In this application note, various U.S. Pharmacopeia (USP) compendial methods are used as examples to highlight a new method translation strategy to facilitate the transfer of methods to and from any LC-based instrument with ease.
Pharmaceutical research and development (R&D) organizations were early adopters who recognized the many benefits of UltraPerformance LC (UPLC) Technology including resolution, sensitivity, throughput, and productivity as compared to HPLC. Today, the number of projects involving new drug entities are increasingly performed utilizing UPLC.
Adopting UPLC for R&D activities is less complex than for laboratories involved with routine analysis, where its use requires consideration about the need to re-file methods for existing products. Routine analysis areas such as Quality Control (QC) laboratories own a vast supply and variety of HPLC instrumentation. Asset procurement regarding new technologies within these groups often requires convincing financial as well as scientific justification.
Although information illustrating UPLC’s return on investment (ROI) for solvent consumption and analysis per unit time can be convincing for R&D, the QC environment requires key practical-use considerations. Managers and end users within QC laboratories require new instrumentation to provide dual purposes: first, the ability to perform both legacy methods and, second, the ability to use sub-2-μm particle columns and methodology in a routine analytical environment without complications. UPLC’s adoption must also strategically provide seamless integration within current laboratory practices and decrease learning curves of the end users.
In this application, various U.S. Pharmacopeia (USP) compendial methods are used as examples to highlight a new method translation strategy to facilitate the transfer of methods to and from any LC-based instrument with ease.
References to LC conditions are addressed as per USP Monographs, whereas specific utilization of LC instrumentation for each application is discussed in the figure captions.
Empower 2 CDS
Successful method translation requires understanding three key chromatographic attributes before implementation. The analyst must consider the differences between LC instrumentation, column selectivity, and the resolving capability of the original methodology versus the target methodology. By understanding these three essential aspects of method translation, the benefits of increasing productivity and decreasing costs while maximizing asset utilization of present and future instrumentation can be realized.
The QC laboratory frequently utilizes a variety of LC instruments for API and drug product analysis. Therefore, instrumentation flexibility is essential. Direct transfer of methods to newer technology may result in retention time and selectivity differences that may be related to decreases in instrument dwell volume.
To illustrate the flexibility provided by the Waters ACQUITY UPLC H-Class System, the USP method for galantamine hydrobromide and related substances was performed on an HPLC instrument (Figure 1). USP system suitability requirements for the related substances assay specify USP tailing of galantamine NMT 2.0 and a resolution of galantamine and 6-alphagalantamine NLT 4.5. When utilizing the same HPLC column on each instrument, the ACQUITY UPLC Columns Calculator (Figure 2) can be used to calculate the differences in the instrument dwell volume. The resulting data yielded no compromise in chromatographic integrity during the translation of the method for use on a UPLC instrument of less dwell volume (Figure 3).
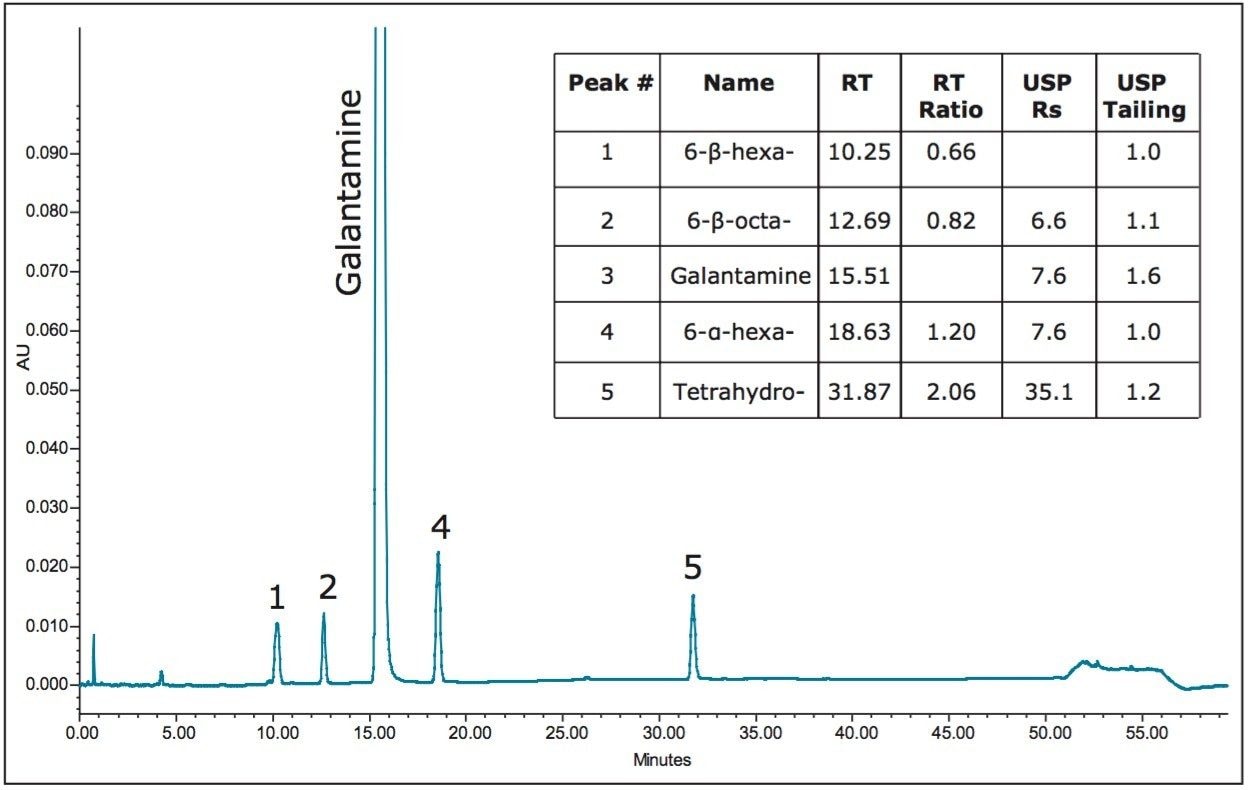
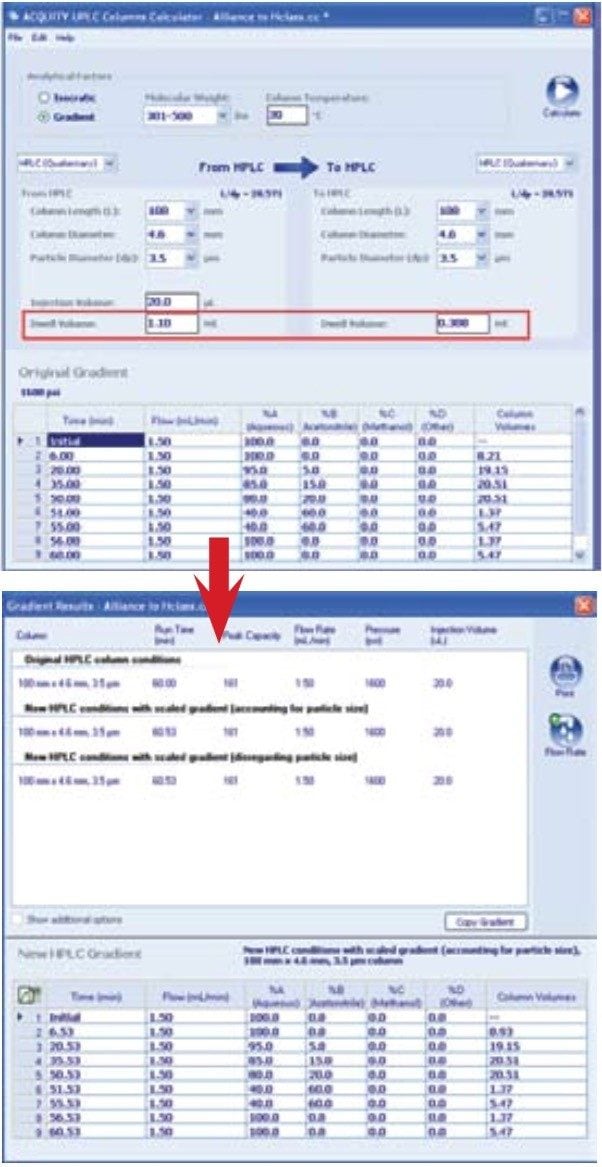
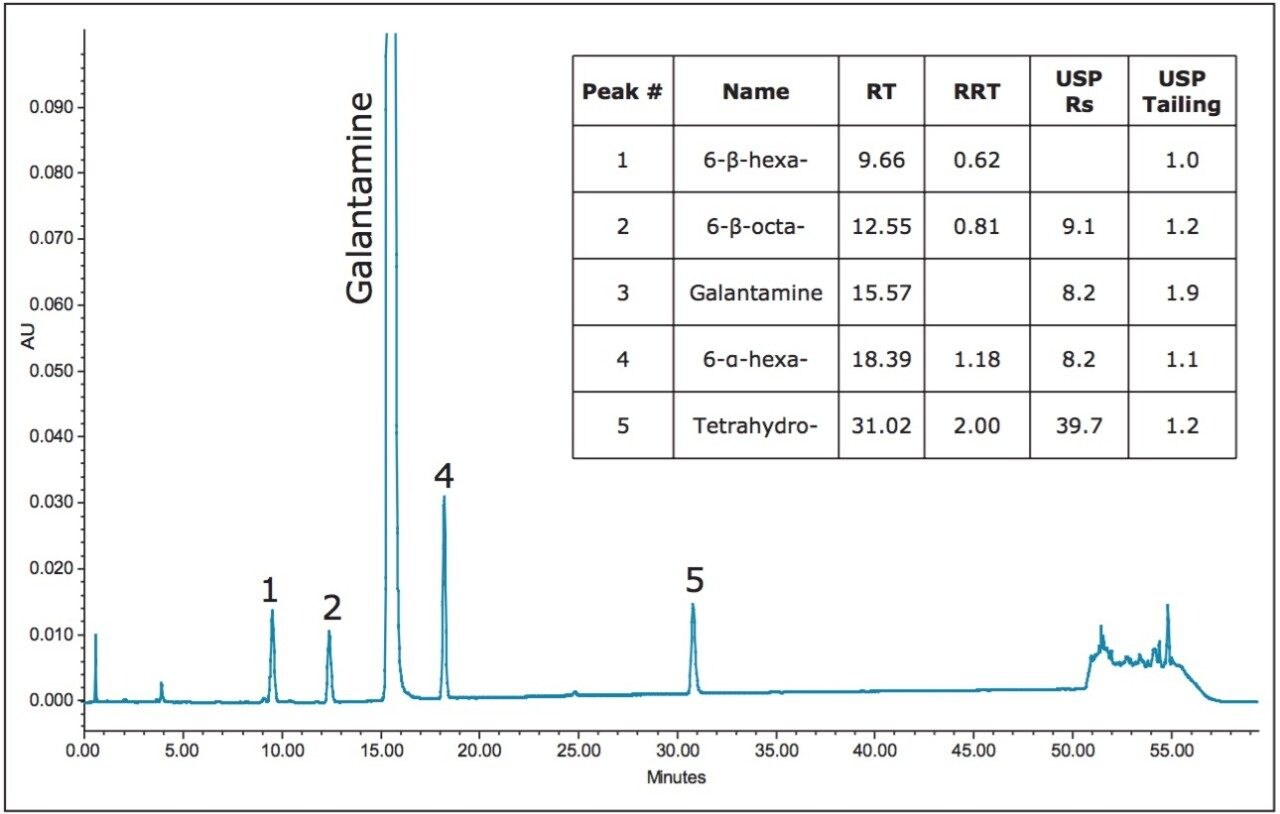
Differences between instrumentation dwell volumes can be easily accounted for with calculated adjustments to the gradient table with the ACQUITY UPLC Columns Calculator. However, the challenging method translations of original methodology to a target methodology reside with differences in column stationary phase selectivity.
Ideally, when scaling from an HPLC column to a UPLC column, the stationary phase should remain constant (i.e., “equal” stationary phase) to maintain the selectivity of the separation. Unfortunately, many original HPLC columns are not available in the same chemistry in sub-2-μm particle sizes. Therefore, an equivalent stationary phase that is available in sub-2-μm particle size must be determined. This task is facilitated using the Waters Reversed Phase Column Selectivity Chart (www.waters.com/selectivitychart) (Figure 4).
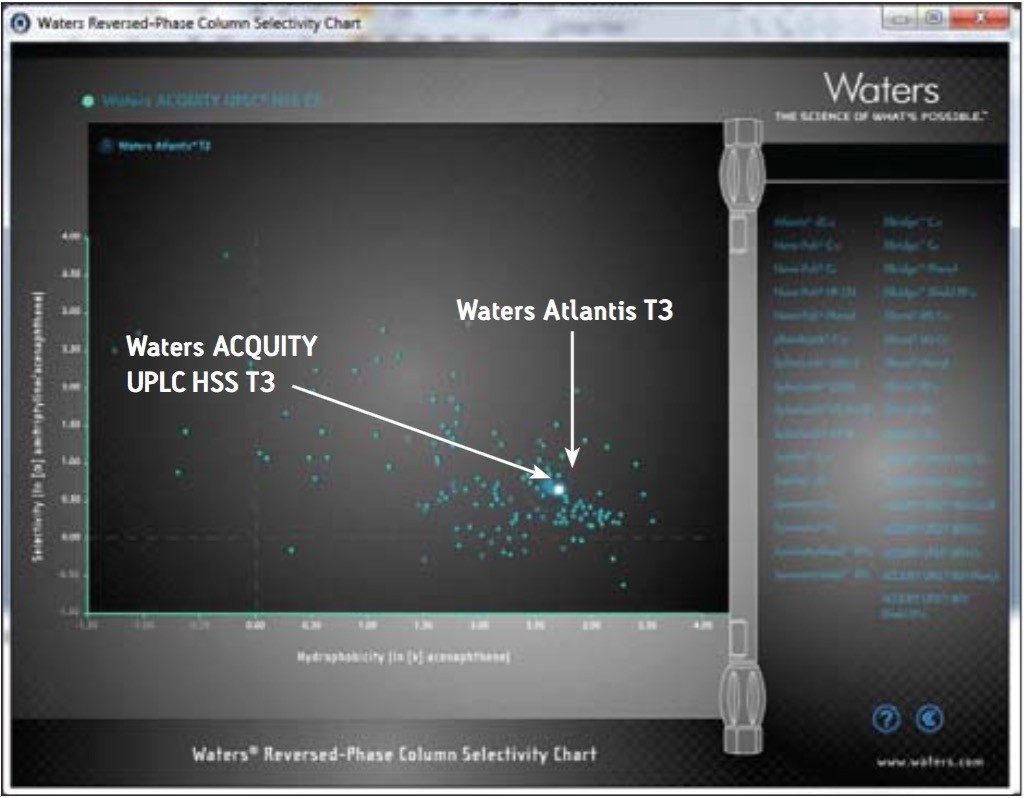
New pressures in the pharmaceutical industry have created a need for QC laboratories to become more productive. It is important to reduce costs in QC, but not at the expense of R&D or any other part of the organization. Additionally, the reduction in cost cannot come at the expense of chromatographic accuracy, robustness, or reliability.
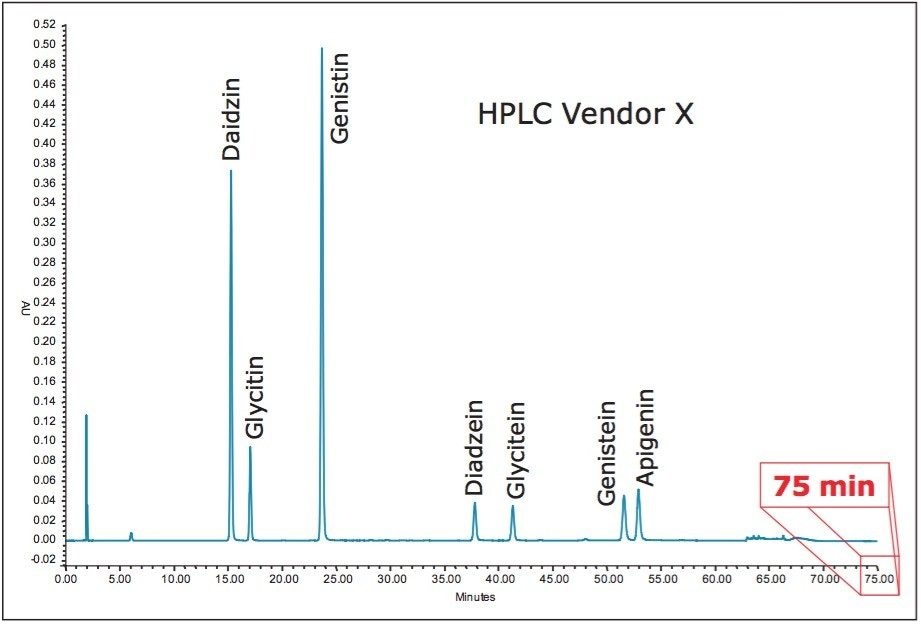
Dietary supplement manufacturers routinely use HPLC to analyze soy extracts for isoflavone content. The current USP compendial method uses a long, shallow gradient that takes 75 minutes per injection. This long run time limits the ability of manufactures to release products quickly. In addition, a sample set run consisting of a blank, five calibration standards, and two retention time check solutions requires more than 10 hours before running the analysis of the first sample. The benefits of analyzing isoflavones using a faster solution that maintains data quality are improved productivity, increased revenues, enhanced efficiency, faster sample turnover, and reduced labor and training costs.
Using the ACQUITY UPLC Columns Calculator, the HPLC USP method for soy isoflavones shown in Figure 5 was transferred to a UPLC method employing an ACQUITY UPLC HSS T3 Column, 2.1 x 100 mm, 1.8 μm. The geometrically scaled method had a runtime of 24.3 minutes. Because the scaled flow rate of 0.319 mL/min is below the optimum linear velocity for the sub-2-μm particle column, the columns calculator was used to recalculate the gradient at 0.60 mL/min, a flow rate that is closer to optimum (Figure 6).
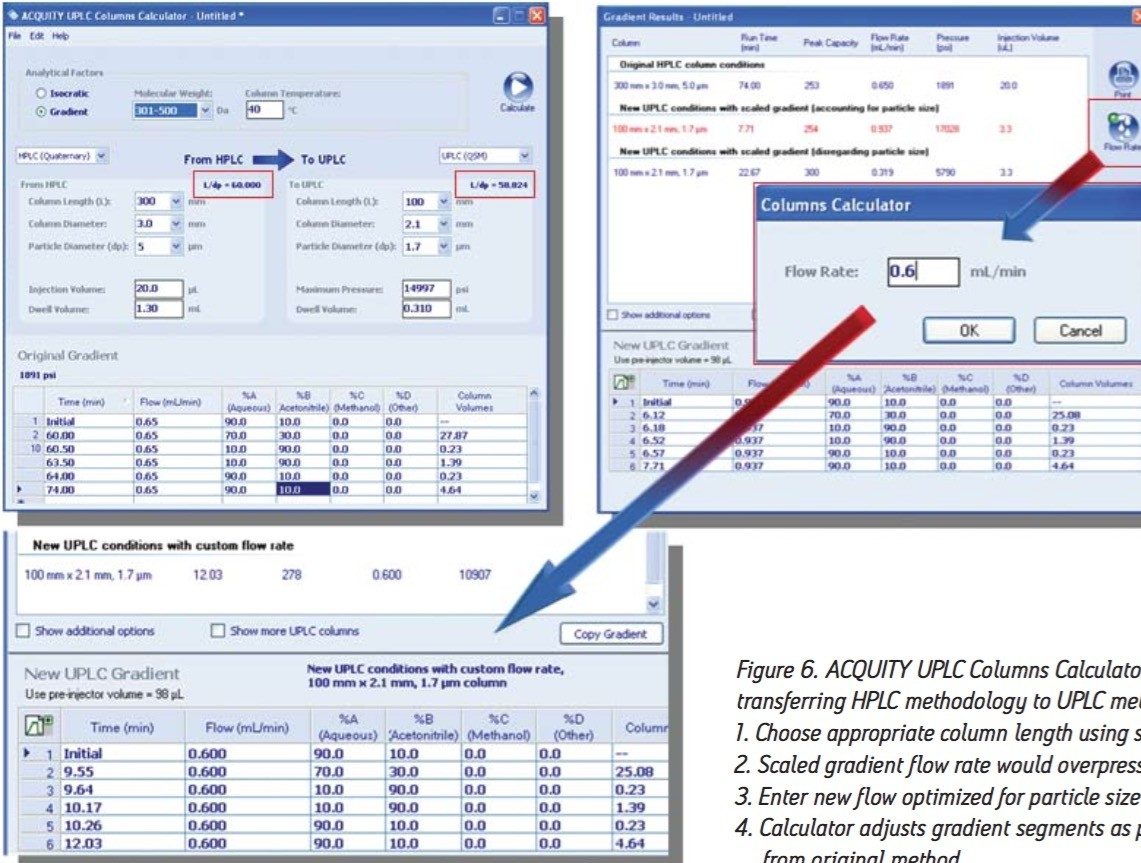
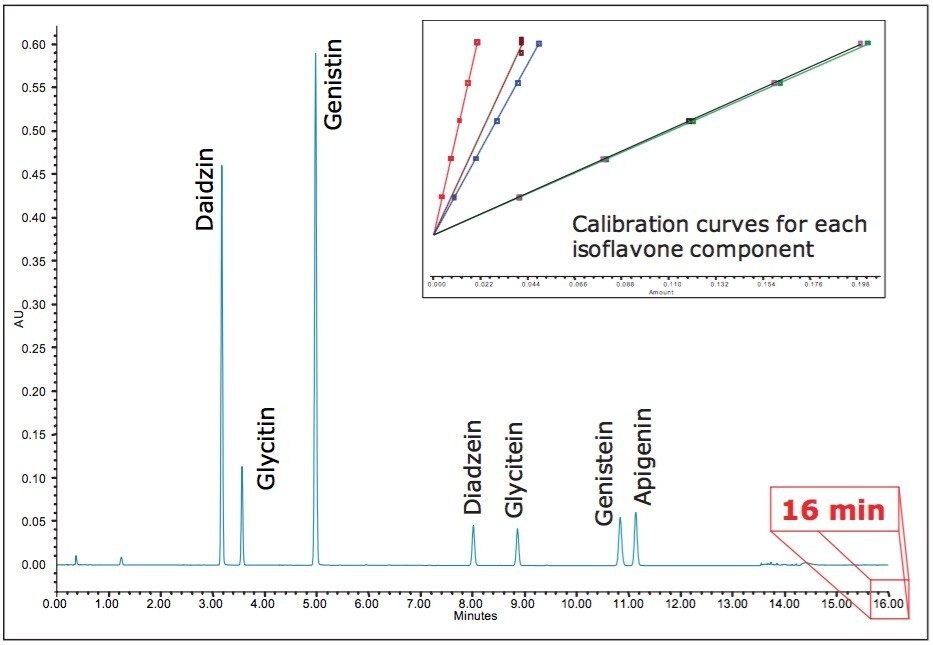
Using the columns calculator, the resolving capabilities of the HPLC column was maintained by choosing a UPLC column dimension with the same column length to particle size (L/dp) ratio. The injection volumes and flow rate were scaled appropriately, and the gradient was corrected to keep the number of column volumes consistent for each time segment. The resulting chromatogram is displayed in Figure 7. We can see that the analysis time has been reduced to 16 minutes. Using this approach, the method was successfully transferred to UPLC with both improved throughput and assay performance. The quality of the analytical results using this new and significantly faster UPLC method were not compromised, and thus met the specified USP criteria.
Analytical development organizations have decreased their method development time by implementing UPLC, however their customers in many situations across the globe have not yet implemented UPLC technology. Maximizing the utilization of the current instrumentation is key to their productivity until appropriate justifications and budgeting is available to adopt the new technology.
In such cases, the method innovator must adapt the UPLC methodology for HPLC use. Implementing the method translation strategy combining the ACQUITY UPLC Columns Calculator and the appropriate column Method Transfer Kit can facilitate the translation of UPLC methodology to HPLC methodology.
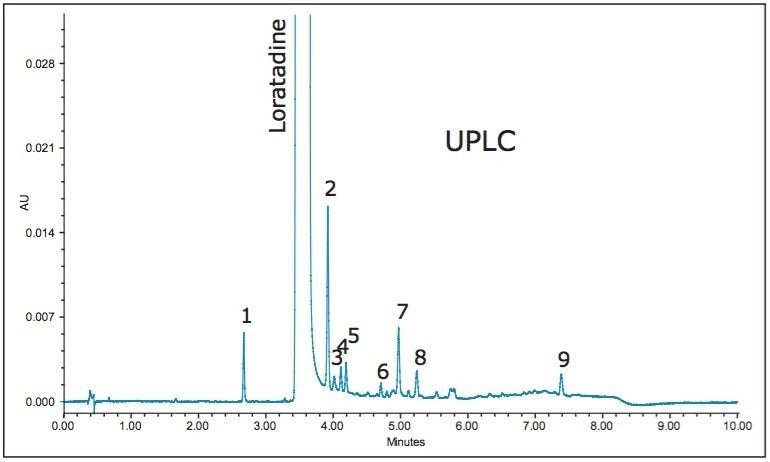
A UPLC method developed for loratadine and its related substances separated nine impurities and the API to meet a set of system suitability criteria as specified in the USP within 10 minutes (Figure 8). The methodology was translated utilizing the ACQUITY UPLC Columns Calculator and ACQUITY UPLC BEH Method Transfer Kit. The key aspects in allowing the transferability to HPLC from UPLC are similar to those stated in the previous example, such that the target column dimensions must have equivalent L/dp values and the column stationary phase selection is equivalent to the originating methodology. The resulting HPLC chromatogram (Figure 9) was compared to the UPLC chromatogram in terms of relative retention time ratios of the related substances.

In addition to the UPLC to HPLC transfer of loratadine, the UPLC and HPLC methods were compared on three different instruments (Alliance HPLC 2695, ACQUITY UPLC, and ACQUITY UPLC H-Class) in order to evaluate the accuracy of the entire method transfer process (Table 1).
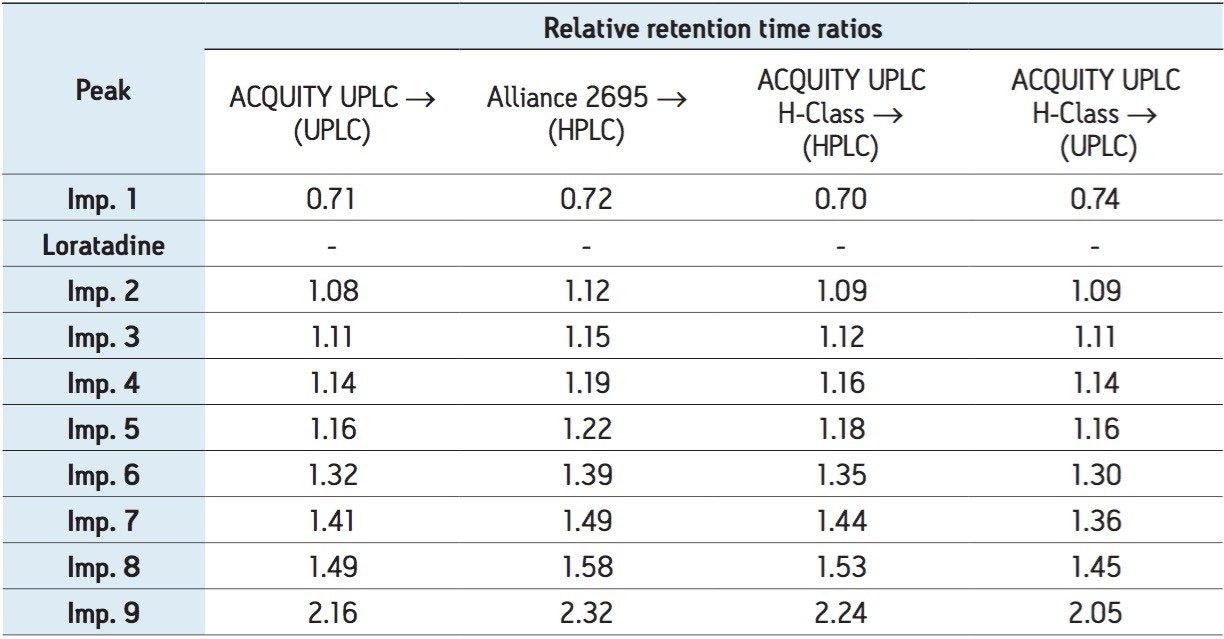
The compendia methods translation experiments were facilitated using a method translation strategy comprised of software tools, column Method Transfer Kits, and thorough knowledge of the instrumentation used. In each example, the chromatographic attributes and integrity of the original methodology were maintained.
Choosing a compatible column stationary phase exhibiting “equivalent” or “equal” selectivity and resolution characteristics was key when transferring from legacy HPLC methodology to UPLC methodology. The process of translating from HPLC to UPLC can be difficult due to the availability of a sub-2-μm particle size equivalent columns with the same originating HPLC stationary phase, especially if the originating HPLC stationary phase was introduced many years prior. The reversed-phase selectivity chart can facilitate proper stationary phase selection in many of these instances, however, some selectivity differences may be observed.
The process of translating methodology from UPLC to HPLC is made easier with columns that are available in both UPLC and HPLC particle sizes, as in the case of ACQUITY UPLC BEH and XBridge, ACQUITY UPLC CSH, and XSelect CSH, and HSS UPLC and HPLC columns.
In an effort to streamline method translation, QC organizations should open communications with R&D organizations presently implementing UPLC for methods development. Discussions should focus on the intricacies of maintaining column selectivity for UPLC and HPLC, as well as the importance of L/dp values for maintaining resolving capabilities of a column. These discussions would help devise a cohesive implementation strategy that can supplement the method translation strategic approach earlier within development.
720003721, September 2010