Determination of Estrogens in Drinking and Surface Water at Low ng/L Levels by Direct Injection Using UPLC-MS/MS
Ofure Idialua, Peter Hancocka, Claudia Rathmannb, Frank Dormanc
a Waters Corporation, Wilmslow, United Kingdom
b Waters Corporation, Eschborn, Germany
c Waters Corporation, Milford, MA, United States
Published on February 08, 2026

Abstract
This application note describes the analysis of 17α-ethinylestradiol (17αEE2), 17α-estradiol (17αE2), 17β-estradiol (17βE2), estrone (E1) and estriol (E3) in drinking and surface water using the ACQUITY™ Premier System coupled to Xevo™ TQ Absolute Mass Spectrometer. Excellent sensitivity was achieved for the quantification of all target estrogens in drinking and surface water at trace levels (1 ng/L). Baseline separation was achieved for 17αE2 and 17βE2. Excellent retention, resolution, and peak shapes were obtained for all the target analytes within a run time of 8 minutes, indicating that this method can be used to accurately quantify target estrogens in water.
Benefits
- Excellent sensitivity was achieved for target estrogens at trace levels (1 ng/L) by direct injection of drinking and surface water samples.
- Method efficiency that allows reduced sample extraction volumes to be used in laboratories that work in compliance with the revised EU Water Framework Directive (WFD) to reach ultra-trace levels (pg/L) set by Directive 2008/105/EC on environmental quality standards.
- Baseline separation was achieved for 17αE2 and 17βE2. Excellent retention, chromatographic resolution and method performance was achieved for all target estrogens in water samples within an 8-minute run cycle without the need for derivatization prior to chromatographic analysis.
Introduction
Estrogens are steroid hormones that present a risk to the aquatic environment and human health because of their interference with the endocrine system function.1,2 Effluent discharge from wastewater treatment plants, disposal or run offs of animal waste in freshwater have been identified as major sources of estrogens in the aquatic environment.1,2 17α-ethinylestradiol (17αEE2), 17α-estradiol (17αE2), 17β-estradiol (17βE2), estrone (E1), and estriol (E3) are the most abundant and widely monitored estrogens.3,4,5
The Directive 2008/105/EC recently proposed extremely low annual average environmental quality standard (AA-EQS) target values for estrogens in inland surface waters (0.017 ng/L for 17αEE2, 0.18 ng/L for 17βE2 and 0.36 ng/L for E1).6 Analytical laboratories have found it quite difficult to reach the required levels due to the low response obtained for estrogenic compounds via LC-MS/MS analysis. Pretreatment processes such as derivatization, extraction, enrichment, and clean up are often needed to improve method sensitivity. Possible methods for the determination of selected estrogens in whole water samples are described in ISO 13646:2025. Sample preparation involves the use of large sample volumes for extraction and enrichment prior to analysis which is time consuming and generates a lot of chemical waste.7 Derivatization of target analytes prior to analysis is also time consuming.8 Thus, there is a need to establish an efficient method with increased sensitivity for the quantification of estrogens at ultra-trace levels in water.
In this application note, a method for analysis of 17αEE2, 17αE2, 17βE2, E1, and E3 in drinking and surface water is presented using the ACQUITY Premier System coupled to Xevo TQ Absolute Mass Spectrometer. This method facilitates the quantification of estrogens at ultra-trace levels without the need for derivatization of the target analytes and use of large sample volumes for extraction prior to analysis. The Xevo TQ Absolute Mass Spectrometer is extremely sensitive for quantification of all the target estrogens in drinking and surface water at low ng/L levels. In addition to the regulated analytes, E3 and 17αE2 are included in the method with baseline separation of 17αE2 and 17βE2, all within a run time of 8-minute that allows for high-throughput analysis.

Experimental
Method performance was assessed using analytical standards prepared in drinking and surface water and analyzed via direct injection of matrix-matched standards. Drinking water (mineral water) was purchased from a UK retail outlet and stored in its original container. Surface water samples were collected from Wilmslow, UK and stored in plastic containers. All samples were stored in refrigerated conditions.
Matrix-matched standards were prepared in drinking and surface water at concentrations ranging from 1–500 ng/L and used for analysis. Internal standards were not included in this method. Blank samples of drinking and surface water were evaluated for the presence of target analytes.
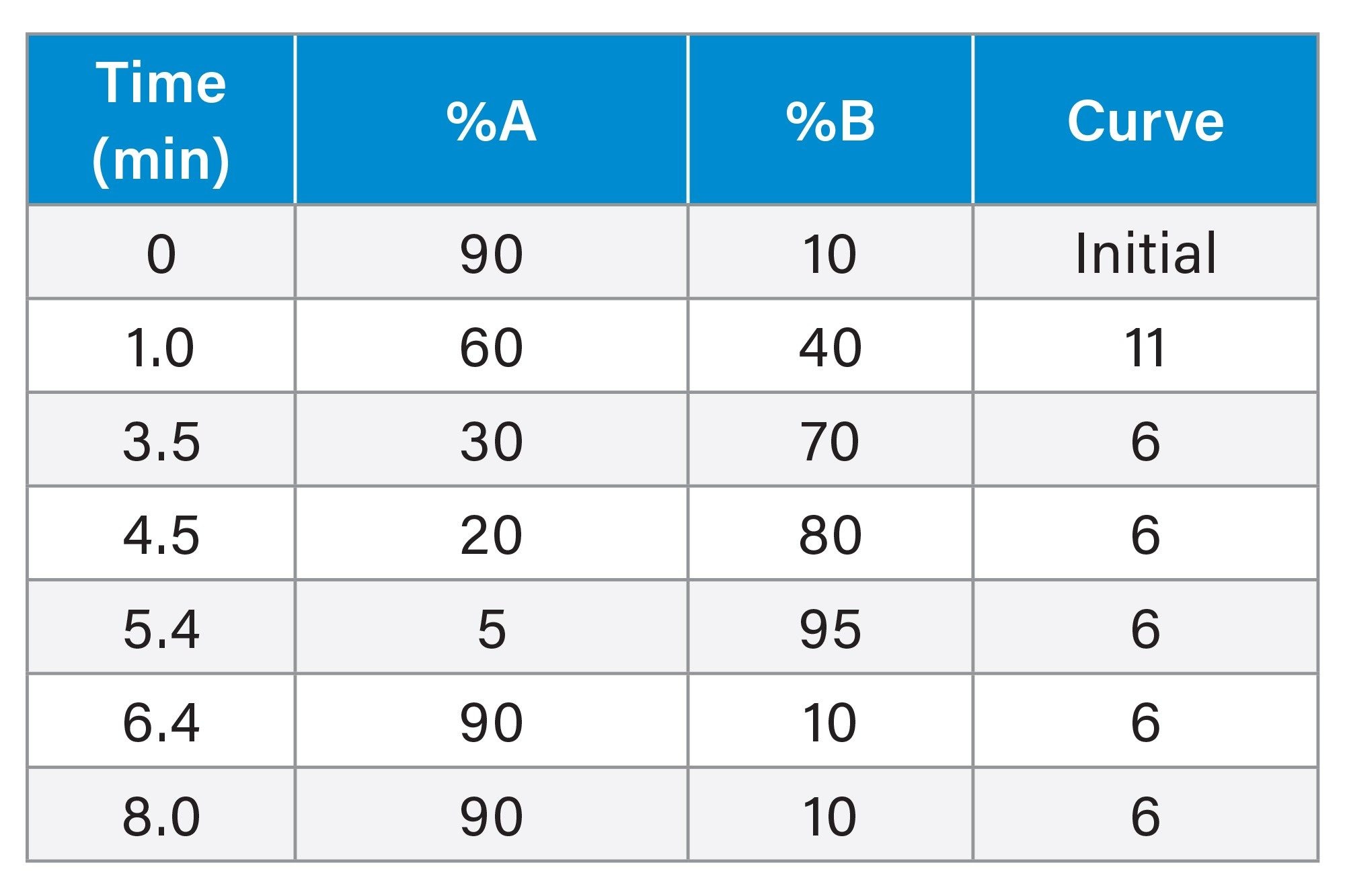
LC Conditions
|
LC system: |
ACQUITY Premier System with Flow-Through Needle (FTN) Sample Manager and 250 µL extension loop (p/n: 700011706) |
|
|
Column: |
ACQUITY™ Premier BEH™ C18 Column, 2.1 x 150 mm, 1.7 µm (p/n: 186009454) |
|
|
Vials: |
TruView™ pH Control LCMS Certified Clear Glass, 12×32 mm (p/n: 186005669CV) |
|
|
Mobile phase A: |
0.01 mM ammonium fluoride in water |
|
|
Mobile phase B: |
Acetonitrile: methanol (50:50) with 0.01 mM ammonium fluoride |
|
|
Flow rate: |
0.3 mL/min |
|
|
Column temperature: |
50 °C |
|
|
Sample temperature: |
10 °C |
|
|
Injection volume: |
100 µL |
Gradient Table
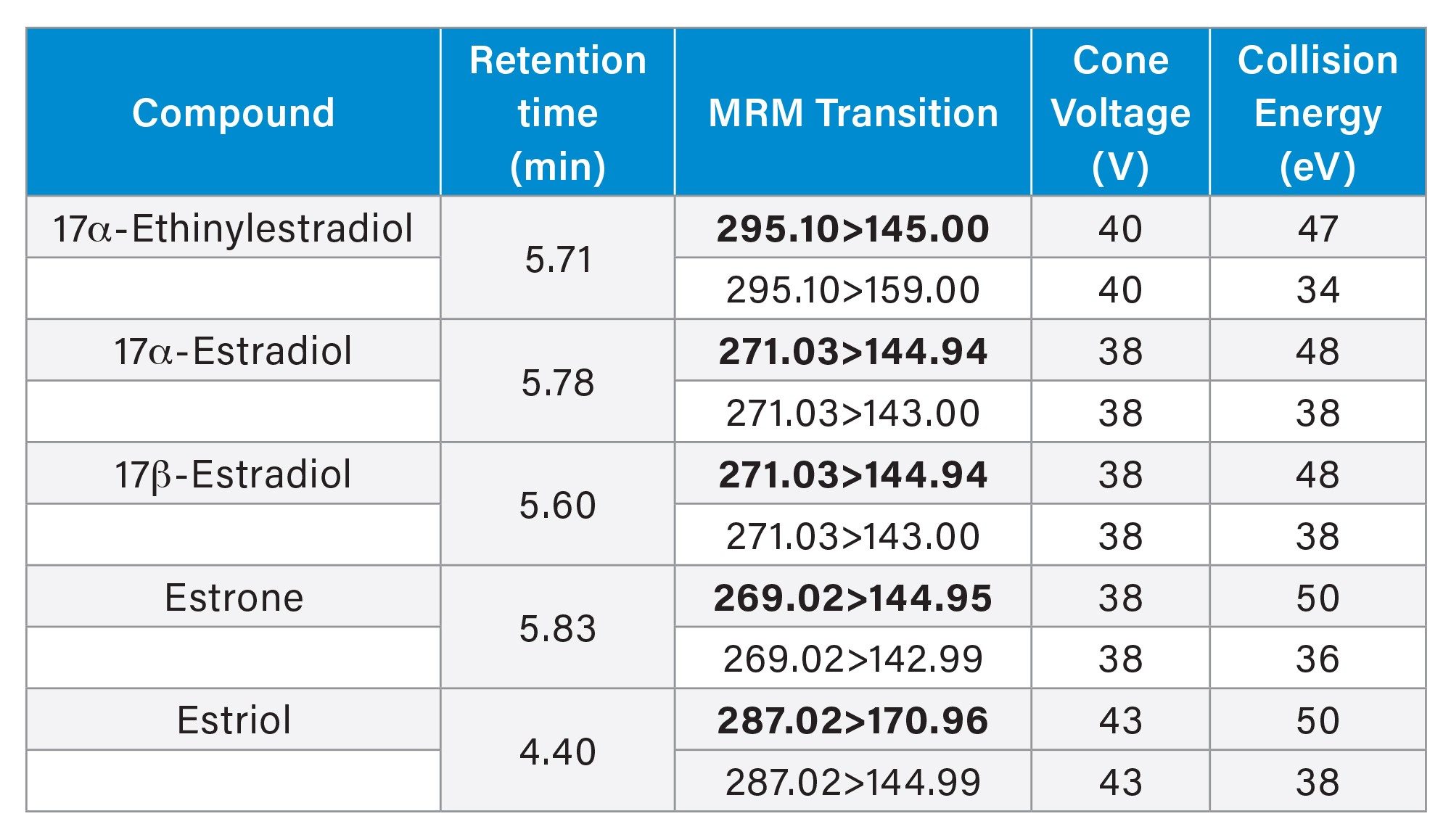
MS Conditions
|
MS system: |
Xevo TQ Absolute Mass Spectrometer |
|
Ionization mode: |
ESI - |
|
Acquisition mode: |
MRM |
|
Capillary voltage: |
2 kV |
|
Desolvation temperature: |
600 °C |
|
Desolvation gas flow: |
1200 L/hr |
|
Cone gas flow: |
150 L/hr |
|
Source temperature: |
150 °C |
|
Software: |
waters_connect™ for Quantitation Software |
Results and Discussion
Method performance was successfully evaluated using matrix-matched calibration curves in drinking and surface water. Excellent sensitivity (1 ng/L) and selectivity were achieved for the analysis of the five target estrogens.
The AA-EQS values range from 0.017–0.36 ng/L for target estrogens in water. The high sensitivity achieved by this method allows lower enrichment factors and consequently smaller sample volumes to be used in laboratories that aim to meet the EU WFD requirements for 17αEE2, 17βE2 and E1 at low ng/L EQS levels in surface water thereby reducing time spent and waste generated.
Figure 2 shows chromatograms obtained for all estrogens in surface water at mid-level. Figures 3 and 4 show chromatograms obtained for all estrogens in drinking and surface water at the lowest level reported respectively. None of the target estrogens exceeded the method lower limit of quantitation (LLOQ) at 1 ng/L in the analyzed samples of drinking and surface water. Matrix effect was evaluated for drinking and surface water. Matrix effects ranging from 16–29% were observed for target estrogens in drinking and surface water.
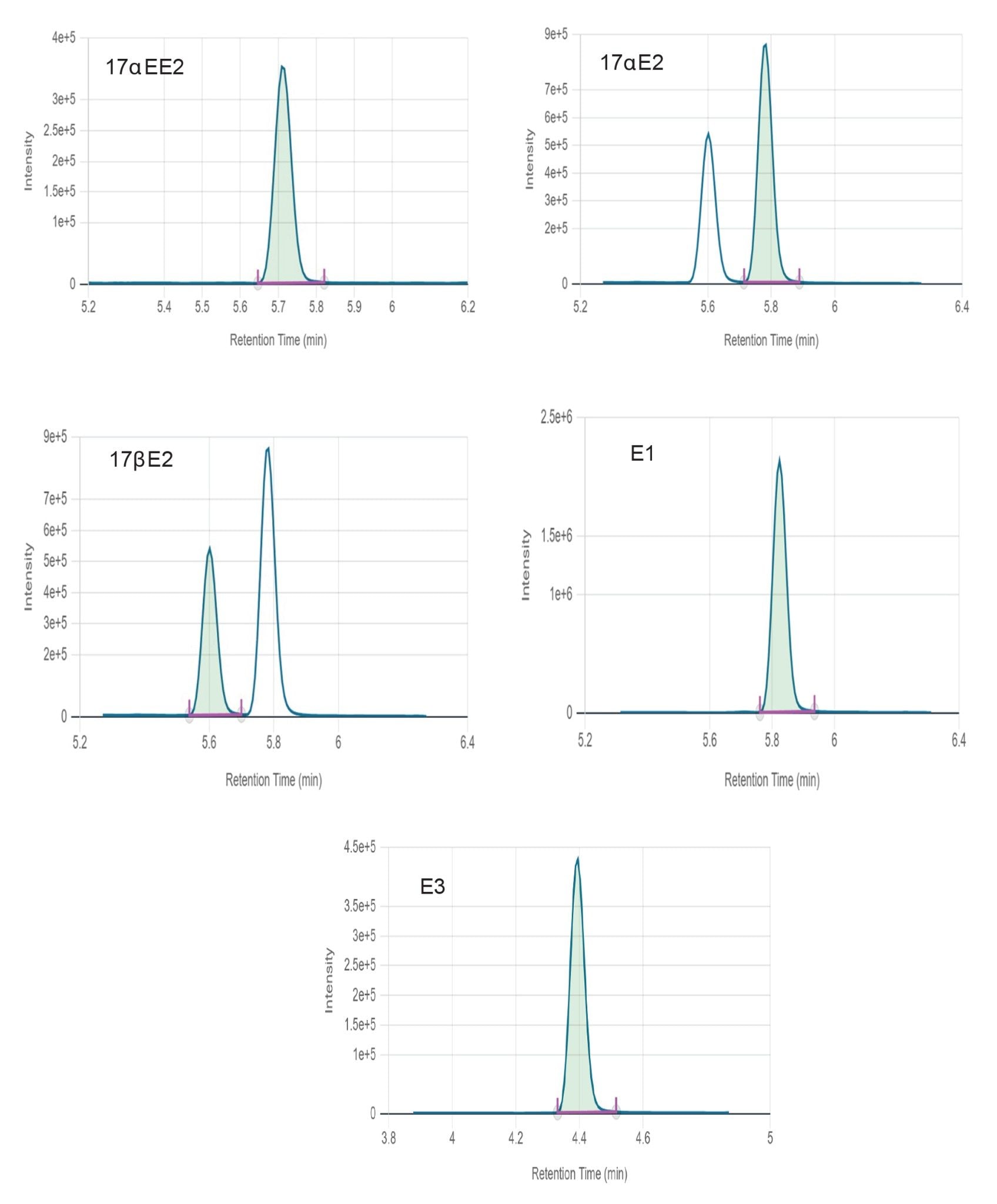
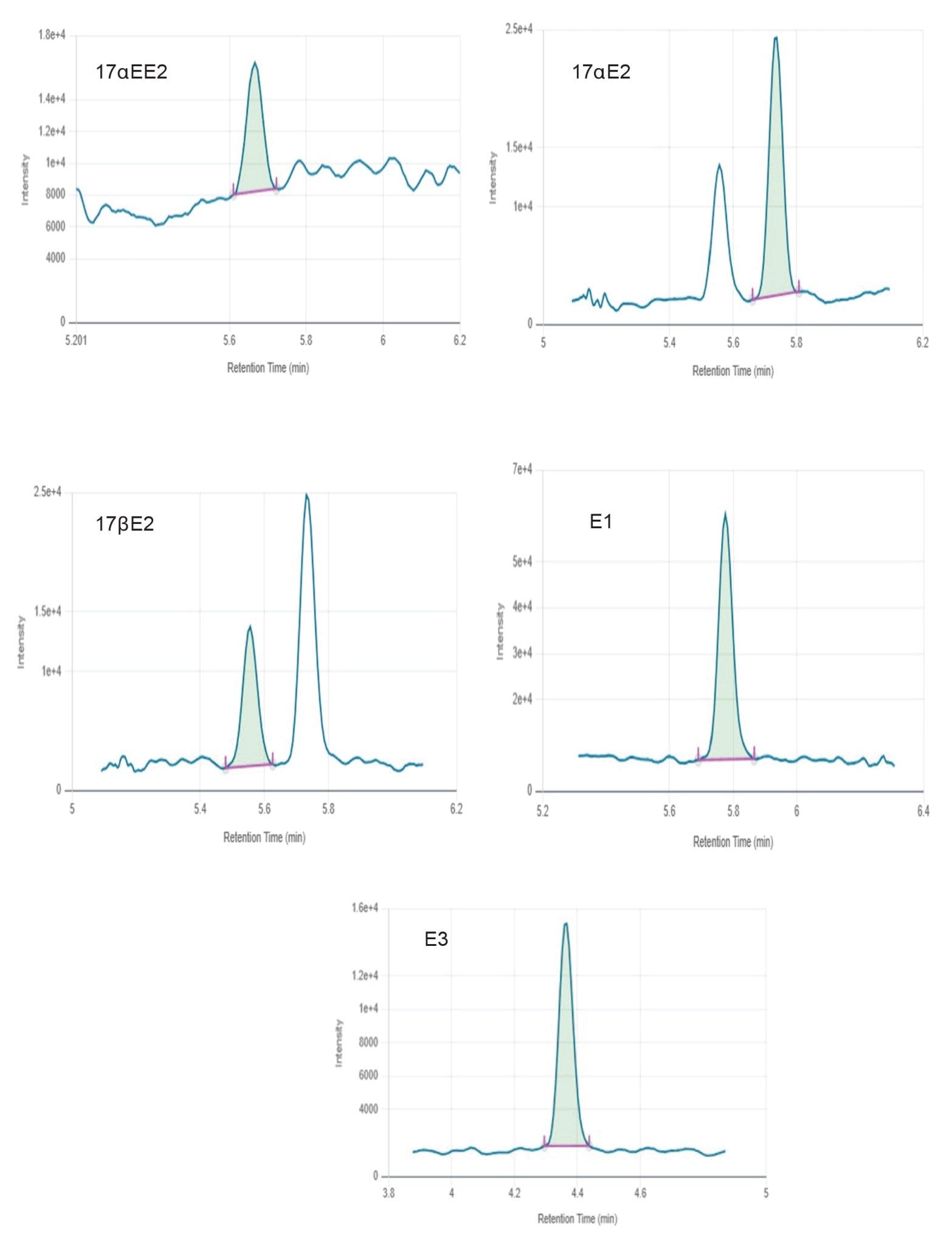
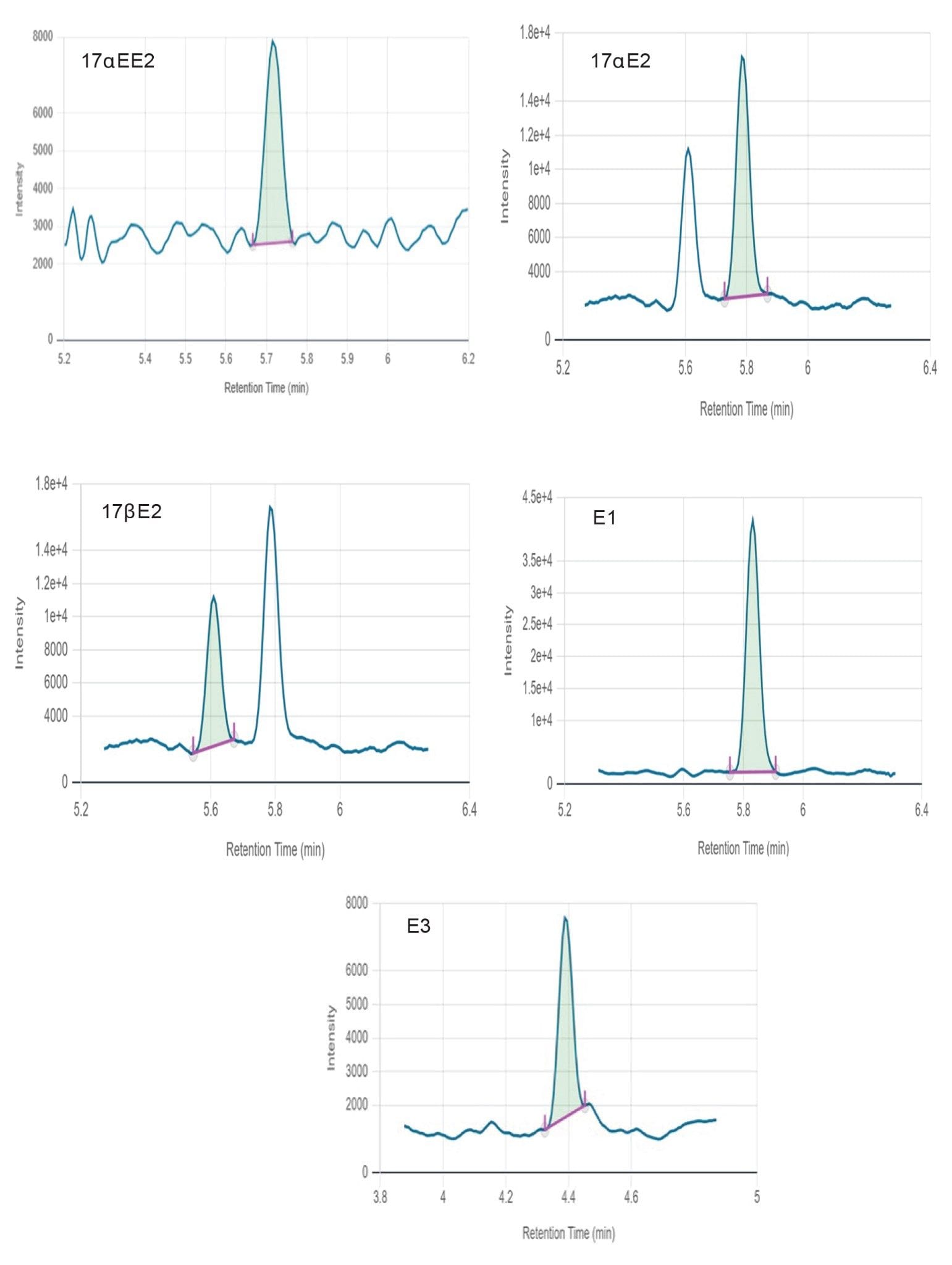
A 150 mm analytical column was selected to facilitate baseline separation of 17αE2 and 17βE2 epi-isomers. Chromatographic resolution was achieved for all target analytes. Response was linear over the range of 1–500 ng/L for all analytes in drinking and surface water as shown in Figures 5 and 6, respectively.
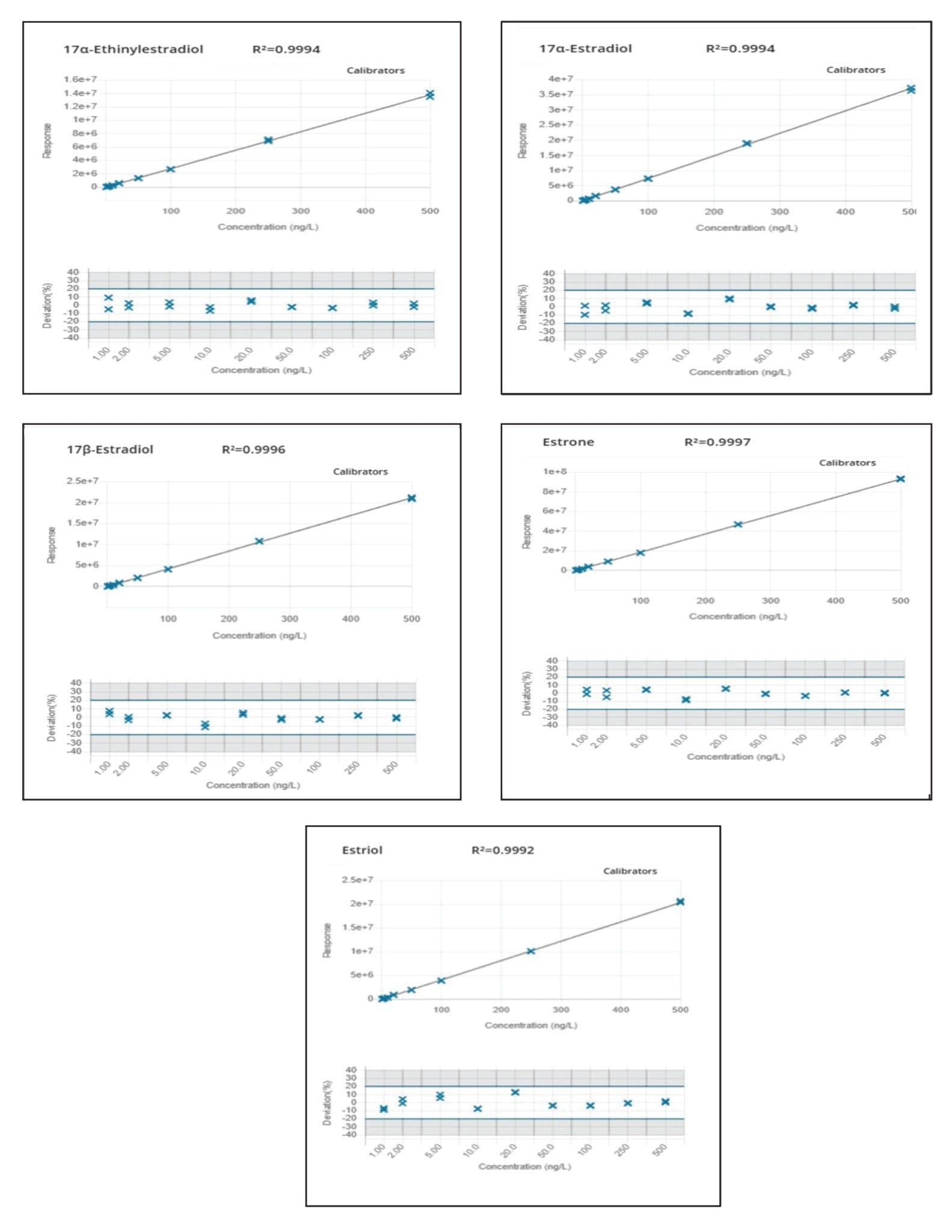
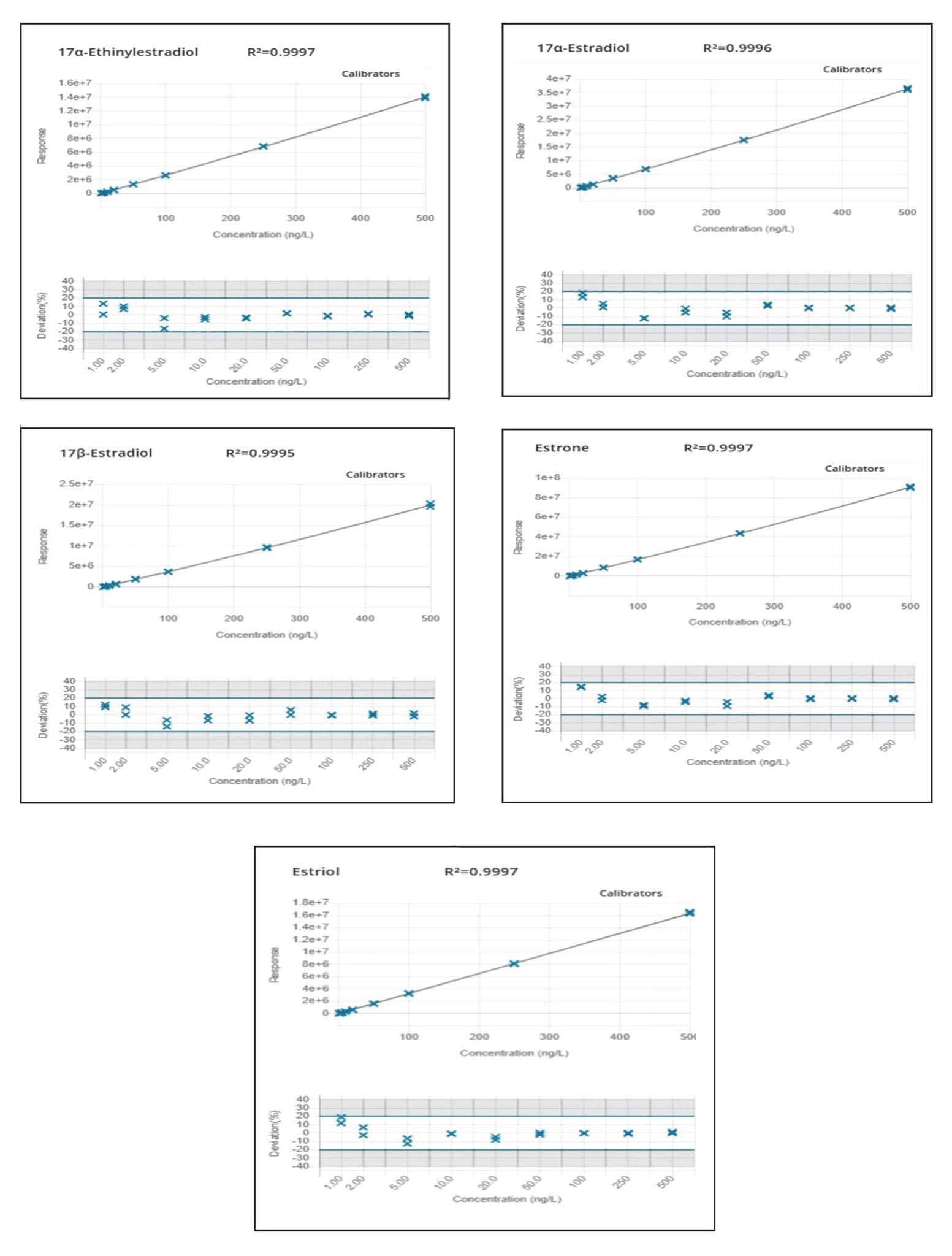
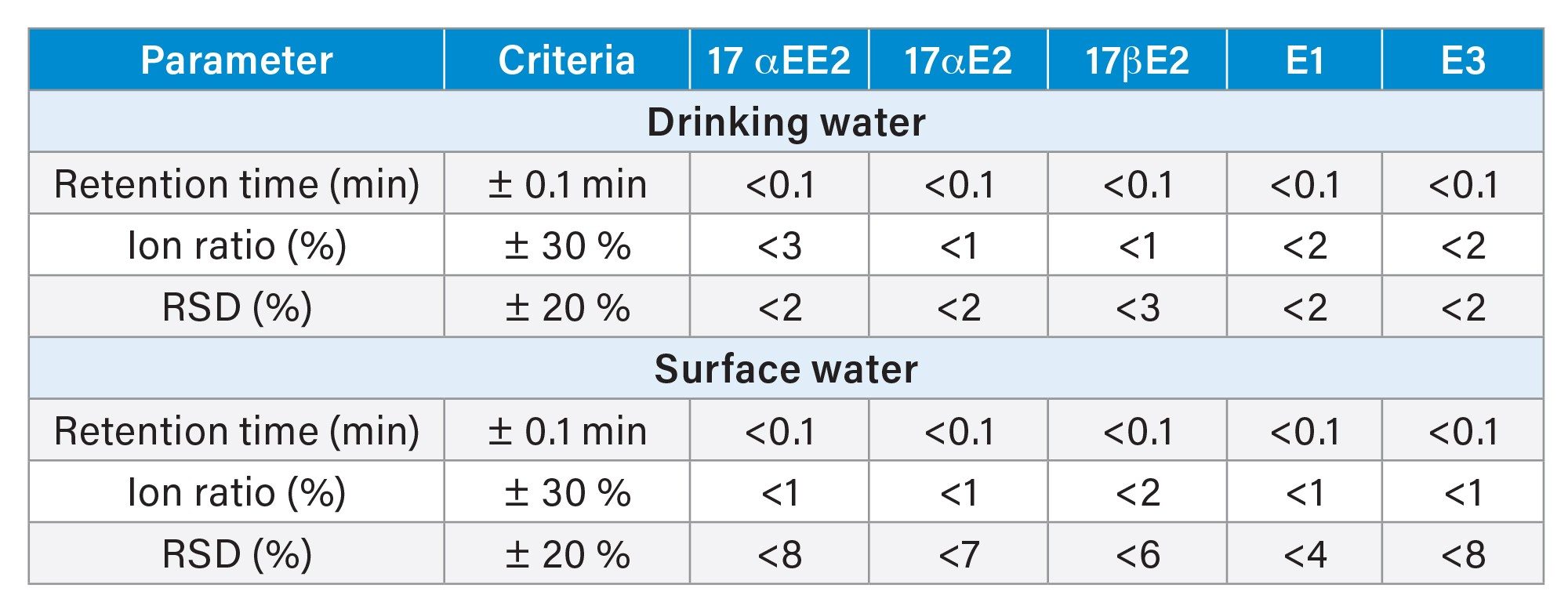
Excellent peak widths and peak shapes were obtained for all the target estrogens. Retention times were stable for all analytes throughout the run and within the required tolerances. Ion ratios and %RSD in repeated injections were also within the required tolerances demonstrating good repeatability as summarized in Table 2. All the target analytes could be accurately quantified at low ng/L levels (method LLOQ) indicating that this method can be employed for the quantification of target estrogens in water at the EU EQS levels.
Conclusion
Excellent sensitivity (with an LOQ of 1 ng/L) as well as chromatographic retention and resolution have been achieved for all estrogens using the ACQUITY Premier System and Xevo TQ Absolute Mass Spectrometer without the need for derivatization prior to analysis. The high sensitivity achieved by this method allows lower enrichment factors and consequently smaller sample volumes to be used in laboratories that aim to meet the EU WFD requirements for 17αEE2, 17βE2 and E1 at low ng/L EQS levels in surface water, thereby reducing time spent and waste generated.
Method performance was successfully evaluated for the five target analytes using matrix-matched standards prepared in drinking and surface water. Matrix blanks in drinking and surface water were free of target estrogens above the method’s detection limit. Matrix effects ranging from 16–29% were observed for target estrogens in drinking and surface water. Small peak widths and symmetric peak shapes were obtained using a 150 mm analytical column with low flow rate that also enabled baseline separation of 17αE2 and 17βE2 epi-isomers. Coefficients of determination, retention times, ion ratios, and %RSD values were all within the required tolerances indicating that this method can be used to accurately quantify target estrogens in different types of water samples at ultra-trace levels to ensure water quality standards are met in Europe.
References
- Junyue W, Yang C, Zikang Z, Jiayi Y, Wanting L, Xuwen C, Yanzheng G. Occurrence, fate and impact of steroidal estrogens and xenoestrogens in plants, A review. Journal of environmental management 2025.
- Fan H, Fan G, Chaoran L, Luiza C. Photodegradatiion of free estrogens driven by UV light: Effects of operation mode and water matrix. Science of the total environment 2022.
- Alex G, Katherine N, Phillipe C, Sebastien R. Development and validation of a method for determining estrogenic compounds in surface water at the ultra-trace level required by the EU Water Framework Directive Watchlist. Journal of Chromatography A 2020.
- Marie S, Jakub J, Ludek S, Zuzana T, Barbora J, Eliska S, Olga L, Klara H. Potential risk of estrogenic compounds produced by water blooms to aquatic environment. Chemosphere 2023.
- Deisi S, Ann M, Gemima A. Estrogenic compounds in drinking water; A systematic review and risk analysis. Chemospere 2024.
- Directive 2022/540/EC of the European Parliament and of the Council of 26 October 2022 amending Directive 2000/60/EC Establishing a Framework for Community Action in the Field of Water Policy, Directive 2006/118/EC on the protection of groundwater against pollution and deterioration and Directive 2008/105/EC on environmental quality standards in the field of water policy.
- Ross E, Wuyts B, Boag A. Quantitative Analysis of Natural and Synthetic Estrogens in Surface and Final Effluent Waters at Low ppq Levels Using UPLC-MS/MS. Waters Application Note. 720006030. November 2020.
- Szilard M, Gyozo K, Pal P. Determination of steroid hormones in water samples by liquid chromatography electrospray ionization mass spectrometry using parallel reaction monitoring. Microchemical Journal 2022.
720009193, December 2025