High Temperature Gas Chromatography Analysis of Polycyclic Aromatic Hydrocarbons (PAHs) Using Atmospheric Pressure Chemical Ionization GC/MS/MS
Douglas Stevensa, Frank Dormanb, Bryan Jamesc, Cara Megillc, Christopher Reddyd, Robert Nelsond
aWaters Corporation, Milford, MA United States
bDepartment of Chemistry, Dartmouth College, Hanover, NH United States
cDepartment of Chemical Engineering, Northeastern University, Boston, MA United States
dWoods Hole Oceanographic Institution, Woods Hole, MA United States
Published on February 19, 2026
Abstract
High temperature (HT) GC/MS analyses, using temperatures of up to 400 °C, span a range of applications including food analysis, environmental forensics, petroleum research, and polymer characterization. The need for high temperature is because analytes with the lowest volatility and highest boiling points tend to remain absorbed within complex matrices for longer periods and under more severe conditions compared with more volatile species. This makes these high boiling point analytes important and useful for the thorough chemical characterization of a broad range of sample types.
In this work, the HT GC analysis of high molecular weight (HMW) polycyclic aromatic hydrocarbons (PAHs) of up to 424 Da was used to evaluate the performance of gas chromatography atmospheric pressure chemical ionization (GC-APCI) combined with tandem quadrupole mass spectrometry (MS/MS). Because of the limited availability of analytical standards in this range, a class-specific PAH acquisition mode was developed and evaluated using standard reference materials (SRMs). The instrument used for this work had a single supply of nitrogen for carrier gas, reagent gas and collision induced dissociation (CID) gas.
The optimized method had a 31-minute run time and a late eluting analyte of 424 Da at 21.3 minutes and 395 °C with symmetric peak shape and a width of <5s. A semi-quantitative comparison was made by comparing the HMW PAH response to reference values reported for the SRMs. Individual peaks at 326 Da and 374 Da were compared with dibenzo[a,e]pyrene at 302 Da and found to be 57% and 23% as intense, respectively. This demonstrates that the HMW PAHs targeted in this work are present at abundances that make their further study relevant for multiple applications and that the GC-APCI MS/MS system is readily adaptable to this HT GC application.
Benefits
- Improved characterization of PAHs through the combination of high temperature GC with atmospheric pressure chemical ionization (HT GC-APCI) has application in research topics such as toxicity studies and environmental analyses such as the study of fate, transport and occurrence
- Use of a single gas for chromatographic separation, ionization and MS/MS fragmentation provides a simpler, more sustainable system
- Use of nitrogen reduces concerns over cost and supply chain reliability that periodically interfere with methods that rely on helium carrier gas
- Atmospheric pressure ionization with MS/MS helps overcome some limitations imposed by changes to chromatography required to facilitate high temperature analyses
Introduction
Common GC/MS PAH methods target only the 16 parent PAHs in the U.S. EPA Priority Pollutant List with a maximum molecular weight of 278 Da.1 Even methods with expanded analyte lists of 50 analytes or more frequently analyze no higher than 302 Da.2 However, a number of publications using a variety of instrumentation have reported the presence of PAHs of up to 424 Da in both SRMs and sample extracts.3-6 These HMW PAHs are of interest because toxicity modeling based on analysis of the EPA 16 alone has, in some instances, shown poor correlation to predicted health effects associated with the PAH content of samples.7 It is advisable then to investigate the possibility that inclusion of HMW PAHs in research on topics such as toxicity or environmental forensics may reveal more reliable correlations between specific HMW PAHs and health impacts or with determination of characteristics such as a sample’s petrogenic or pyrogenic origin.8
However, monitoring HMW PAHs is not without its challenges, especially if HT GC is employed. The columns used are typically shorter with a thinner stationary phase than those used in more conventional GC/MS methods. This is primarily to reduce excessive column bleed at high temperature, but can result in reduced sample loading capacity and reduced chromatographic resolution. Using tandem quadrupole mass spectrometry (GC-MS/MS), as opposed to GC/MS, often adds sufficient specificity to a HT GC-MS/MS method to regain separation of high boiling analytes not fully chromatographically resolved on shorter, thin film columns. The sensitivity of MS/MS also helps address the reduced loading capacity of these columns. This makes MS/MS a key enabling technology for successful HT GC-MS/MS method implementation.
While carrier gas flow rate is another important condition for optimization of chromatographic separations, it is often overlooked when using electron ionization (EI) instruments because the vacuum in the ionization source imposes flow rate limitations on the GC. Atmospheric pressure ionization interfaces, such as the atmospheric pressure chemical ionization GC (APGCTM) source, not only tolerate high carrier gas flows, but also adapt well to the use of either helium or nitrogen carrier gas. This is another way in which the GC-APCI technology facilitates HT GC/MS/MS because it is common in these methods to ramp the carrier gas flow rate to efficiently separate analytes that elute near the maximum temperature of the oven ramp.
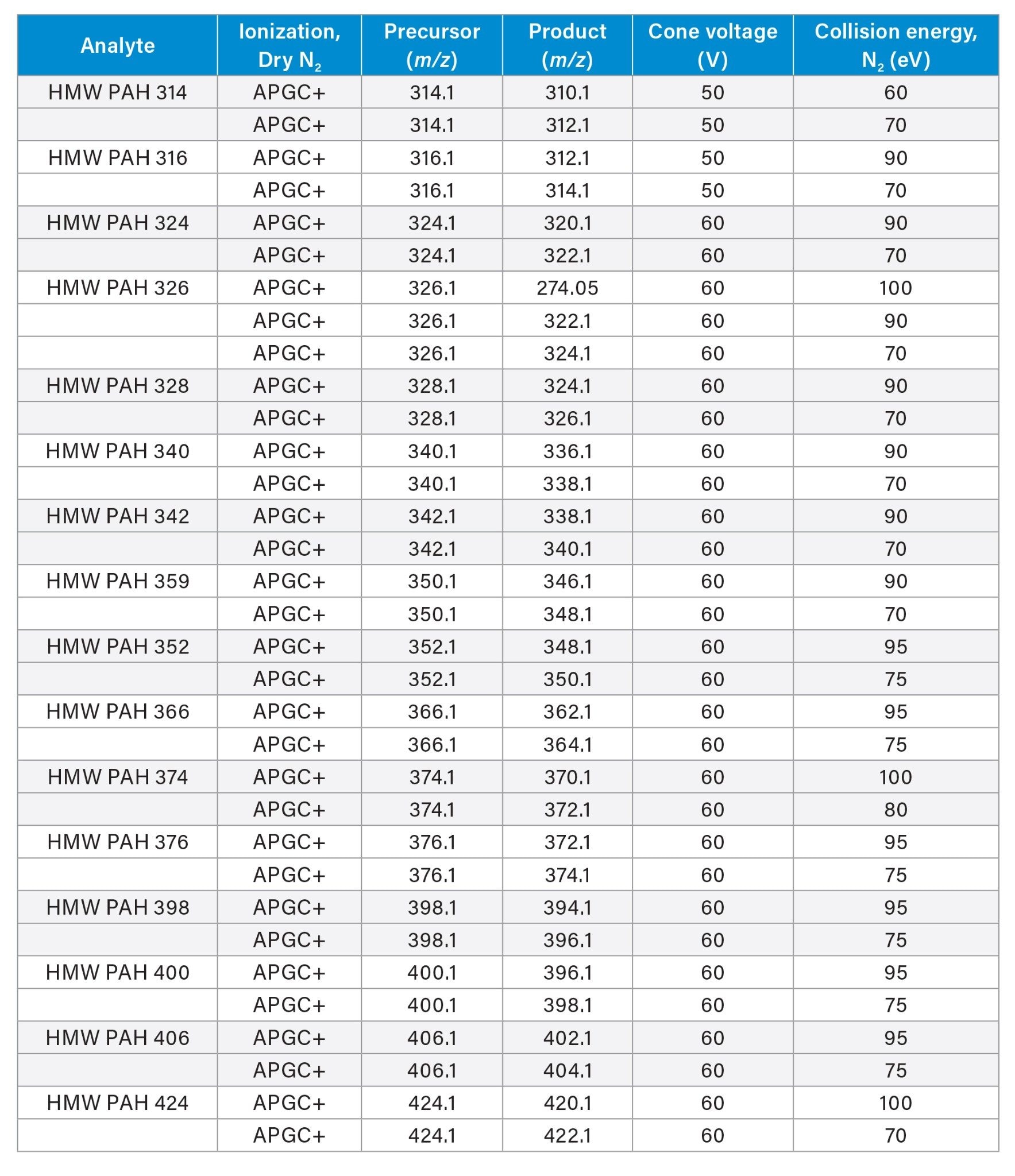
Coal tar SRMs from the National Institute of Standards and Technology (NIST) contain significant amounts of well characterized PAHs of ≤302 Da. In addition, they are reported to contain HMW PAHs ≥314 Da of high abundance but lack quantitative reference values and individual analyte identification. So, while the presence of the HMW PAHs in the SRMs make them well suited to the evaluation of HT chromatographic separations, target analysis using MS/MS may be challenged by the absence of individual standards for each analyte in this high mass range. However, optimization of parameters for the lower mass, well-characterized individual PAHs contain trends that allowed the development of a HMW PAH class-specific acquisition scheme using multiple MRM transitions for monitoring of PAHs in the range of 314 to 424 Da.
In this work, the analysis of HMW PAHs was demonstrated to evaluate the HT GC/MS/MS performance of APGC on Xevo™ TQ Absolute tandem quadrupole Mass Spectrometer using NIST SRMs 1597a and 1991.
Experimental
Sample Preparation
NIST SRMs used in this work were 1991 Mixed Coal Tar/Petroleum Extract, and 1597a Complex Mixture of Polycyclic Aromatic Hydrocarbons from Coal Tar. Each of these was diluted 10:1 in methylene chloride (DCM; Acros Organics, 326760010; purity >99.9%) containing 200 ppb SV internal standard mix (Restek Corporation) 1,4-dichlorobenzene-d4, naphthalene-d8, acenaphthene-d10, phenanthrene-d10, chrysene-d12, perylene-d12. The weight of 100 µL used for each dilution was also recorded.
GC Conditions
The use of the HT column led to the need for minor changes to the injection port configuration. The split/splitless (SSL) injection port was configured with a high temperature septum (400 °C max), 100% graphite liner o-ring and a straight 4 mm inner diameter, wool packed liner (450 °C max). Furthermore, carrier gas flow programming was used to decrease the hold-up time of analytes eluting near the column’s maximum temperature. The final nitrogen carrier gas flowrate of 3.0 mL/min does not interfere with ionization in the atmospheric pressure source because it contributes a trivial amount of additional gas in comparison to the standard nitrogen reagent gas flow of 350 mL/min. The high carrier gas flow of nitrogen necessary to achieve narrow chromatographic peaks is incompatible with GC/MS systems that use an electron ionization (EI), vacuum source due to pumping limitations and the formation of interfering azides.
|
GC system |
8890 (Agilent Technologies, Inc.) |
|
Column: |
Rxi®-5HT, 15 m x 0.25 mm ID x 0.10 µm film (Restek Corporation) |
|
Injection: |
SSL injection port, 1 µL split 10:1 at 380 °C, 4 mm ID liner with wool, 100% graphite o-ring, high temp septum (400 °C max) |
|
Temperature program: |
40 °C for 0.5 minutes, ramp to 160 °C at 14 °C/min, ramp to 395 °C at 22 °C/min, hold 11 minutes (30.75 minutes runtime) |
|
Carrier gas program: |
Nitrogen starting at 0.60 mL/min then ramped to 0.90 mL/min at 0.015 mL/min, ramp to 3.0 mL/min at 0.150 ml/min, hold to end of run |
|
Vials: |
Amber Glass Screw Top Vials with 300 µL insert (p/n: 186001130C) |
MS Conditions
One challenge to method optimization was the high cost and lack of commercially available standards for PAHs in the 314 to 424 Da mass range. This prevented the usual individual analyte-specific optimization of MS/MS conditions. Fortunately, higher mass PAHs maintain some MS/MS characteristics observed among the lower mass members of this class of compounds. Namely, the stability of PAHs results in the presence of common neutral losses of both 2 and 4 Da that require relatively high collision energies (CE) of 60–100 eV in comparison with other compound classes that commonly optimize in the range of 10–40 eV. Their stability further allows use of a high cone voltage (CV) in the source region that helps dissociate more labile potential interferences. This characteristic of PAHs has also been leveraged with electron ionization GC/MS in a “pseudo-MRM” approach using precursor-to-precursor MRM transitions.9,10 However in this work, high energy offset voltages were used in both the source and in the collision cell and were combined with the more conventional and specific precursor-to-product ion MRM transitions. Multiple steps of increasing CE were evaluated for each transition using repeat injections of SRMs. The system was also modified from the standard system set up by replacing the usual plastic tubing used for supplying N2 to the GC and MS with stainless steel tubing and fittings.
A list of 16 prospective PAHs in the range of 314 to 424 Da that have been previously reported in either NIST SRMs or bitumen extracts, was used to create multiple MRM transitions for each precursor mass. A common aspect of each of these previous publications is on-line or off-line fractionation prior to the analytical chromatographic separation. The approach demonstrated here used only sample dilution and split injection without the requirement for sample fractionation.
|
MS system: |
Xevo TQ Absolute Mass Spectrometer |
|
Ionization source: |
APGC at 150 °C |
|
Heated transfer line: |
380 °C |
|
Corona current: |
2.0 µA |
|
Cone gas: |
270 L/hr, Nitrogen |
|
Auxiliary gas: |
200 L/hr, Nitrogen |
|
Makeup gas: |
350 mL/min, Nitrogen |
|
Collision gas: |
0.40 mL/min, Nitrogen |
|
Acquisition: |
See Appendix for MRM method details |
Results and Discussion
Initial chromatographic development included evaluation of the separation of 50 PAHs in the range of 128 to 302 Da to assess the resolving power of the HT column. Four coelutions were observed in this range as compared with typical PAH separations. These were among the isomeric forms of the methylchyrsenes, benzofluoranthenes and dibenzo anthracenes. This was determined to be an acceptable reduction in separating power to achieve the elution of higher mass PAHs with a reasonable analysis time since priority was placed on determining the upper mass range of analytes that could be determined rather than on complete chromatographic resolution of all individual isomers. Alternative high temperature compatible columns with different stationary phases and column dimensions should be evaluated, with helium or nitrogen carrier gas, if resolution of these analytes is critical to the goals of an analysis.
Figure 1 shows the final separation of PAHs in the range of 128–424 Da for NIST SRM 1597. Note the significant intensity, >2e8, of the peak at 326 Da and 17.65 minutes. NIST SRM 1991 was also analyzed using the final method. The profiles of both low and HMW PAHs are distinctly different between the two SRMs as can be seen by comparing Figure 1 and Figure 2. Absolute and relative distribution differences are evident as are individual HMW PAH peaks in each sample such as the 424 Da peaks at 21.33 and 21.51 minutes, respectively.
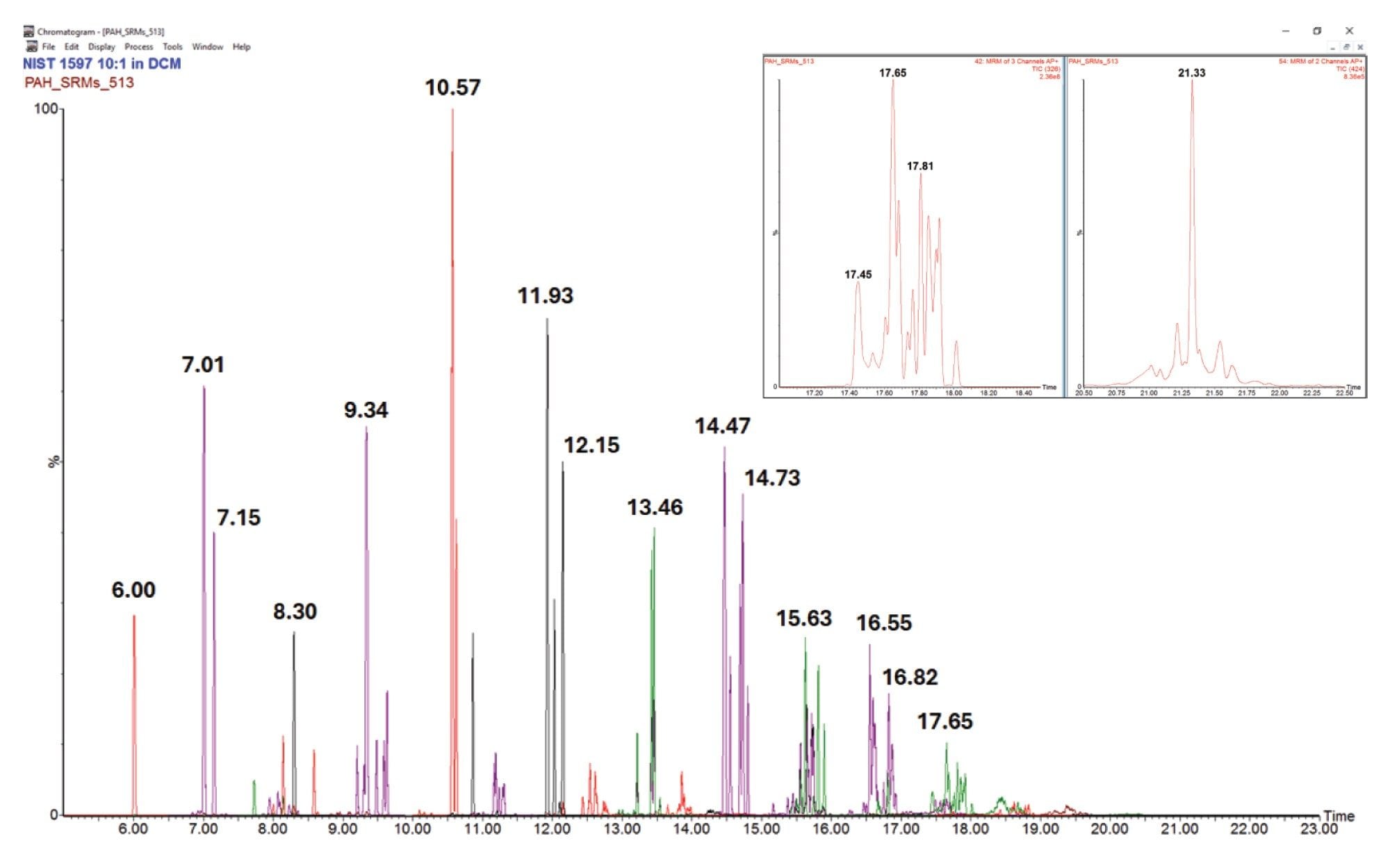
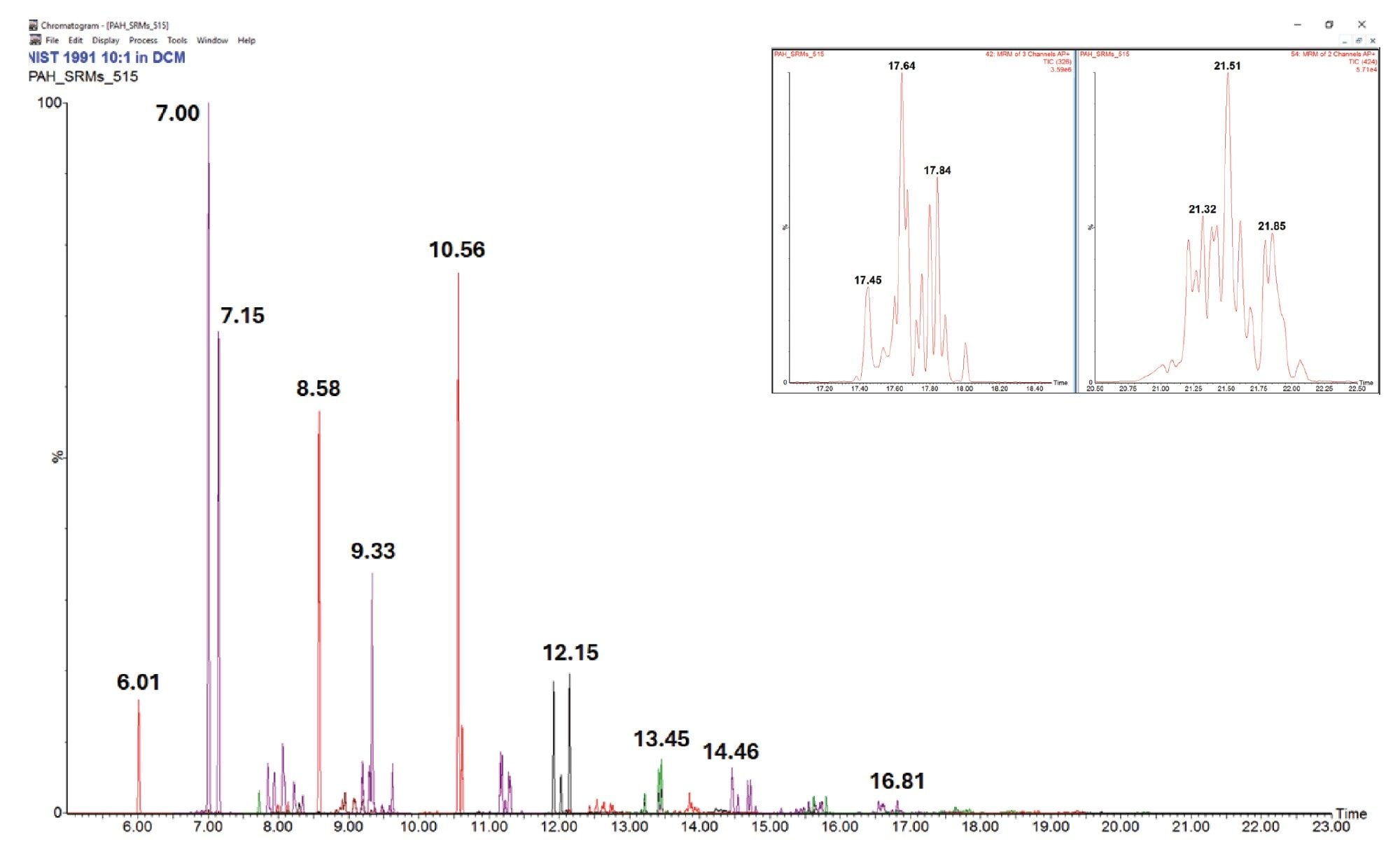
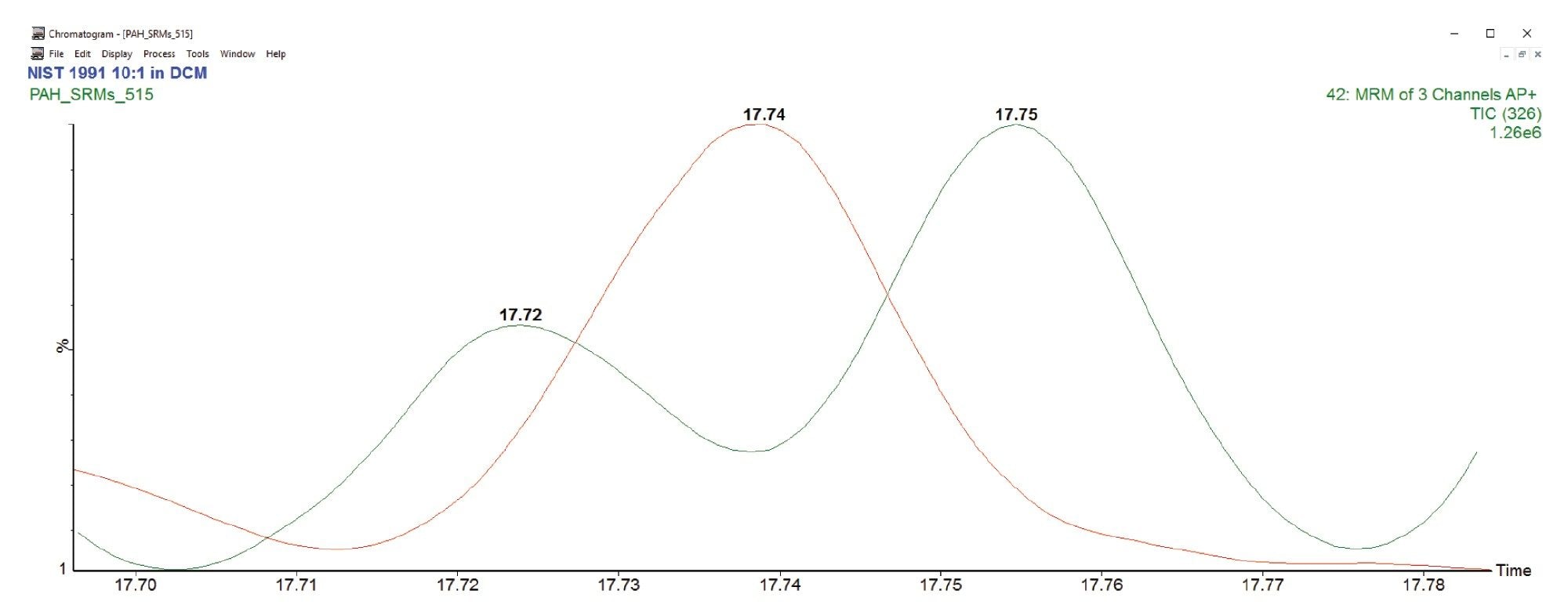
Figure 3 is an illustration of MS/MS specificity facilitating the resolution of near eluting analytes. The three analytes elute within a narrow <5 s window but are separated across MRM acquisition functions for 324 and 326 Da.
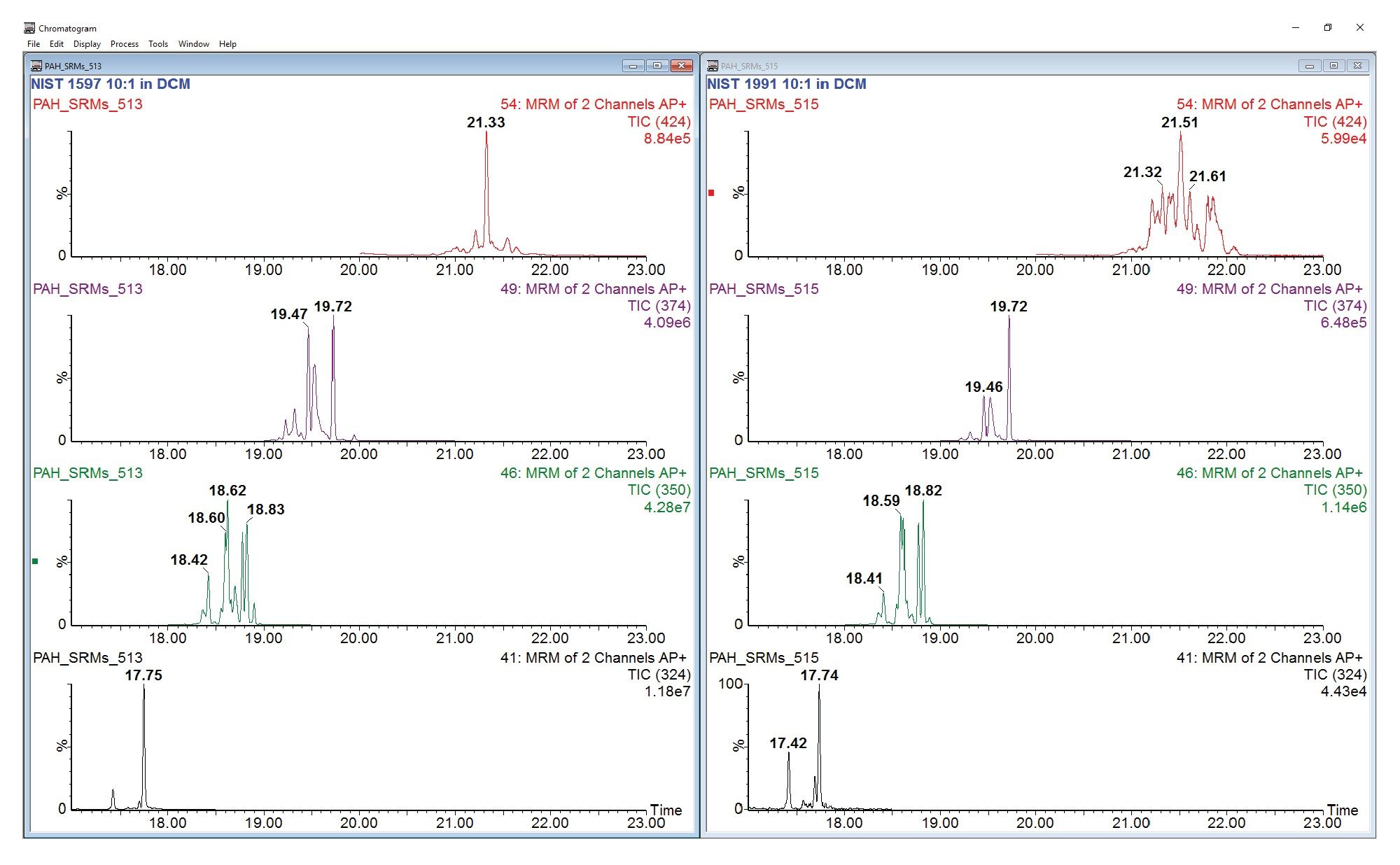
To better assess the differences between the two SRMs, the individual HMW PAH MRM TICs for 314–424 Da were compared. Figure 4 shows a selection of these chromatograms side-by-side. In its certificate of analysis, NIST 1597a is described as “… a natural, combustion-related mixture of polycyclic aromatic hydrocarbons (PAHs) isolated from a coal tar sample…”. NIST 1991 is described as a “mixed coal tar/petroleum extract”. The presence or absence of characteristic peaks as well as the relative abundances between peaks in this range demonstrate their potential use for discriminating between samples of different sources. These examples further demonstrate the successful implementation of a fast, efficient HT GC-APCI MS/MS method for the analysis of PAHs in the range of up to 424 Da. In subsequent work, this method has been applied to study the HMW PAHs in pyroplastics, a novel form of plastic pollution.11
Conclusion
Combining APGC MS/MS with high temperature GC separations allows the detection of an expanded list of PAHs. This expanded analyte list provides researchers in food and environmental applications with more comprehensive chemical characterization of PAH profiles in complex matrices with the potential for higher specificity. This increased specificity may contribute to improvements in toxicity assessment and environmental forensic analyses. Because of the analyte properties of the HMW PAHs investigated, such as high boiling point, low vapor pressure and poor water solubility, they present a promising compound class for improving the accurate chemical characterization of samples across longer periods and for samples exposed to harsh conditions. Further study of extracted samples and the evaluation of standards of individual HMW PAHs is warranted to definitively establish relationships between expanded PAH profiles and specific sample characteristics.
References
- Office of the Federal Registration (OFR). "Appendix A: priority pollutants." Fed. Reg. 47 (1982): 52309.
- Stevens, Douglas, Dorman, Frank, and Hancock Peter. “Converting Quantitative Analysis of Polycyclic Aromatic Hydrocarbons in Environmental Matrices From Electron Ionization GC-HRMS Using Helium Carrier Gas to Atmospheric Pressure Ionization GC-MS/MS Using Nitrogen Carrier Gas” Waters Corporation application note document 720008357, 2024.
- Fetzer, John C., et al. "Identification of large PAHs in bitumens from deep-sea hydrothermal vents." Polycyclic Aromatic Compounds 9.1-4 (1996): 109–120.
- Bergvall, Christoffer, and Roger Westerholm. "Determination of 252–302 Da and tentative identification of 316–376 Da polycyclic aromatic hydrocarbons in Standard Reference Materials 1649a Urban Dust and 1650b and 2975 Diesel Particulate Matter by accelerated solvent extraction–HPLC-GC-MS." Analytical and Bioanalytical Chemistry 391 (2008): 2235–2248.
- Oña-Ruales, Jorge O., Arun K. Sharma, and Stephen A. Wise. "Identification and quantification of six-ring C26H16 cata-condensed polycyclic aromatic hydrocarbons in a complex mixture of polycyclic aromatic hydrocarbons from coal tar." Analytical and Bioanalytical Chemistry 407 (2015): 9165–9176.
- Oña-Ruales, Jorge O., Yosadara Ruiz-Morales, and Stephen A. Wise. "Identification and quantification of seven fused aromatic rings C26H14 peri-condensed benzenoid polycyclic aromatic hydrocarbons in a complex mixture of polycyclic aromatic hydrocarbons from coal tar." Journal of Chromatography A 1442 (2016): 83–93.
- Samburova, Vera, Barbara Zielinska, and Andrey Khlystov. "Do 16 polycyclic aromatic hydrocarbons represent PAH air toxicity?." Toxics 5.3 (2017): 17.
- James, Bryan D., et al. "Fire and Oil Led to Complex Mixtures of PAHs on Burnt and Unburnt Plastic during the M/V X-Press Pearl Disaster." ACS Environmental Au 3.5 (2023): 319–335.
- Shang, Dayue, Marcus Kim, and Maxine Haberl. "Rapid and sensitive method for the determination of polycyclic aromatic hydrocarbons in soils using pseudo multiple reaction monitoring gas chromatography/tandem mass spectrometry." Journal of Chromatography A 1334 (2014): 118–125.
- Lian, Wenliu, et al. "Analysis of polycyclic aromatic hydrocarbons in cigarette samples using gel permeation chromatography clean-up by gas chromatography–tandem mass spectrometry." Microchemical Journal 129 (2016): 194–199.
- Megill, Cara, et al. “Gas Chromatography-Atmospheric Pressure Chemical Ionization (GC-APCI) Expands the Analytical Window for Detection of Large PAHs (≥24 Ringed-Carbons) in Pyroplastics and Other Environmental Matrices”, ACS Omega (2026): DOI: 10.1021/acsomega.5c11703.
Apprendix:
MRM transitions for the analytes included in this app note. Optimum dwell time for target compounds was set automatically using the auto-dwell function so values will vary depending on acquisition windows.
720009252, February 2026