
In this application note, we discuss preliminary tests on a novel, high conductance inlet/conjoined ion guide system and restricted APCI nebulizer design that demonstrate a large improvement in sensitivity when compared to a conventional inlet MS.
LOQ improvements of up to 100 times were observed with the UPLC-MS method, which led to sub-picogram on-column quantitation.
The first commercially available API sources employed relatively small ion inlet orifices (Ø0.1 to 0.2 mm) to admit ions and gas molecules from an ionization region that was held at atmospheric pressure. Since the number of ions entering the MS, and hence sensitivity, both increase with increasing orifice diameter, API sources have evolved to incorporate inlet orifices of typically Ø0.4 mm and a number of forepumping regions to gradually reduce the pressure from atmosphere to the MS analyzer pressure. In order to reduce the level of detector noise from droplets and energetic neutrals that invariably increase with increasing gas throughput, some source designs employed orthogonal ion sampling from the first vacuum stage. In this study, we present APCI data obtained on a high gas conductance inlet system that features an Ø0.8 mm orifice and a novel conjoined ion guide that maximizes ion transmission and reduces spurious detector noise from microdroplets. This inlet has been combined with an improved APCI heated nebulizer design that incorporates a laser-drilled nebulizer gas flow restrictor to optimize aerosol generation prior to desolvation.
All test samples were analyzed via a standard UPLC method at a mobile phase flow rate of 0.8 mL/min. A Waters ACQUITY UPLC System and an ACQUITY UPLC BEH C18 (1.7 μm, 2.1 x 50 mm) Column were used with a full loop injection volume of 5 μL and a column temperature of 50 °C.
Mobile phase A was 2 mM aqueous ammonium formate and mobile phase B was 100% methanol. Mobile phase A was held at 90% between 0.00 and 0.25 min, ramped linearly to 10% at 1.50 min, held at 10% to 2.00 min, and then stepped back to 90% between 2.01 and 2.50 min. The test analytes were caffeine, sulphadimethoxine, and 17-alpha hydroxyprogesterone, which were injected as a mixture at relative concentrations of 1:1:5, respectively.
A schematic of the prototype Waters Xevo TQ-S System is shown in Figure 1. Here, the entire gas flow from the Ø0.8 mm inlet orifice is directed into the entrance of the conjoined ion guide under the influence of a 100 m3/hr vacuum rough pump. The conjoined ion guide utilizes a combination of rf containment and a transverse dc component between the major and minor tunnels of the lens structure, as shown in Figure 2. This serves the purpose of separating the highly mobile ions from unwanted droplets and reduces the effects of gas streaming into the first turbo pumped region of the MS.
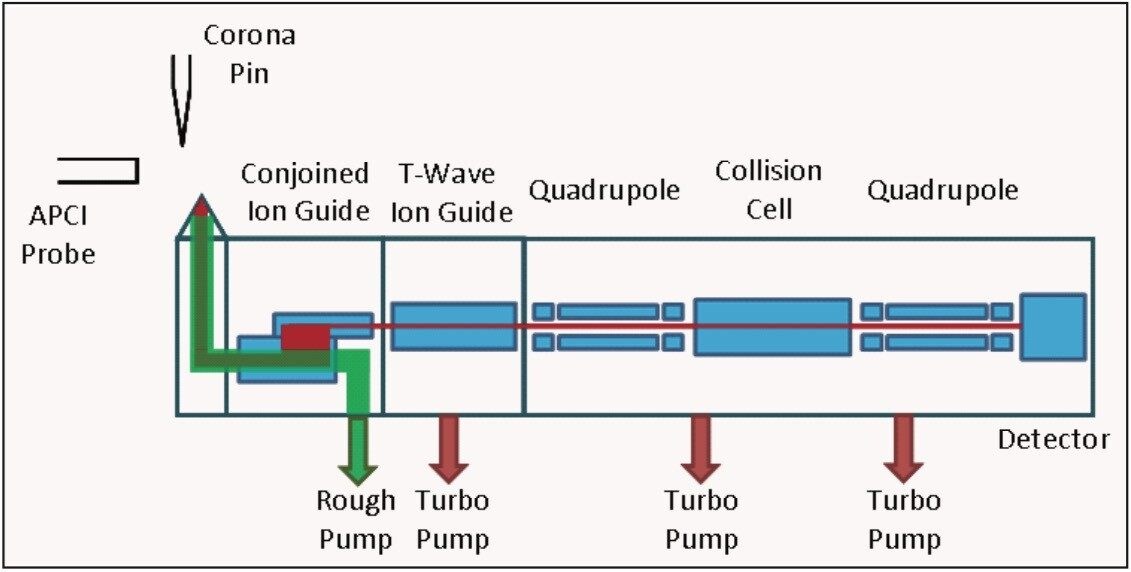
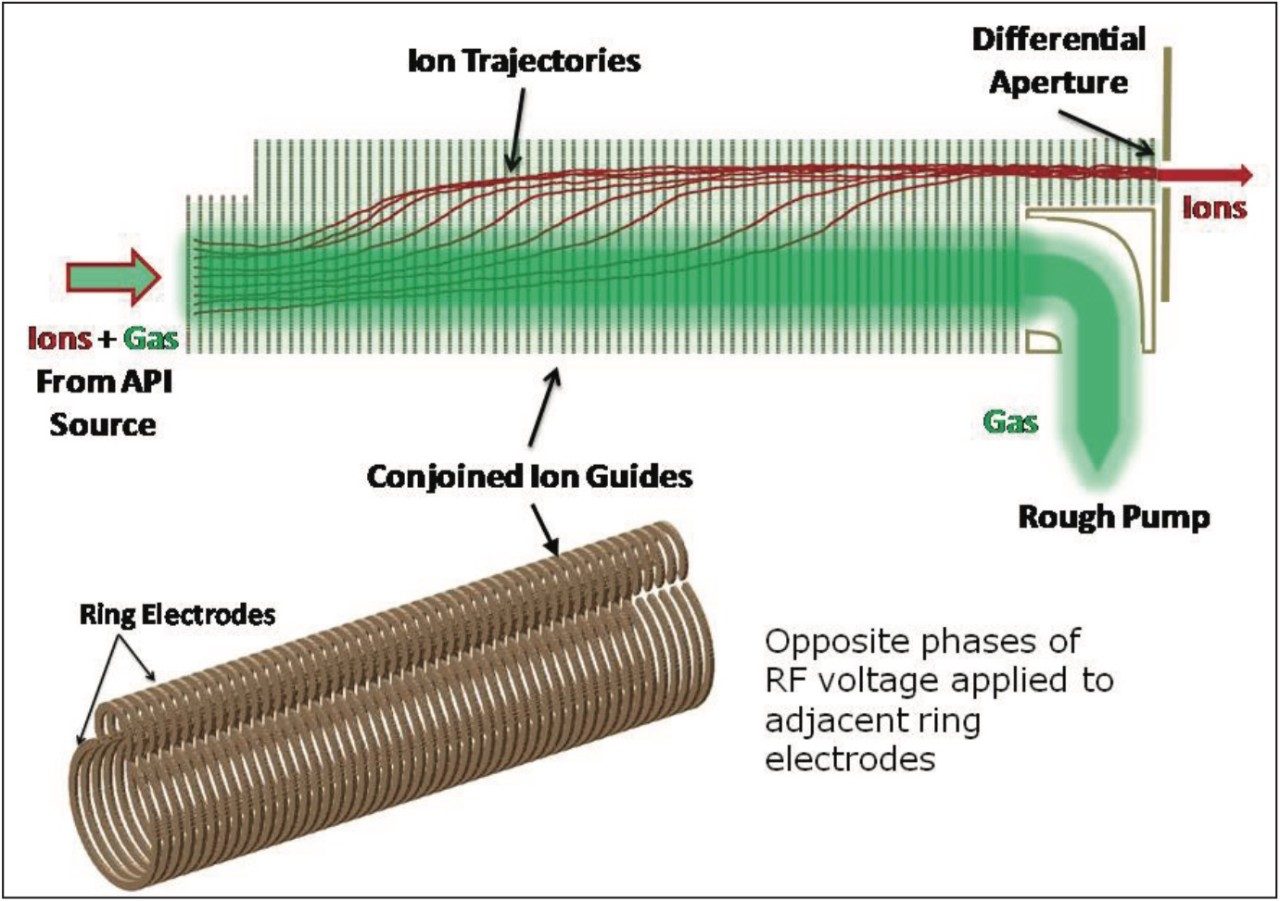
This MS system also incorporates an improved APCI heated nebulizer design that utilizes a precision, Ø0.4 mm laser-drilled nebulizer gas flow restriction that surrounds the liquid inlet capillary and includes the ability to tune the nebulizer gas pressure (40 to 100 psi). Furthermore, the corona pin position is relocated 5.5 mm upstream of the ion inlet orifice when compared to the conventional design.
The conventional MS used in this comparative study was a standard Waters Xevo TQ MS System. This MS incorporates a Ø0.35 mm inlet orifice and splits the incoming gas flow prior to admitting ions into the first rf containment device. Furthermore, this MS did not include the improved APCI heated nebulizer probe. Here, the nebulizer gas was delivered to the liquid capillary tip via an unrestricted 0.60 mm I.D. stainless steel tube at a fixed nebulizer gas pressure of 100 psi.
Data were acquired in MRM mode: caffeine, m/z 194 to m/z 138, sulphadimethoxine, m/z 311 to m/z 156, and hydroxyprogesterone, m/z 331 to m/z 109. All data were obtained with an APCI probe heater temperature of 650 °C, and a corona current of 10 μA.
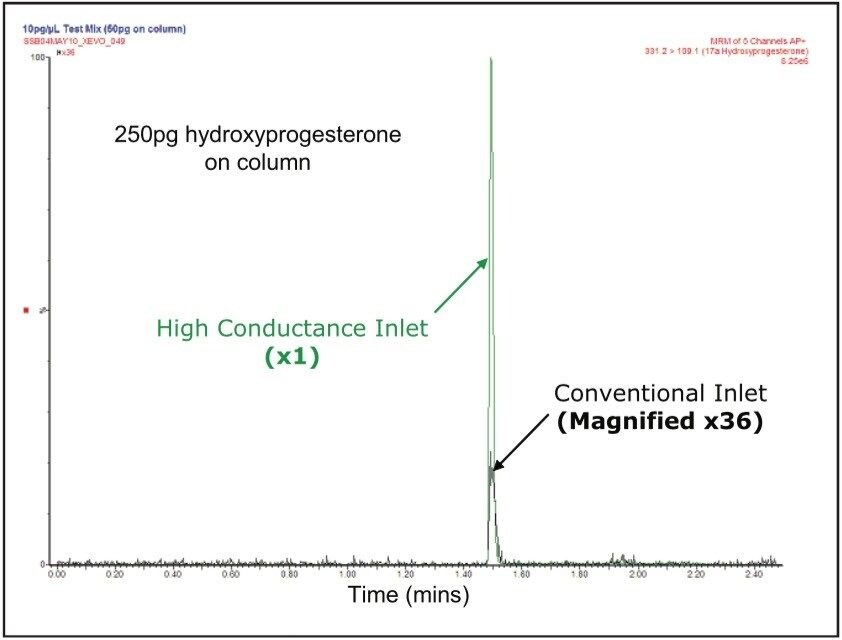
A back-to-back sensitivity comparison was conducted between the conventional and high conductance sources using the same sample set within a 12 hour analysis period. Each sample concentration was replicated six times, and the lowest concentrations used were 0.01 pg/μL. Figure 3 shows the magnitude of the sensitivity enhancement typically obtained with the high conductance inlet (~180 x). Similar enhancements were obtained with all three analytes used in this study. Table 1 compares the MRM chromatographic peak areas obtained using the two different inlet systems and the resulting total enhancement. The total signal enhancement observed with the high conductance system can be attributed to an APCI probe contribution from the nebulizer restriction design and a contribution from the ion inlet design. To determine the contribution from the APCI probe, a series of infusion experiments was conducted with both probe types on the high conductance system, where each analyte was infused under mobile phase conditions that were equivalent to their elution composition. Figure 4 shows the dependence of APCI probe enhancement (restricted nebulizer signal/unrestricted nebulizer signal) on mobile phase composition for the three analytes used in this study and an additional two compounds.
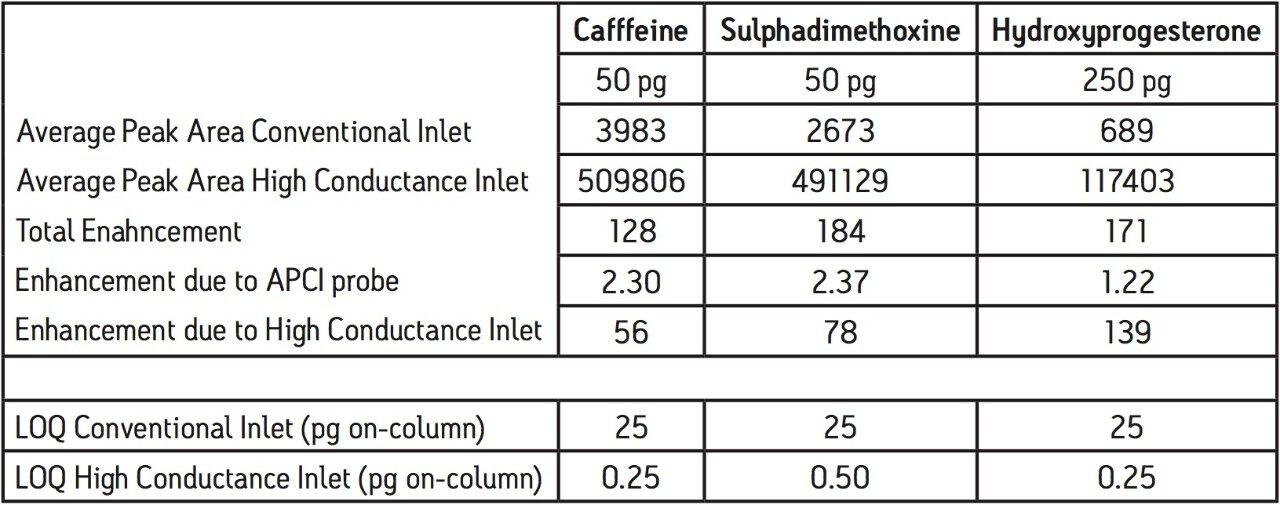
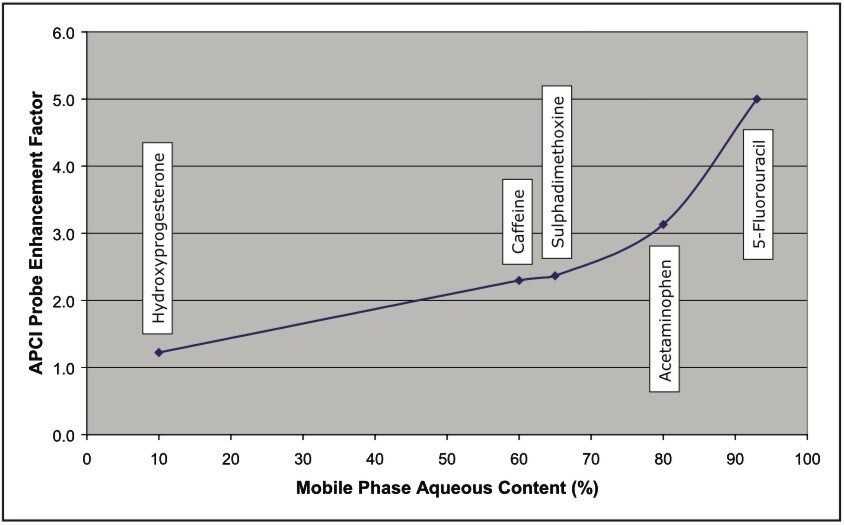
Here, it was observed that the probe enhancement factor rose with increasing aqueous content at a constant probe temperature (650 °C); however, it should be noted that enhancements at low aqueous contents can be increased by reducing the probe temperature across the UPLC gradient (not used in this study). Referring to Table 1, when we apply the probe enhancement contribution to the figures obtained for the total enhancement, we can deduce that that contribution to signal enhancement arising from the use of the high conductance inlet is in the range 56 to 139 times that of the conventional inlet. These figures compare favorably to those obtained in a similar study on the two inlet types, where signal enhancements in ESI+ and ESI– modes were found to be typically in the range 25 to 125 times.
Table 1 summarizes the limit of quantitation (LOQ) values obtained with this UPLC-MS method using both inlet types. The conventional inlet was shown to give LOQs of 25 pg on-column for all three analytes. Here, LOQ is defined as < 20% chromatographic peak area deviation from the least squares fit standard curve for each analyte with a further requirement on blank peak areas to be < 25% of the LOQ value.
In comparison, the combination of the high conductance inlet and improved probe design reduced LOQs by 50 to 100 times, resulting in sub-picogram quantities of 0.25, 0.50, and 0.25 pg for caffeine, sulphadimethoxine, and hydroxyprogesterone, respectively.
These preliminary tests are very encouraging from a sensitivity viewpoint, and extend the routine use of APCI to sub-picogram UPLC-MS analyses. It is our intention to extend the current study to determine the LOQs and linear dynamic ranges under real-life conditions, such as the analysis of target compounds in human plasma, food extracts, and more.
720003607, June 2010