In this application note, we describe a simple LC-MS method for the analysis of a Vitamin B12 standard. The determination of Vitamin B12 is important, but can be challenging largely because of its chemical instability and the complexity of the matrices in which it is usually found. Ideally, methods for Vitamin B12 analysis should be simple, selective and sensitive to overcome the above issues. The most commonly reported method for Vitamin B12 analysis is reverse-phase HPLC separation with various detection schemes such as UV, fluorescence, chemiluminescence, etc.
Acrylic polymers have broad applications in the manufacture of paints and paper coatings, emulsion adhesives, plastics, lighting, glass replacements, personal care products and even dental fillings. However, acrylic acid and some acrylate oligomers and monomers adversely affect human health as eye and skin irritants. Exposure to monomers occurs primarily in the workplace via inhalation and dermal contact during its manufacture or use.1 Consumers may be exposed to residual monomers in polishes, paints, coatings, rug backings, adhesives, plastics, textiles and paper finishes.1
Because of these issues, manufacturers closely monitor and control the residual monomer content in acrylic polymers. High Performance Liquid Chromatography (HPLC) is the techique of choice to measure free acrylic monomers in polymers before cross-linking. Typically, sample preparation to extract a variety of residual monomers from the polymer matrix is complex, tedious and time-consuming2,3,4. If the extract contains both polar and non-polar monomers as analytes of interest, then it may be necessary to perform two separate HPLC experiments.5
This note illustrates an easy-to-use protocol to quantify polar and non-polar free monomers in a polymer matrix using reverse-phase HPLC. Polymer samples were directly dissolved in tetrahydrofuran, eliminating monomer extraction from the polymer matrix. In the same run with only a single injection, polar and non-polar monomers were separated, thereby reducing the total sample analysis time. This protocol enables manufacturers to increase the efficiency of laboratory processes related to the analysis of residual monomers which facilitates release of final product.
Acetonitrile (MeCN), tetrahydrofuran (THF, stabilized with 159 ppm BHT, butylated hydroxytoluene), toluene (J. T. Baker). o-Phosphoric acid (85%, A.C.S. reagent grade), acrylic acid, and alkylacrylate monomers
(Sigma-Aldrich, St. Louis, MO). Water (generated by Millipore Milli-Q Gradient A10 system), acrylic polymers in toluene (Scientific Polymer Products, Inc., Ontario, NY).
|
System: |
Waters Alliance 2695 with 2996 Photo Diode Array |
|
Software: |
Empower 2 |
|
Column: |
Sunfire C18, 4.6 x 15 mm |
|
Sample temp.: |
40 °C |
|
Detection: |
210 nm |
|
Time constant: |
0.2 s |
|
Sampling rate: |
1 Hz |
|
Filter Response: |
0.1 s |
|
Injection: |
5 μL |
Stock calibration solution was prepared by weighing approximately 20 mg each monomer into a glass container into which approximately 20 mL of THF was added. The volume of the solution was calculated from the total mass of solution by using the density of THF at room temperature (0.889 g/mL). Calibration stock solutions were prepared by diluting the stock solution with THF approximately 40%, 10%, 3%, 1% and 0.5%. The exact dilution factor was calculated from the mass of the stock solution and the total solution mass. Samples were prepared by dissolving the acrylic polymers into 20 ml of THF, making approximately 5 mg/mL solutions.
The polymer samples were dissolved in THF, injected and the residual monomers separated with gradient elution.
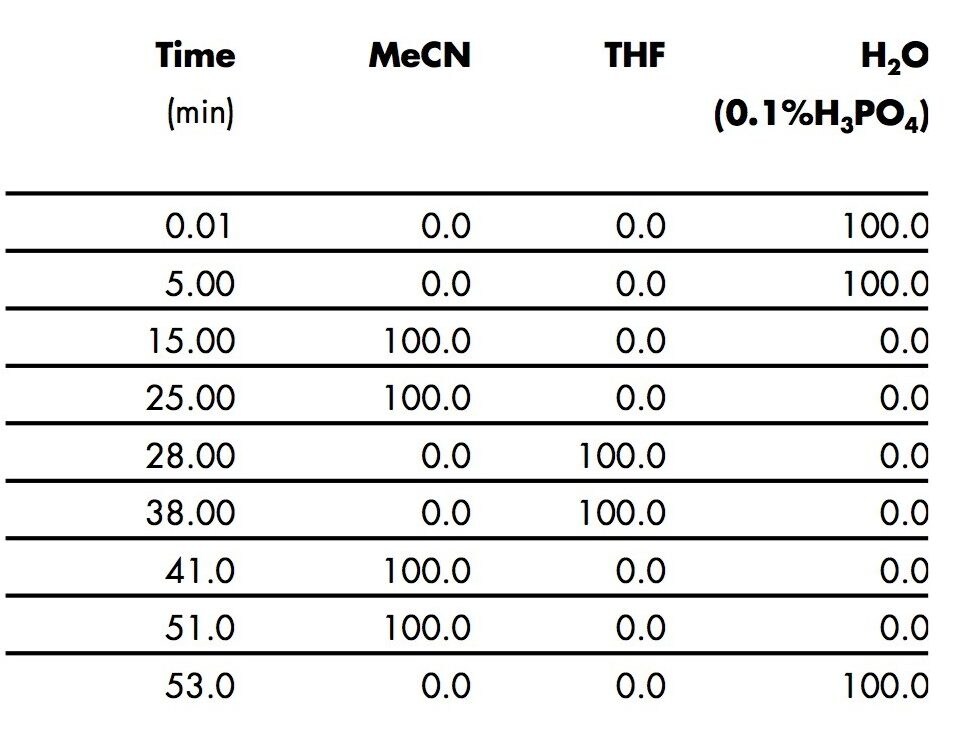
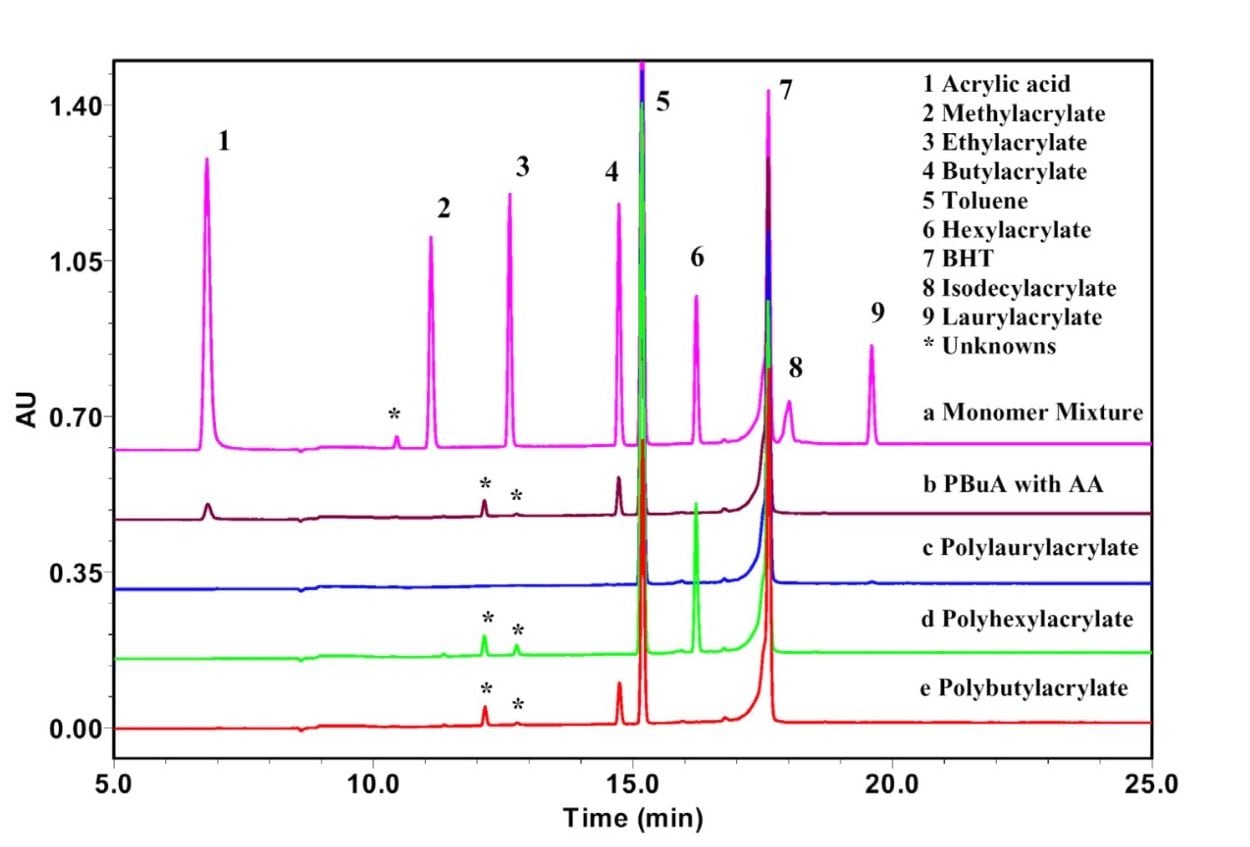
The gradient program (Table 1) consists of 2 gradient elution profiles. The first gradient (water to acetonitrile) separates the acrylic monomers but retains the polyacrylates on the column whereas the second gradient (acetonitrile to THF) elutes the polyacrylates from the column.7 Figure 1 shows chromatograms of a mixture of common acrylic monomers (a) and several commercial polyacrylate standards (b-e) in toluene. Peaks were identified by obtaining individual chromatograms of the monomers, the anti-oxidant (BHT) and toluene under the same conditions.6 This second gradient eliminates the need for a separate procedure to extract polar and non-polar monomers from polymer samples.
An example of the usefulness of this step is shown in Figure 1 Chromatogram b, where both acrylic acid (polar) and butylacrylate (non-polar) peaks are separated, identified and available for quantification. The remaining gradients in the program (Table 1) return the column to initial conditions and equilibrate.
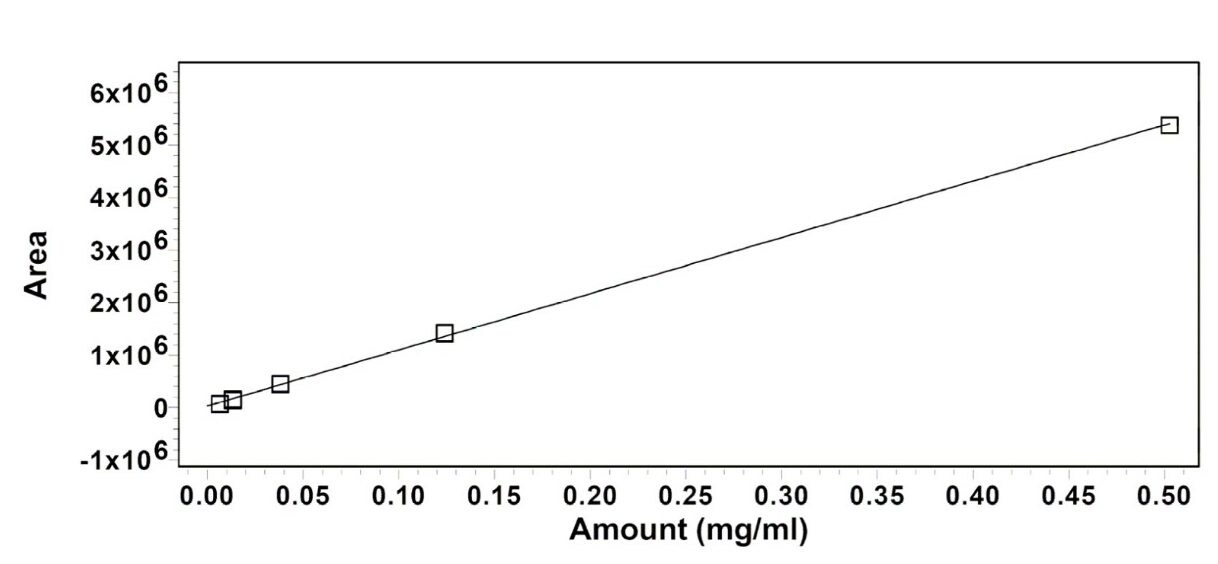
The calibration curves of peak area vs. concentration were obtained for acrylic acid, methylacrylate, ethylacrylate, butylacrylate, hexylacrylate, isodecylacrylate and laurylacrylate. Figure 2 shows a typical calibration curve. The range of the calibration curves, the slope and the intercepts, and the square of the correlation coefficient are in Table 2. The Limit of Detection (LOD) and the Limit of Quantification (LOQ) were estimated from the noise level in the chromatogram of the THF blank.
These values support the utility of this approach for obtaining data relevant to the acrylate manufacturing community.1 The observed monomer content in commercial polyacrylate polymer standards as concentration and weight % are in Table 3. To illustrate using this methodology for identifying and quantifying residual acrylic acid, a polybutylacrylate sample was spiked with a known concentration of acrylic acid, then analyzed. The measured acrylic acid concentration (Table 3) is within 2.5% of the spiked concentration.
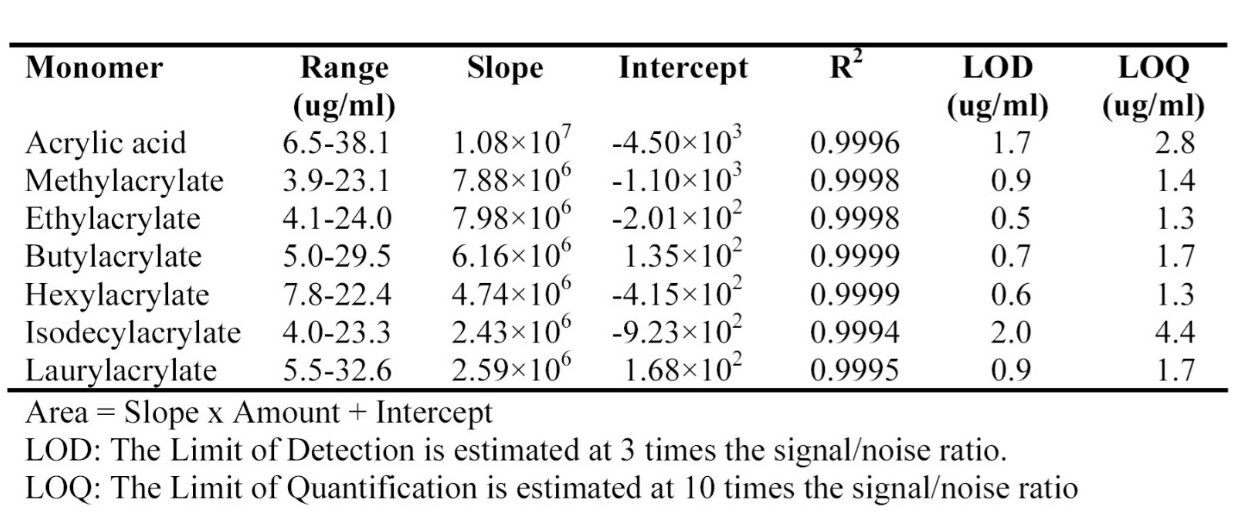
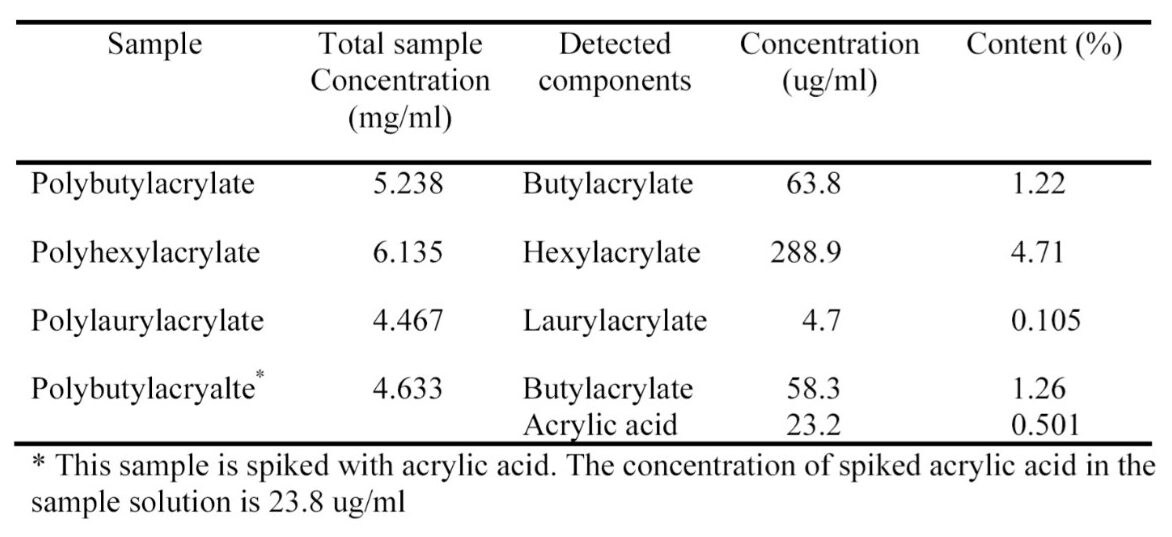
Polar and non-polar compounds found as residual monomers in polyacrylates can be easily separated and quantified. There was no need for a separate step to extract the monomers from polymer and perform two separate HPLC experiments. Acrylic acid can be easily quantified; LOD and LOQ were estimated at 1.7 μg/mL and 2.8 μg/mL, respectively.
This methodology simplifies sample preparation and reduces total analysis time. The approach not only provides information critical to polyacrylate manufacturers but does so at a higher level of efficiency than existing methods. This enables chemical analysis lab managers to increase work capacity as lab personnel and instruments may be utilized for additional analyses.
720001909, October 2006