieve the spectra of the peak eluting at 1.44 min and establish that the most abundant component of the coeluting peak had a m/z of 200 (inset).
Figure 3 shows the MRM trace for propranolol and full-scan data from the UPLC run. The smaller particle-size columns show greater chromatographic efficiency in the separation, leading to more resolution between components in the matrix. In the UPLC full-scan data even the matrix components are more resolved.
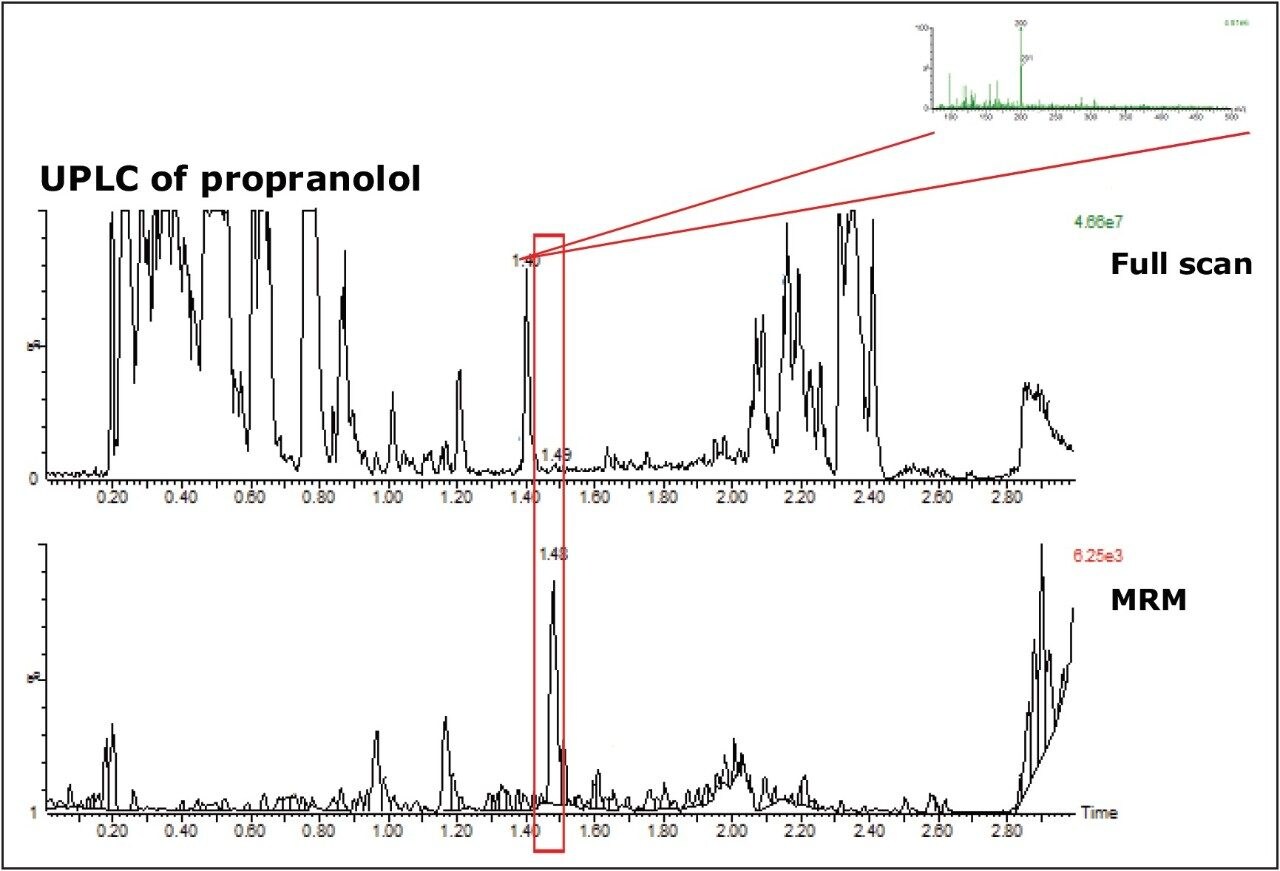
The full-scan data from the UPLC run shows the propranolol peak is fully resolved from interferences, in particular the peak that coelutes in the fast HPLC run. The spectra for the peak eluting at 1.4 min (inset) was very similar to the coeluting peak in the HPLC run, indicating that the interference is resolved away from the propranolol peak in the UPLC run.
In the UPLC run we are confident that, under these conditions, the increased resolution has resulted in interference-free chromatography. We have not compromised our productivity goals and have gained a factor of 5X sensitivity.
When fast chromatography is required to meet productivity goals:
720003464, June 2010