This is an Application Brief and does not contain a detailed Experimental section.
This application demonstrates the rugged, high-resolution separations of proteins using reversed-phase techniques, four-solvent blending, Protein Separation Technology columns, and the ACQUITY UPLC H-Class Bio System.
A robust separation of intact proteins is readily achieved with this biocompatible UPLC system that streamlines use of desired mobile phase combinations.
The characterization and analysis of protein samples must be sensitive to chemical differences in a molecule that, while but a small fraction of the large molecule, can have large biological effects. The general approach to this analytical challenge is to use an array of separation techniques, each sensitive to a specific physical or chemical property. The ACQUITY UPLC H-Class Bio System was designed to be suitable for use with all the common protein analysis techniques, including ion exchange, size exclusion, HILIC, and reversed phase. The experiments described here focus on reversed phase analysis of large, intact proteins.
Reversed phase separations of proteins typically use wide pore columns with short chain bonded phases. The ACQUITY UPLC BEH 300 C4, 1.7-μm column was selected for these experiments. The samples are eluted with a gradient of increasing organic solvent in the presence of a polar acid modifier. There are several choices in identity and concentration for both the solvent and the modifier. We can manage these choices by using the four-solvent blending capability of the ACQUITY UPLC H-Class Bio System’s quaternary solvent manager to prepare the different desired mobile phase combinations on-demand from bottles of pure solvents and concentrated modifiers. This Auto•Blend Plus Technology is only useful if it reliably produces the programmed mobile phase gradient accurately and precisely. The experiments described here focus on that system performance.
A robust method for protein analysis must include an instrument, a column chemistry, and the set of operating conditions. For developing the separation discussed here, UPLC technology was chosen as the underlying principle. The use of sub-2-μm particle packing materials on instruments that minimize dispersion ensures the best resolution, sensitivity, and sample throughput. The ACQUITY UPLC family of instruments provides the required performance.
The ACQUITY UPLC H-Class Bio System was chosen for these experiments due to its biocompatible materials of construction, and because its four-solvent blending combines with Auto•Blend Plus Technology for easy methods development and execution. The UPLC Protein Separation Technology BEH300 C4, 1.7-μm column was selected for the separation because its combination of large pores, short chain bonded phase, and inert base particle gives the best resolution and recovery. Auto•Blend Plus Technology was used to find the best combination of modifier concentration and organic solvent. The convenience of method development with Auto•Blend Plus requires a high degree of accuracy and precision in solvent blends over a long series of runs. The selected four-solvent method and the resultant chromatogram are shown in the accompanying figure.
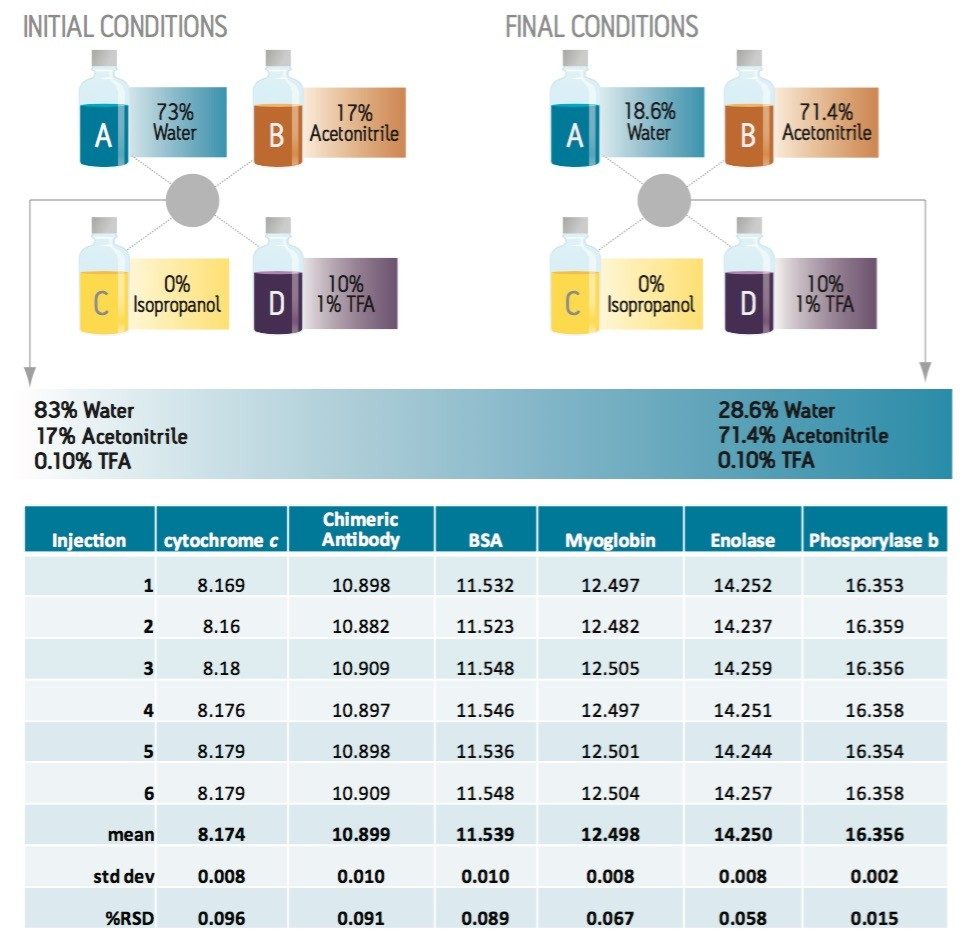
The accompanying table shows the reproducibility of retention times for this protein mixture. The proteins represent a wide range of properties and sizes, providing a good test of separation consistency. The reproducibility of the separation is suitable for routine use. The application of Auto•Blend Technology for preparation of blended mobile phases reduces labor and the possibility of error while providing reliable results.
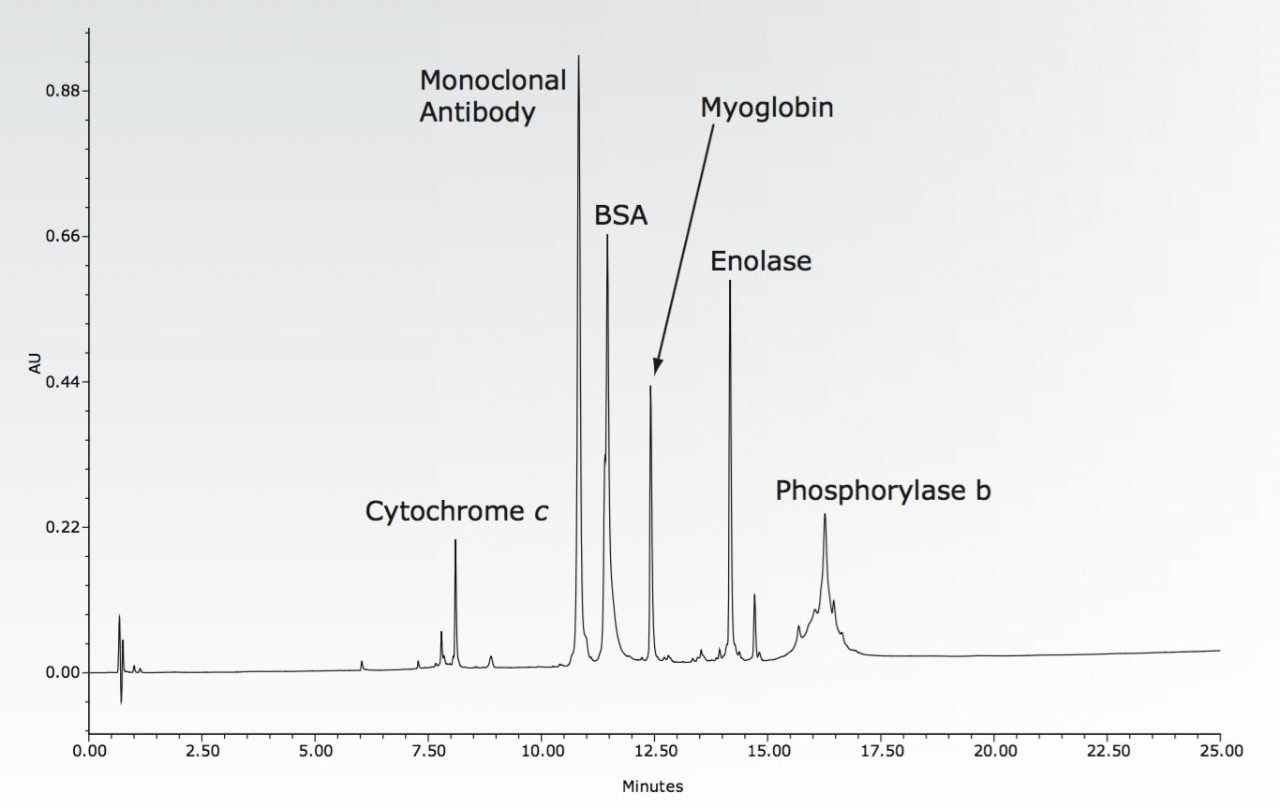
The requirements for a reversed phase analysis of intact proteins are satisfied by the system solution that combines ACQUITY UPLC H-Class Bio instrumentation, UPLC column chemistry, and software. The robustness of the system includes simplified mobile phase preparation, and is demonstrated with reproducibility of the separation.
720003646, July 2010