This application note evaluates the use of the Xevo TQ MS tandem quadrupole mass spectrometer, equipped with a novel collision cell design, for the detection and identification of PDE5 adulterants in herbal supplements.
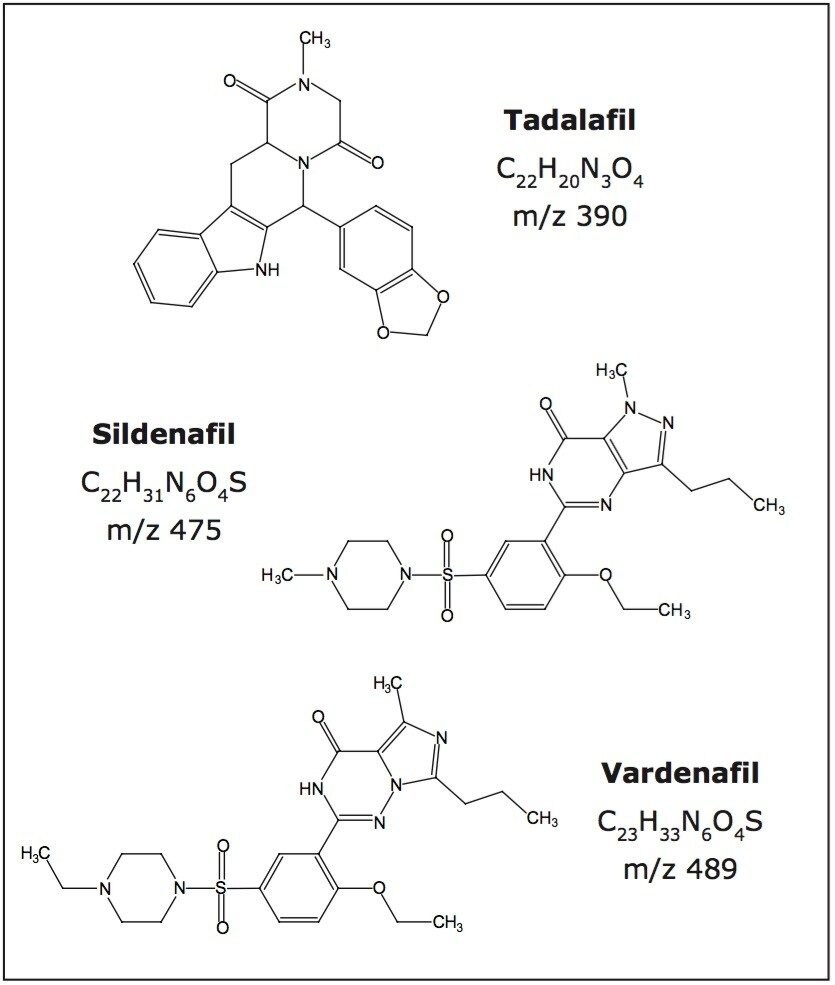
There are currently three synthetic phosphodiesterase type 5 (PDE5) inhibitors that have been approved by the U.S. Food and Drug Administration for the treatment of erectile dysfunction (ED): sildenafil citrate (brand name Viagra), vardenafil hydrochloride (brand name Levitra), and tadalafil (brand name Cialis). Their chemical structures are shown in Figure 3. These medications can be obtained legally by prescription from a licensed physician and they should be used under medical supervision.
It has been extensively reported that there have been adulterations to herbal dietary supplements (HDS) with synthetic drugs.1-4 Natural aphrodisiacs are heavily advertised on the Internet. Some claim to enhance sexual function as natural alternatives to the three approved, synthetic PDE-5 inhibitors. Recently, there have been reports that these supposed natural alternatives have actually been illicitly adulterated with one of the pharmaceutical ED drugs or their structurallymodified analogues.5-16
When an HDS product is labelled as natural, there is also a sense of security that it is safe to use. Given that an HDS could contain undeclared synthetic drugs and that it can easily be obtained over the Internet, without prescription, there is the potential for a threat to public health.
It is therefore very important that analytical methods be able to rapidly detect both known PDE5 adulterants and potentially new analogues that have not yet been reported.
In this paper, we evaluate the use of the Xevo TQ MS tandem quadrupole mass spectrometer, equipped with a novel collision cell design, for the detection and identification of PDE5 adulterants in herbal supplements.

On a conventional tandem quadrupole mass spectrometer, the search for unknowns generally requires multiple injections: one injection in full-scan LC-MS mode, followed by a second injection for targeted LC-MS/MS experiments. This increases the time required to obtain necessary data, in addition to the time the analyst needs to construct MS/MS methods.
The Survey Scan feature of the Xevo TQ MS allows intelligent switching from LC-MS to LC-MS/MS data modes in a single run, thus improving productivity. Conventional MS or ScanWave MS scanning experiments can be used to trigger MS/MS experiments in real time as the peaks are eluting from the LC column. Conventional product ion or enhanced product ion spectra (ScanWave) data can be generated for all the components present in these complex samples.
In ScanWave mode, duty cycle improvements result in signal enhancement in scanning acquisition modes, which facilitates the detection of low-level adulterants.17 A more targeted screen can also be performed, using parent ion or neutral loss spectral acquisition, to screen for compounds that have common structural features.
|
LC system: |
ACQUITY UPLC |
|
Solvent delivery: |
ACQUITY UPLC Binary Solvent Manager |
|
Sample delivery: |
ACQUITY UPLC Sample Manager |
|
Column: |
ACQUITY UPLC BEH C18 , 2.1 x 100 mm, 1.7 μm (P/N 186002352) |
|
Column temp.: |
60 °C |
|
Sample temp.: |
4 °C |
|
Injection volume: |
5 μL |
|
Flow rate: |
550 μL/min |
|
Mobile phase A: |
10 mM Ammonium acetate in water, pH 6.7 |
|
Mobile phase B: |
50:50 Methanol/acetonitrile |
|
Gradient: |
0 to 9 min, 20 to 52% B, hold at 52% B until 10.5 min, 10.5 to 12.5 min 85% B, hold at 85% B until 15 min, then return to initial conditions |
|
MS system: |
Xevo TQ MS |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
3.0 kV |
|
Cone voltage: |
35 V |
|
Collision energy: |
20 to 40 eV |
|
Desolvation temp.: |
400 °C |
|
Desolvation gas: |
800 L/Hr |
|
Collision gas: |
0.15 mL/min |
|
Source temp.: |
150 °C |
|
Acquisition range: |
100 to 700 amu |
|
Scan speed: |
5000 amu/sec |
|
Range: |
210 to 400 nm |
|
Sampling rate: |
20 points/sec |
The crushed tablet sample or powdered capsule contents were transferred quantitatively to a 100 mL volumetric flask, and sonicated in 50:50 methanol/water for 20 minutes. A representative aliquot was transferred to a centrifuge tube and centrifuged at 3000 RPM for 10 minutes. A portion of the supernatant was placed in a vial and used for analysis by LC-MS.
Five products were obtained over the Internet (Figure 2) and analyzed by UPLC-MS/MS using data-directed analysis on the Xevo TQ MS. Four capsules and one tablet supplement were purchased. All five were found to be adulterated, containing sildenafil and/or tadalafil, or analogues of these drugs.
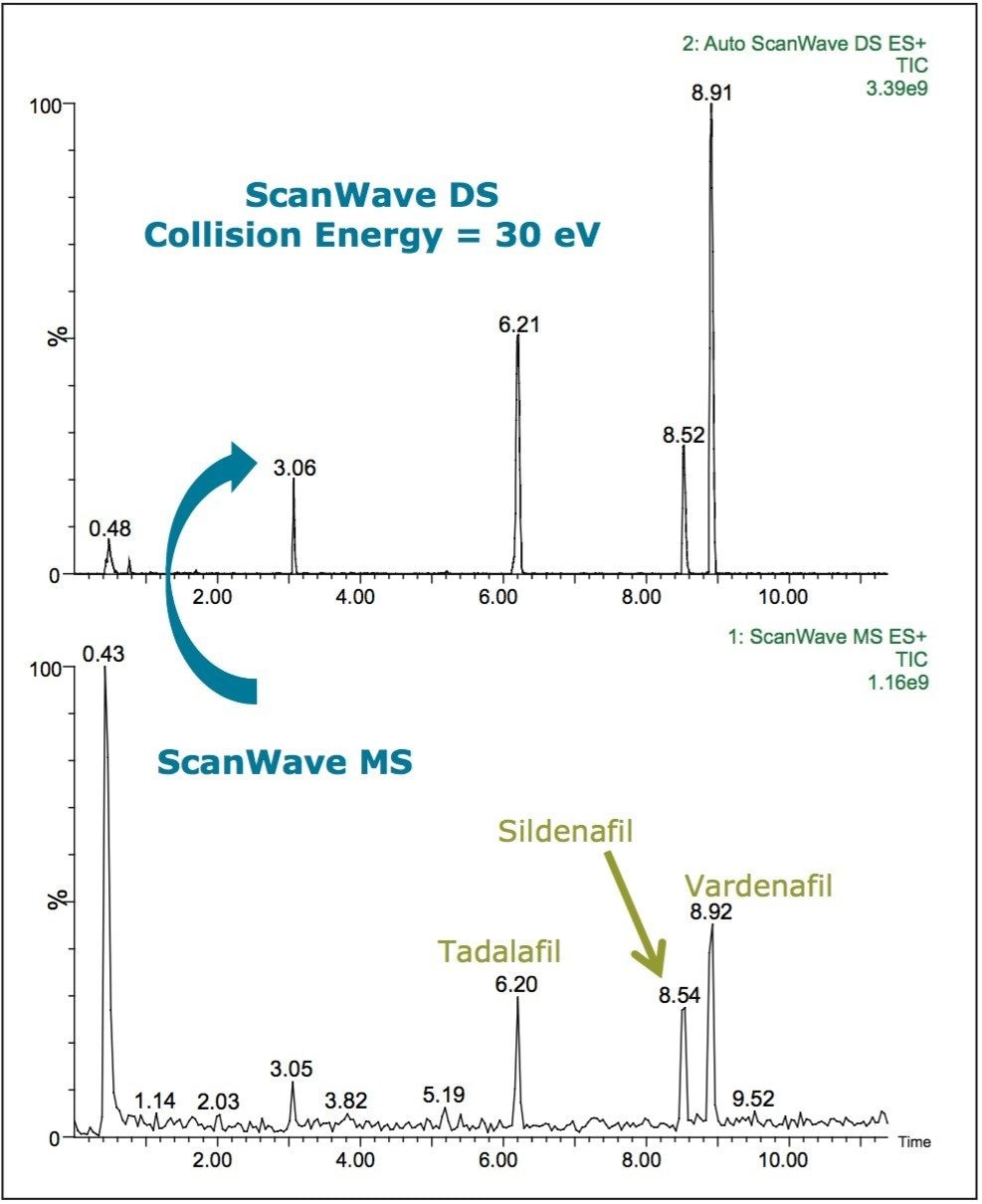
A survey scan of a standard solution of sildenafil, vardenafil, and tadalafil is shown in Figure 4. The primary function is ScanWave MS, which switches to ScanWave DS when a peak is detected to acquire a full MS/MS spectrum from 50 to 700 amu at a collision energy of 30 eV.
In the spectrum for tadalafil (Figure 5) from the peak eluting at retention time (RT) 6.21 min, m/z 390, several diagnostic fragments are seen: m/z 268, m/z 262, m/z 169, and m/z 135.
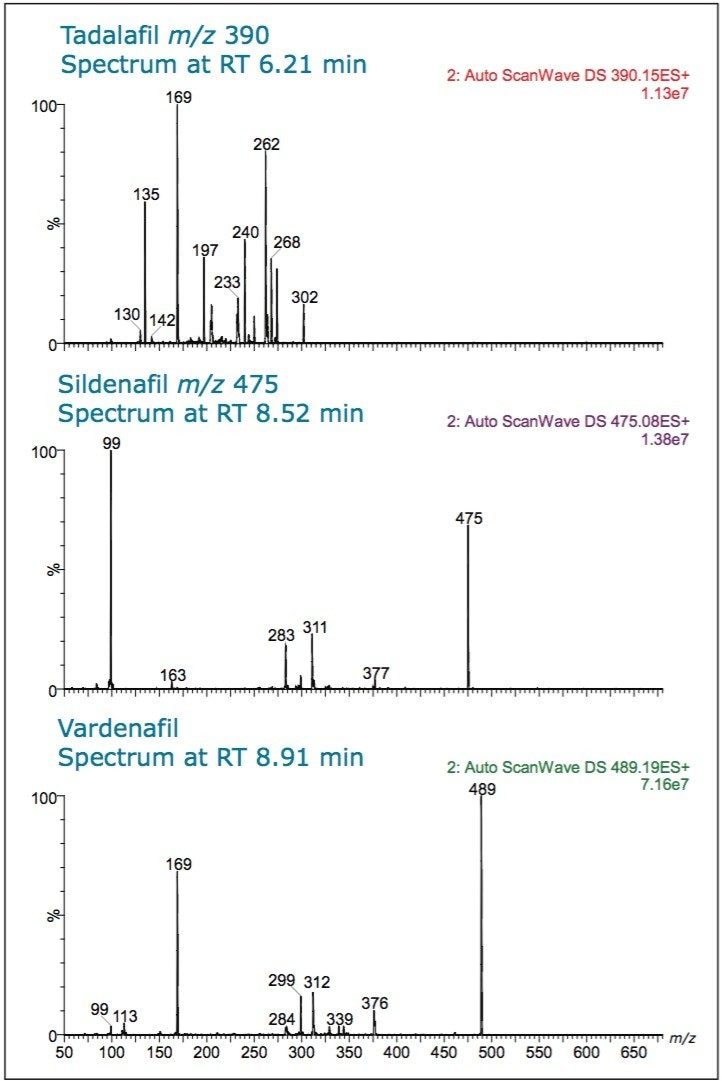
In the spectrum for sildenafil, RT 8.52 min, fragments m/z 377, m/z 311, m/z 283, and m/z 99 are seen.
The spectrum for vardenafil, at RT 8.91 min, shows MS/MS fragments of m/z 312, m/z 299, m/z 169, and m/z 99. These ions can be used to confirm peak identity.
Sample 1 was found to be adulterated with tadalafil, as can be seen in Figures 6 and 7. The spectrum of a tadalafil standard at a collision energy of 35 eV (Figure 7 inset) exactly matches the spectrum taken at 6.18 minutes in the Sample 1 chromatogram. The survey scan was set up to collect two MS/MS functions, one at a collision energy of 25 eV and a second at 35 eV. This is advantageous in the analysis of complex samples, where the range of collision energies required to obtain useful structural information can vary.
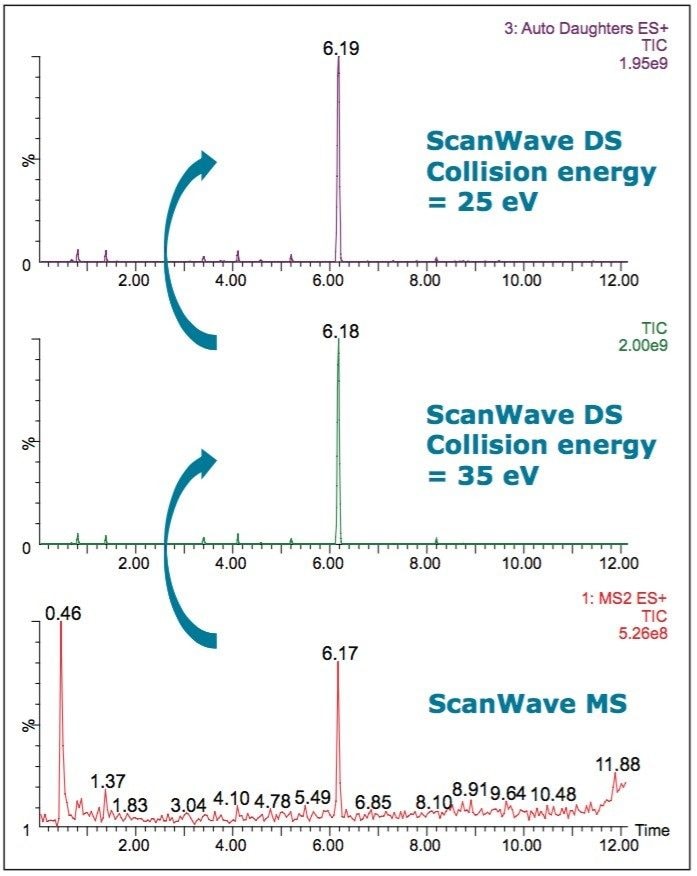
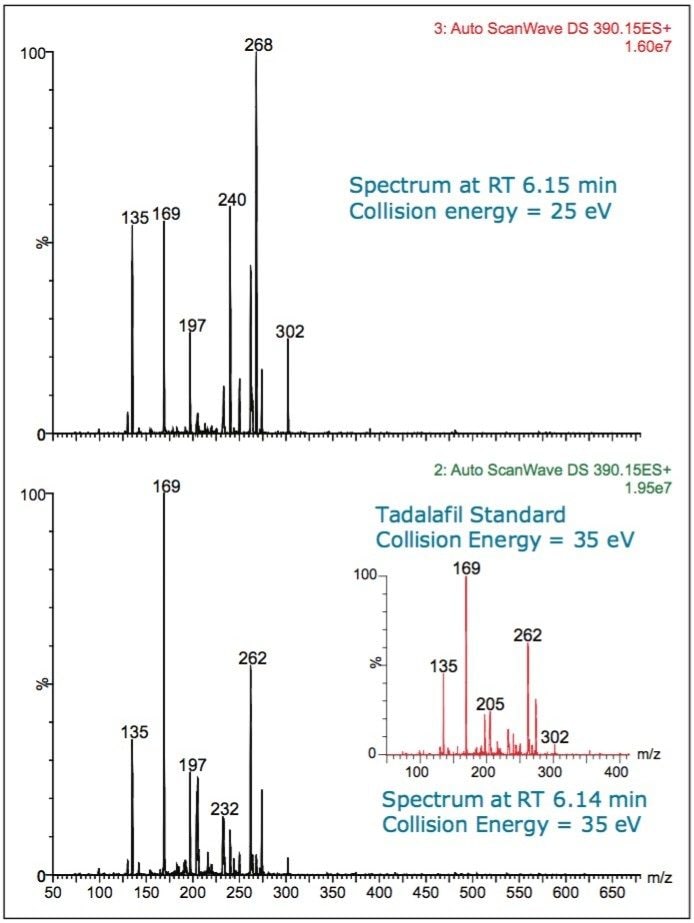
The signature fragments of tadalafil, m/z 268, m/z 169, and m/z 135, can be seen in the spectra.
This sample indicated the presence of many natural ingredients, including Dioscorea spinosina (wolfberry fruit), Glycyrrhiza glabra (liquorice root), as well as others. Neither the patient information nor the packaging declared the presence of tadalafil in the product.
Sample 3, in tablet form, was found to be adulterated with tadalafil and sildenafil. A survey function is shown in Figure 8 and selected spectra are shown in Figure 9. The spectra taken at RT 6.18 min and RT 8.5 min match with the spectra from a standard mix of tadalafil and sildenafil (see Figure 4). The survey scan was set up to collect two collision energy functions, one at 25 eV and a second at 35 eV. The expected diagnostic fragments were apparent in the sildenafil spectrum m/z 311, m/z 283, and m/z 99.
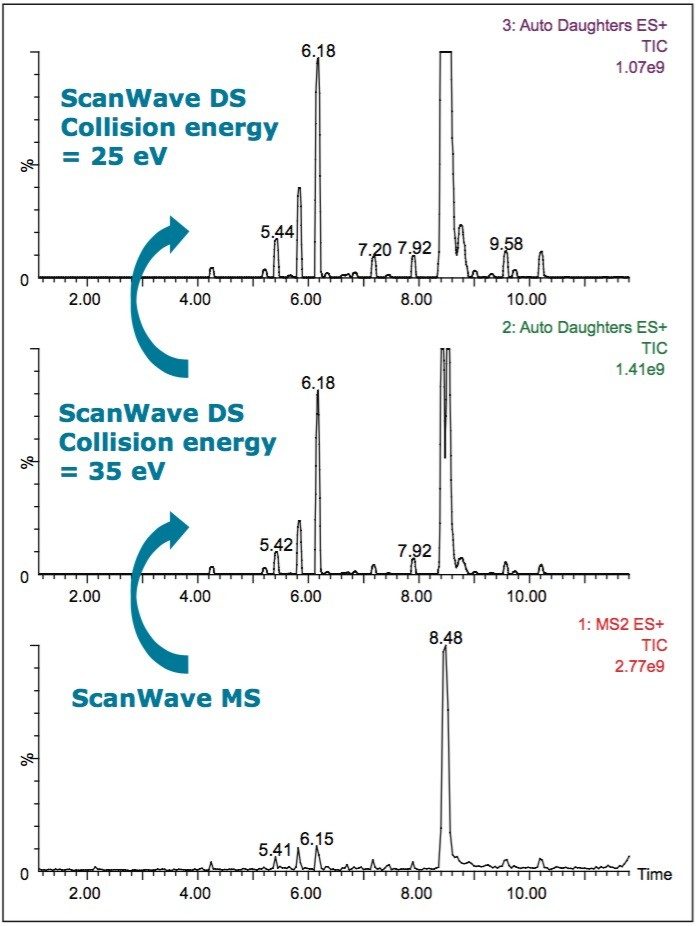

In addition to finding known compounds, it can be seen from the MS/MS fragmentation patterns in Figure 9 that there are other potentially related impurities in the sample. Many of them are above the ICH guideline’s reporting threshold of 0.05% of the active pharmaceutical ingredient (API) peak (measured at 230 nm). In spectrum 1 shown in Figure 9, the precursor mass of m/z 503 gives rise to MS/MS fragments m/z 99, m/z 283, and m/z 311.
In spectra numbers 2, 4, 5, 7, and 8, one or more of these fragments are present. This information was obtained from one survey experiment without the need for extra confirmatory MS/MS analyses. This allows the analyst to acquire important structural information in a single run.
The patient information leaflet for this tablet sample said it contained 10 rare animal and plant extracts. It also stated on the packaging and in the patient information that it was acceptable for persons with heart disease and hypertension to take it. However, use of synthetic PDE5 inhibitors is contraindicated when a patient is currently taking nitrate medications (i.e., nitroglycerin) as it can cause low blood pressure – there can be a synergistic effect of these drugs in relaxing vascular smooth muscle and drastically lowering blood pressure.18 Consequently, men in this group who suffer from ED may resort to seeking out this kind of herbal alternative – not knowing the remedy’s actual risk to their heath.
Sample 5 was analyzed using a more targeted approach. A precursor scan of m/z 99 was used to trigger a ScanWave DS MS/MS function. Sildenafil, tadalafil, and vardenafil were not found in this sample. A chromatographic peak with a RT of 8.38 min had precursor mass of m/z 489. The retention time of this component did not match that of vardenafil, m/z 489 at RT 8.92 min, or homosildenafil, m/z 489 at RT 9.5 min.
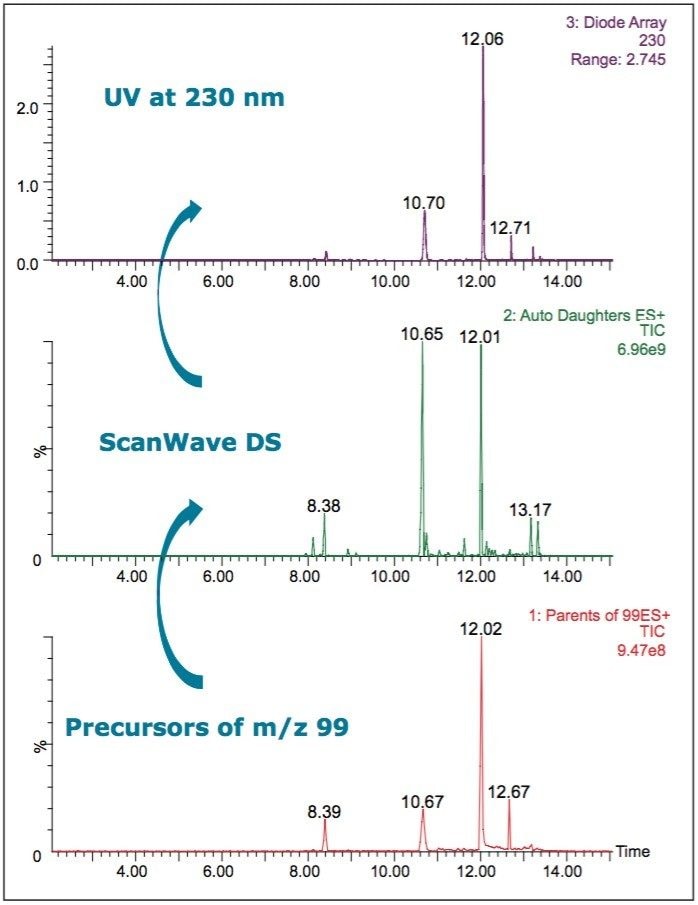
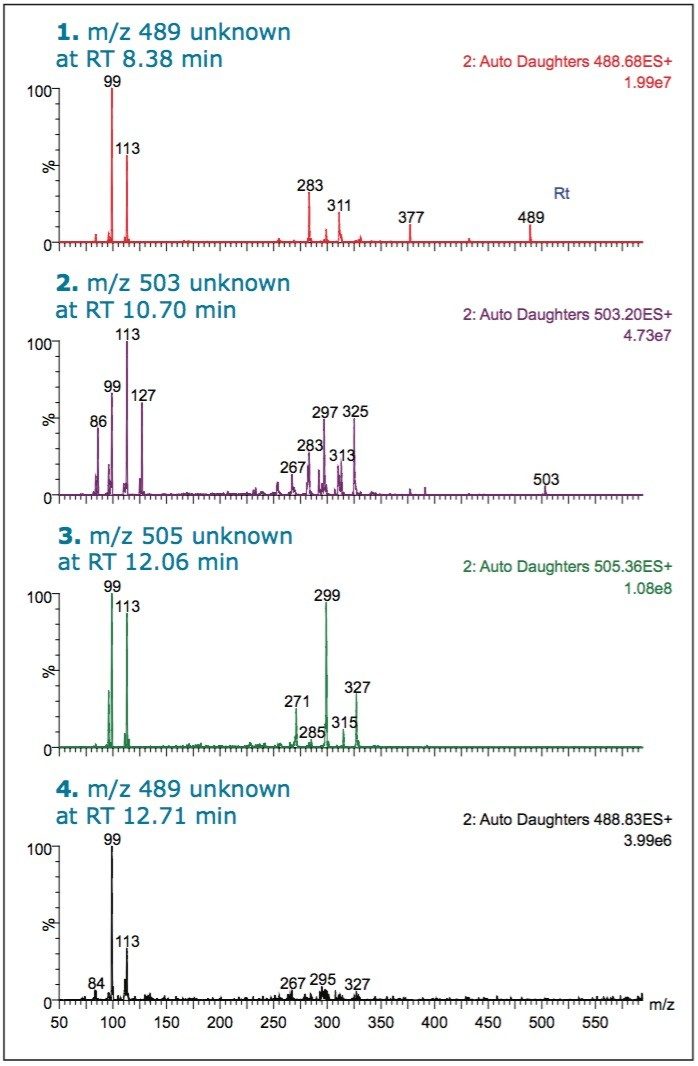
However, when Sample 5 was subjected to MS/MS fragmentation, the characteristic fragments of sildenafil were seen: m/z 99, m/z 283, and m/z 311. There was also another fragment observed, m/z 113. The presence of this ion in certain analogues has been reported previously in literature.13 The other chromatographic peaks, at retention times 10.70, 12.06, and 12.71 min, showed common fragments for the m/z 99 and m/z 113. These peaks are likely to arise from structural analogues of sildenafil.
This sample also declared on its packaging that it is all-natural, stating that it has helped support male performance for centuries. The supplement’s ingredients were supposed to contain wild yam extract, Siberian Ginseng extract, jujube extract, and cayenne extract, as well as others.
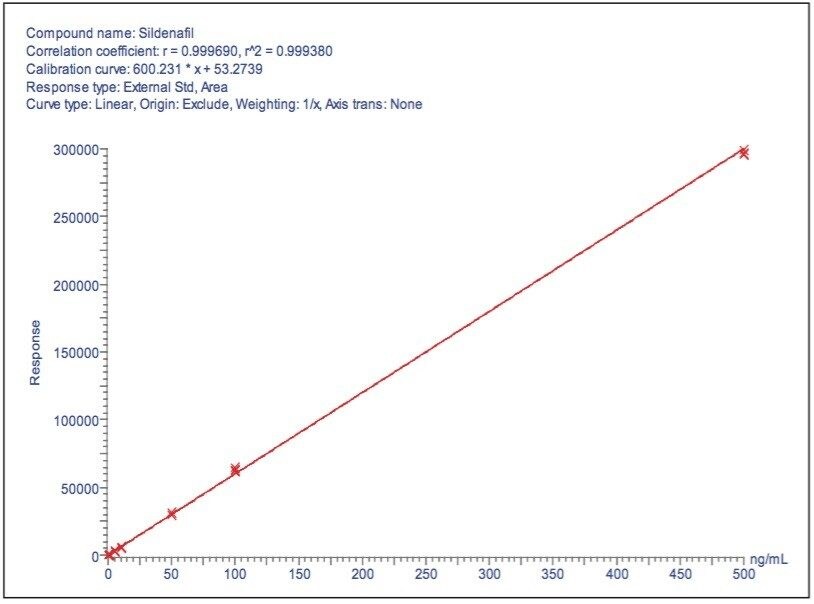
In an effort to understand the levels of adulteration to the samples, sildenafil and tadalafil were quantified. The drug substances for tadalafil and sildenafil citrate were purchased. Stock solutions of sildenafil and tadalafil were prepared at a concentration of 1.0 mg/mL in methanol. Working standard solutions were prepared at concentrations of 0.1 ng/mL to 500 ng/mL in a simulated matrix. This was performed using extraction of a mixture of some of the ingredients listed on the patient information of the HDS samples and subsequently spiking the resulting solution with the appropriate levels. Quantitation curves were injected in triplicate and were linear over the calibration range for both tadalafil and sildenafil (Figures 12 and 13). QC samples were within acceptable limits (< 15%).
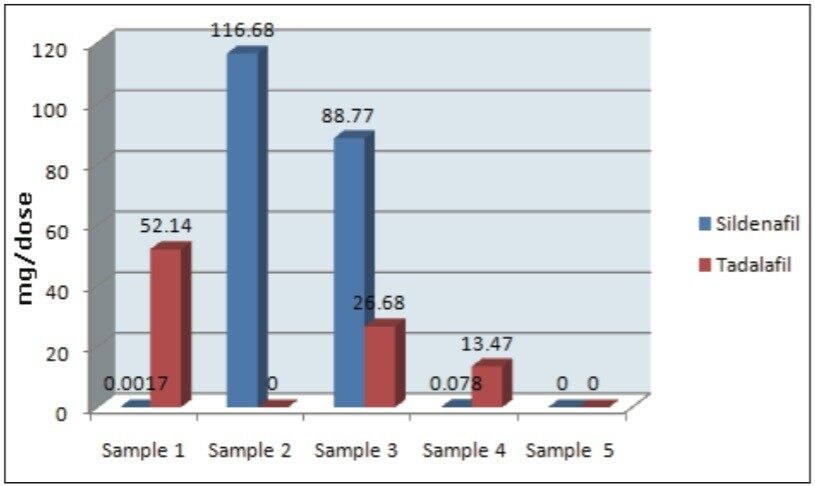
The range of sildenafil measurements quantified in the HDS samples that were purchased via the Internet was 0.078 to 116.68 mg/dose. The range of tadalafil measurements was 13.47 to 52.14 mg/dose.
For samples 1 through 4, these concentrations would be at therapeutic levels.
It should be noted that the analytical method used for quantitation of the samples had not been validated at the time when these results were reported.
Adulteration of herbal dietary supplements with synthetic pharmaceuticals is a growing problem. There have been reports in published literature that show many of the supposed natural alternatives to approved synthetic PDE5 inhibitors used to treat ED are actually adulterated with them. Many reports also indicate that this adulteration of herbal additives is a growing trend. Due to the threat to public health posed by unknowingly ingesting both the known PDE5 inhibitors and their analogues, it is vital that analysts have tools that allow them to characterize these complex samples adequately.
The Waters Xevo TQ Mass Spectrometer, with its unique collision cell design, facilitates the simultaneous acquisition of MS and MS/MS data in one LC-MS run. Its high scan speed of up to 10,000 amu/sec allows for these experiments to be performed with sufficient points across the peak to accurately define the narrow peaks produced by UPLC. This capability facilitates data-dependant experiments where real-time switching between MS and MS/MS allows more information to be acquired from a single injection. This reduces the need for separate experiments and accelerates the process of structural identification and unknown compound determination.
The value of the using the Waters Xevo TQ MS has been demonstrated for the analysis of adulterated herbal dietary supplements. Furthermore, use of the Xevo TQ MS for quantitation of the samples revealed that the doses of the sildenafil and tadalafil are sufficiently high to be therapeutic.
720003159, June 2015