For research use only. Not for use in diagnostic procedures.
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the use of the ACQUITY UPLC Online SPE Manager (OSM) enables analytically sensitive measurement of important vitamin D metabolites in plasma.
Improved workflows for the measurement of vitamin D metabolites in plasma using the ACQUITY UPLC (OSM).
Recent studies have shown that vitamin D deficiency is common in adults and children in many parts of the world. In addition to the well known effects of vitamin D deficiency such as calcium mal-absorption, there is growing evidence that vitamin D deficiency may increase the risk of certain types of cancer, and play a role in many other diseases. As a consequence, the measurement of vitamin D metabolites is of increasing importance to many researchers.
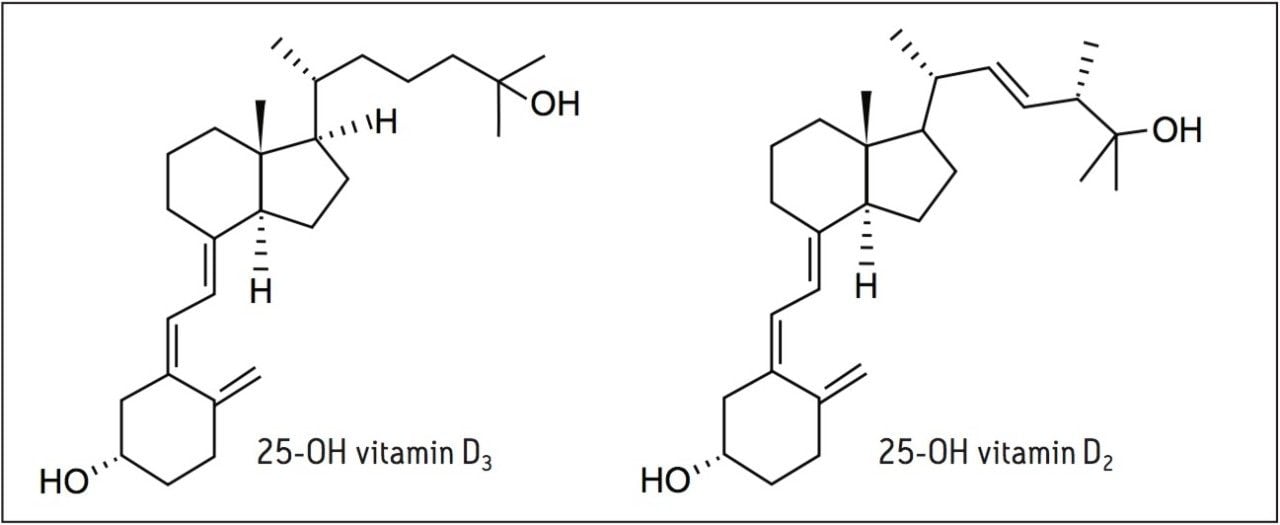
Vitamin D exists in two forms; vitamin D3 (D3), which is produced in the skin on exposure to sunlight, and vitamin D2 (D2), which is a plant derivative that is found in many dietary supplementation products. All forms of vitamin D are metabolized in the liver to form 25-hydroxyvitamin D (25-OH D), which is further metabolized in the kidneys to form the active metabolite 1,25-dihydroxyvitamin D that is responsible for calcium homeostasis.
In many laboratories, the measurement of vitamin D and its metabolites in plasma by LC-MS has gained popularity over other methods such as the competitive binding methods, immunoassays, and HPLC to quantify 25-OH D2 and 25-OH D3. The issues with these other types of measurement are that they may be complex and costly to perform, or they cannot discriminate between 25-OH D2 and 25-OH D3 and measure these vitamers accurately enough to provide the level of information that researchers are seeking. Even when using LC-MS, issues with preparing samples effectively and efficiently must be overcome, to allow for the development of an accurate method that can be used routinely for measuring vitamin D metabolites.
|
LC-MS system configuration: |
|
|---|---|
|
LC: |
ACQUITY UPLC |
|
MS: |
Xevo TQ-S |
|
Column: |
ACQUITY UPLC CSH Phenyl-Hexyl, 1.7 μm, 2.1 x 100 mm (p/n 186005407) |
|
Sample preparation: |
ACQUITY UPLC OSM |
|
SPE cartridge: |
MassTrak C8 Online SPE Cartridges, 10 μm, 1 x 10 mm (p/n 186005673IVD) |
|
Sample pre-treatment: |
150 μL plasma 25 μL internal standard in methanol containing (isotopically labelled 25-OH D2 and D3) 800-μL of aqueous 0.4 M ZnSO4; centrifuge to pellet protein |
|
Solvent A: |
10% methanol + 2 mM ammonium acetate + 0.05% formic acid |
|
Solvent B: |
Methanol + 2 mM ammonium acetate + 0.05% formic acid |
|
Flow rate: |
0.4 mL/min |
|
Column temp.: |
35 ˚C |
|
Time |
%A |
%B |
|---|---|---|
|
0.00 |
35 |
65 |
|
3.75 |
10 |
90 |
|
4.25 |
0 |
100 |
|
5.25 |
0 |
100 |
|
5.50 |
35 |
65 |
|
6.25 |
35 |
65 |
|
Conditioning: |
Step 1: 0.5 mL acetonitrile at 4 mL/min Step 2: 0.5 mL acetonitrile/methanol/ isopropyl alcohol/water + 0.2% formic at 3 mL/min |
|
Equilibration: |
0.5 mL water at 4 mL/min |
|
Sample load: |
0.5 mL water at 2 mL/min |
|
Cartridge wash: |
Step 1: 0.5 mL 20% methanol + 0.1% ammonia at 2 mL/min Step 2: 0.5 mL 30% methanol + 0.1% ammonia at 2 mL/min Step 3: 1 mL 55% methanol + 0.1% formic acid at 2 mL/min |
|
Clamp flush: |
Step 1: 0.5 mL acetonitrile at 3 mL/min Step 2: 1.0 mL acetonitrile/methanol/ isopropyl alcohol/water + 0.2% formic at 3 mL/min Step 3: 0.5 mL water at 3 mL/min |
|
Elution time: |
0.75 min |
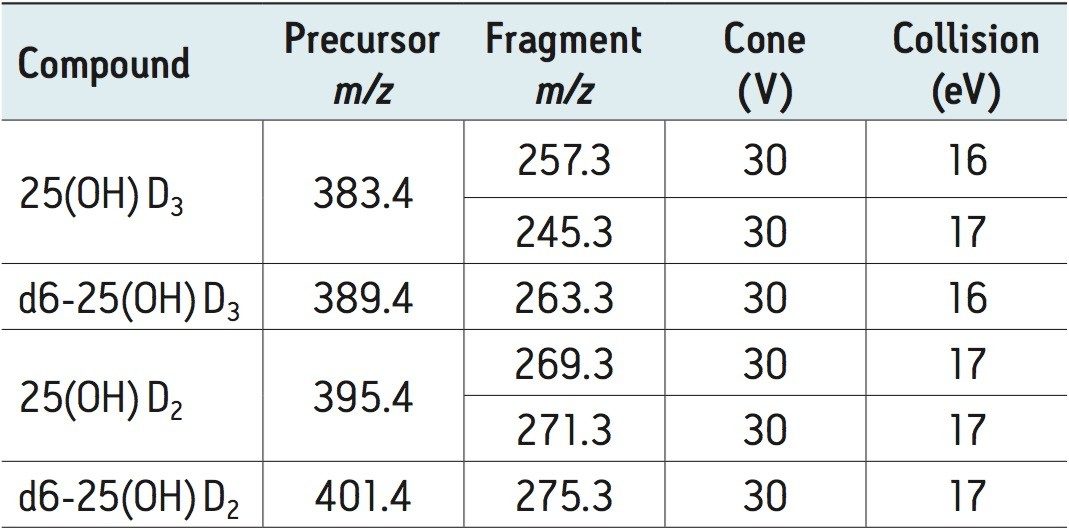
A clinical research method for the measurement of two vitamin D metabolites (25-OH D2 and 25-OH D3) from plasma has been developed. This method takes advantage of the unique capabilities of the ACQUITY UPLC OSM to enable analytically sensitive and reproducible measurements. The method enables direct measurement of these key vitamin D metabolites, and provides valuable information to clinical researchers as they investigate the role these molecules play in normal biological function and disease. Simultaneous measurement of these key metabolites with this method can also provide valuable information to clinical researchers as they investigate novel models of disease.
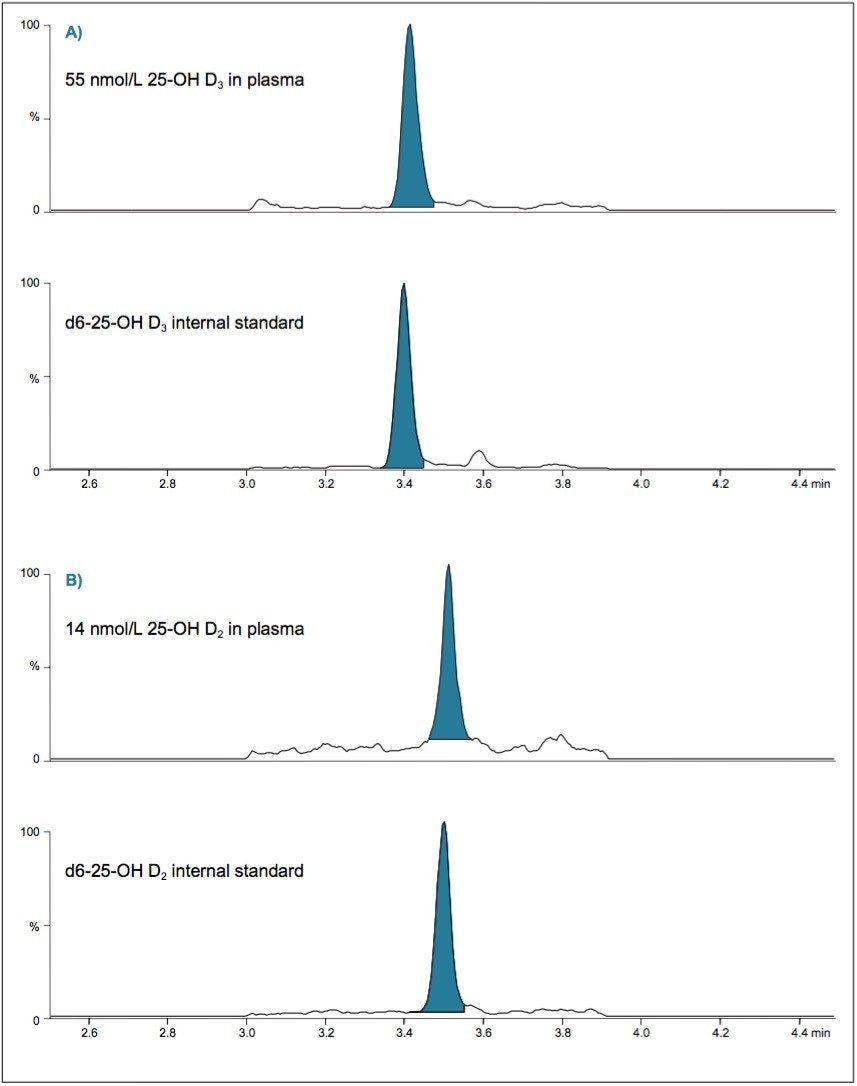
The method described was successfully used to detect and measure two vitamin D metabolites in plasma. Figure 2 demonstrates the typical analysis of these analytes in plasma samples.
Intra-assay variability
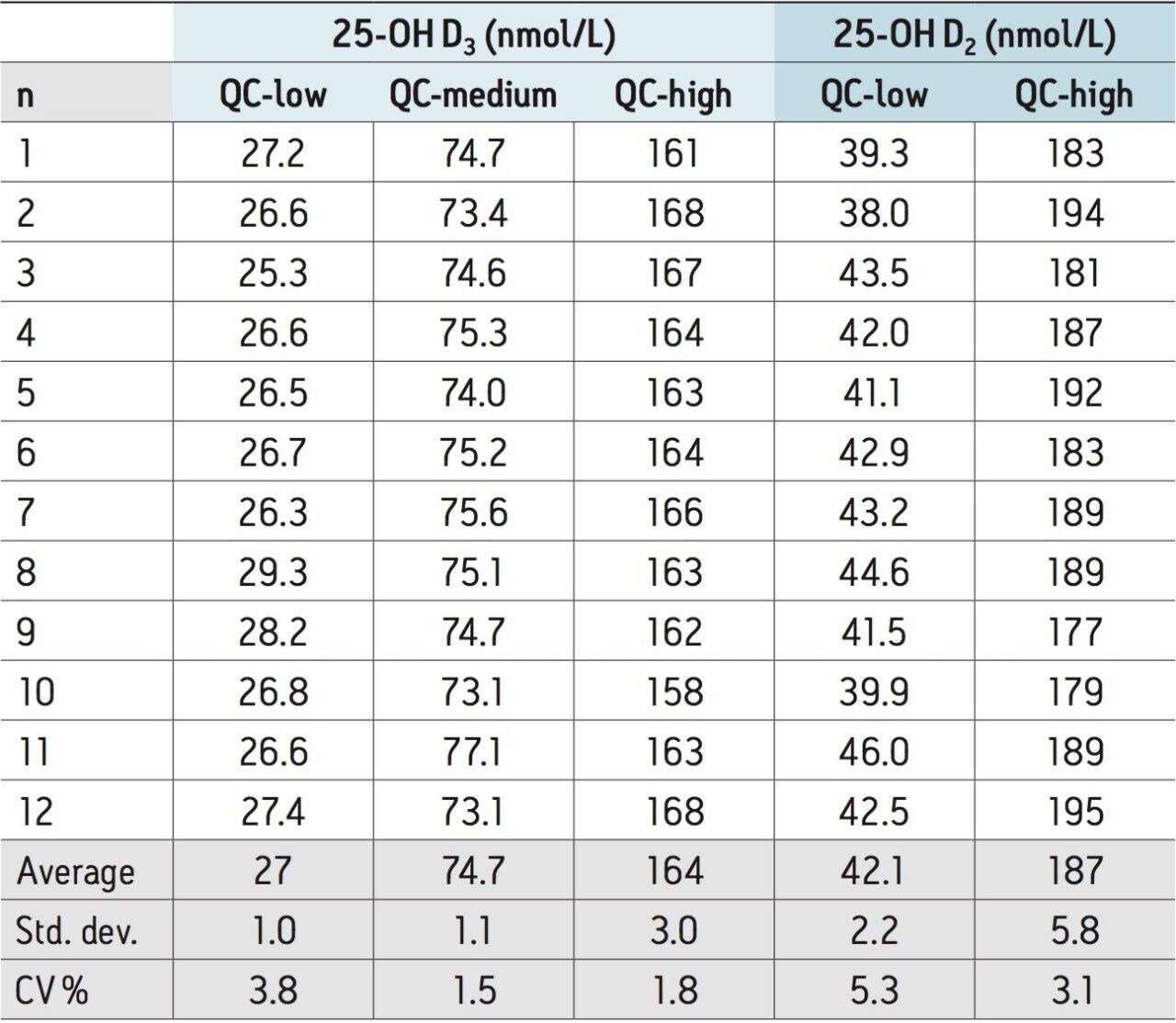
The reproducibility of the method for measuring the vitamin D metabolites was assessed for both intra- and inter-assay variability. For measuring intra-assay variability, 12 samples at three concentrations of 25-OH D3 (27, 75, and 165 nmol/L) and two concentrations of 25-OH D2 (40 and 185 nmol/L) in plasma were analyzed. The results presented in Table 1 indicate that the intra-assay variability of the method ranges from 1.5 to 3.8% for 25-OH D3 and from 3.1 to 5.3% for 25-OH D2.
Inter-assay variability
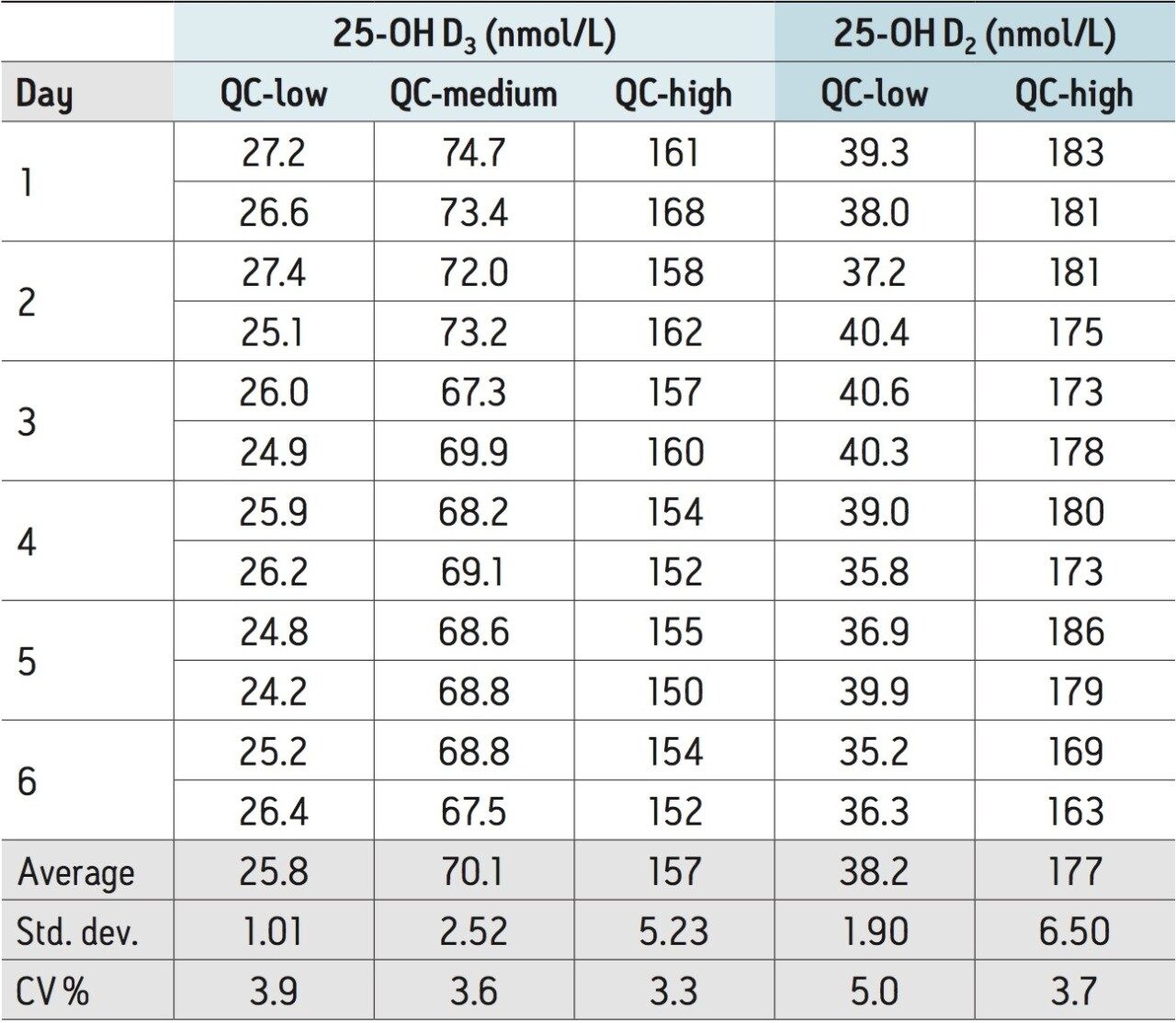
Inter-assay variability of the method was assessed by performing the method daily over the course of six days using duplicate samples of the vitamin D metabolites in plasma at concentrations ranging from 25 to 180 nmol/L. The results presented in Table 2 indicate that the inter-assay variability range from CVs of 3.3 to 3.9% for 25-OH D3 to CVs of 3.7 to 5% for 25-OH D2.
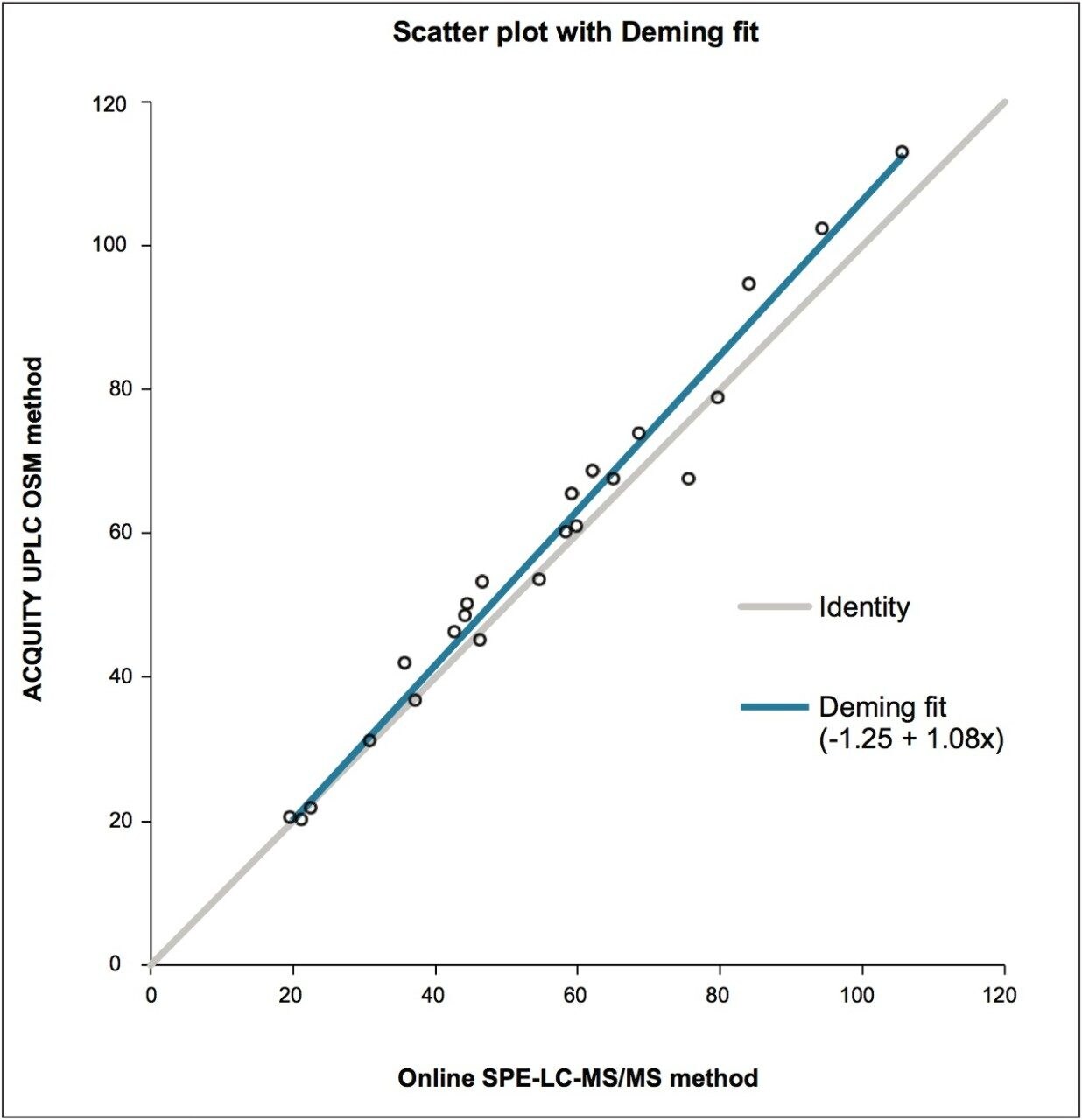
The method described was compared to results obtained from an independent LC-MS/MS method, developed in the same laboratory. This method utilizes a different, less efficient workflow involving the use of an older online SPE system in conjunction with protein precipitation. The results indicate that the developed method displays excellent correlation with the existing LC-MS method.
In many clinical research laboratories, the sensitive and reproducible measurement of vitamin D metabolites can be a challenge. Older methods involving the use of immunoassay or other technologies are unable to provide the level of information on these important vitamin D metabolites that many researchers are seeking to measure. While LC-MS is capable of making the specific and analytically sensitive measurement of these vitamin D metabolites, preparing samples adequately and efficiently has always been an issue that has not been easy to overcome.
The method described here takes best advantage of the automated sample preparation capabilities of the ACQUITY UPLC OSM in the analysis of vitamin D metabolites. This highly automated method will reduce both the direct staff and instrument time required to perform these analyses when compared to other LLE or offline analysis techniques, and allow for rapid, simultaneous analysis of these vitamin D metabolites. The C8 sorbent used for extraction and the ACQUITY UPLC CSH Hexyl-Phenyl Column used in this study also showed excellent performance in terms of sample clean-up and separation of these molecules (Figure 2).
Furthermore, both the intra- and inter-assay variability of the method were assessed to be excellent (Tables 1 and 2). Even at low nmol/L concentrations of either vitamin D metabolite, the calculated CVs were typically less than 5%. The robustness of the method makes it a good option for routine measurement of vitamin D metabolites in a complex matrix such as plasma.
In this study, a rapid clinical research method using the ACQUITY UPLC OSM for the measurement of vitamin D metabolites has been developed. The method is suitable for routine use and the small volume of serum used (150 μL) is desirable for clinical research laboratories that are sample limited.
The method developed here provides:
720005345, May 2015