
This is an Application Brief and does not contain a detailed Experimental section.
This application brief provides general guidelines regarding the choice of sample injection solvents based on the analyte and stationary phase properties when performing separations on an ACQUITY UPC2 System.
To optimize UPC2 separations, choosing the right injection solvent can tremendously impact peak shape.
Diluent effects are extremely important to consider when developing a UPC2 method. First and foremost, the sample must be soluble in a solvent compatible with the mobile phase. In general, if a sample can be dissolved in methanol (with up to a few percent of water) or any other organic solvent, it can be successfully injected into supercritical CO2, notwithstanding any adverse effects to the column. Sample solvent effects of some common diluents were previously investigated on 2-ethylpyridine bonded phases.1 We found that nonpolar sample solvents such as heptane and THF should be used to minimize peak distortion and it is always advantageous to use low dispersion instruments, even when ideal sample solvents are not used. In this work, we examined the former point for ACQUITY UPC2 BEH and HSS C18 SB columns.
Two very different stationary phases were selected for comparison. A polar stationary phase, ACQUITY UPC2 BEH Column, consisting only of polar surface groups (silanols), and what can be considered a mixed polarity stationary phase, ACQUITY UPC2 HSS C18 SB Column, consisting of nonpolar, trifunctional C18, ligands, and silanols. Three sample diluents were chosen for each to demonstrate proper selection of an injection solvent based on the stationary phase and analyte. Two analytes, butylparaben (polar) and d-tocopherol (nonpolar), were chosen. Both are easily soluble in the three sample solvents chosen for each column at sufficient concentrations for this study (0.4 g/L). The two analytes were dissolved individually in each of the sample solvents and injected at volumes between 1.0 and 9.0 μL. Notice the evolution of the peak profile of butylparaben injected onto the ACQUITY UPC2 BEH Column shown in Figure 1. The flow rate was set to 1.2 mL/min of 95:5 CO2/MeOH at 40 °C and 2175 psi (ABPR). The peak height of the heptane-dissolved sample increases linearly to the point of detector saturation. Injections of d-tocopherol above 2.0 μL begin to show peak distortion, but heptane is clearly the best choice of the solvents tested. For the ACQUITY UPC2 HSS C18 SB Column, heptane also gives the best peak shape for butylparaben (Figure 2). However, peak shape for d-tocopherol is best with acetonitrile as the injection solvent instead of heptane, which was the best injection solvent for this compound on the BEH column.
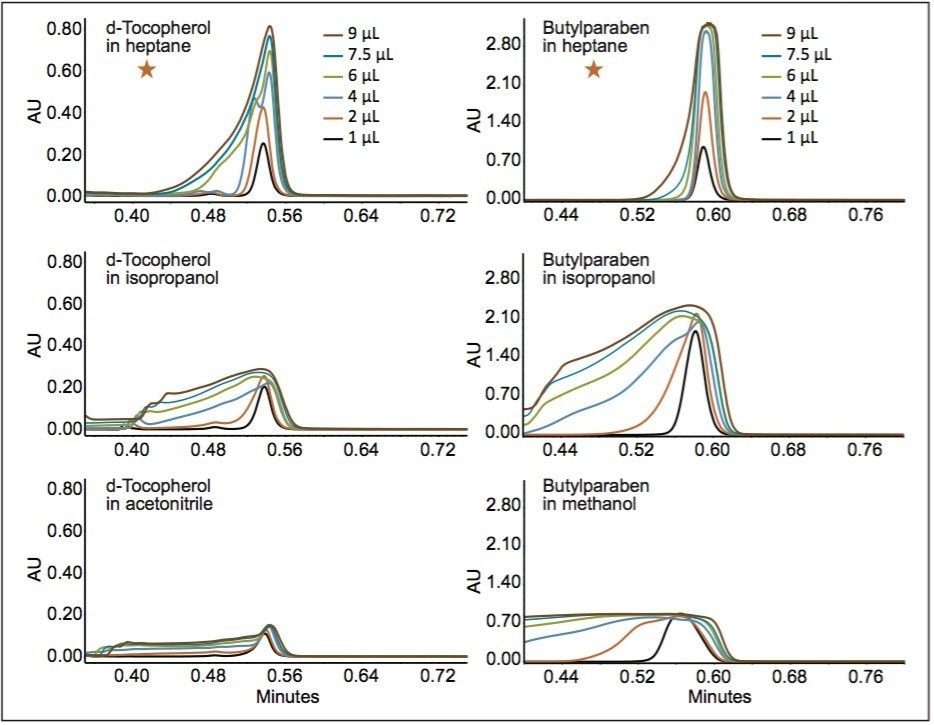
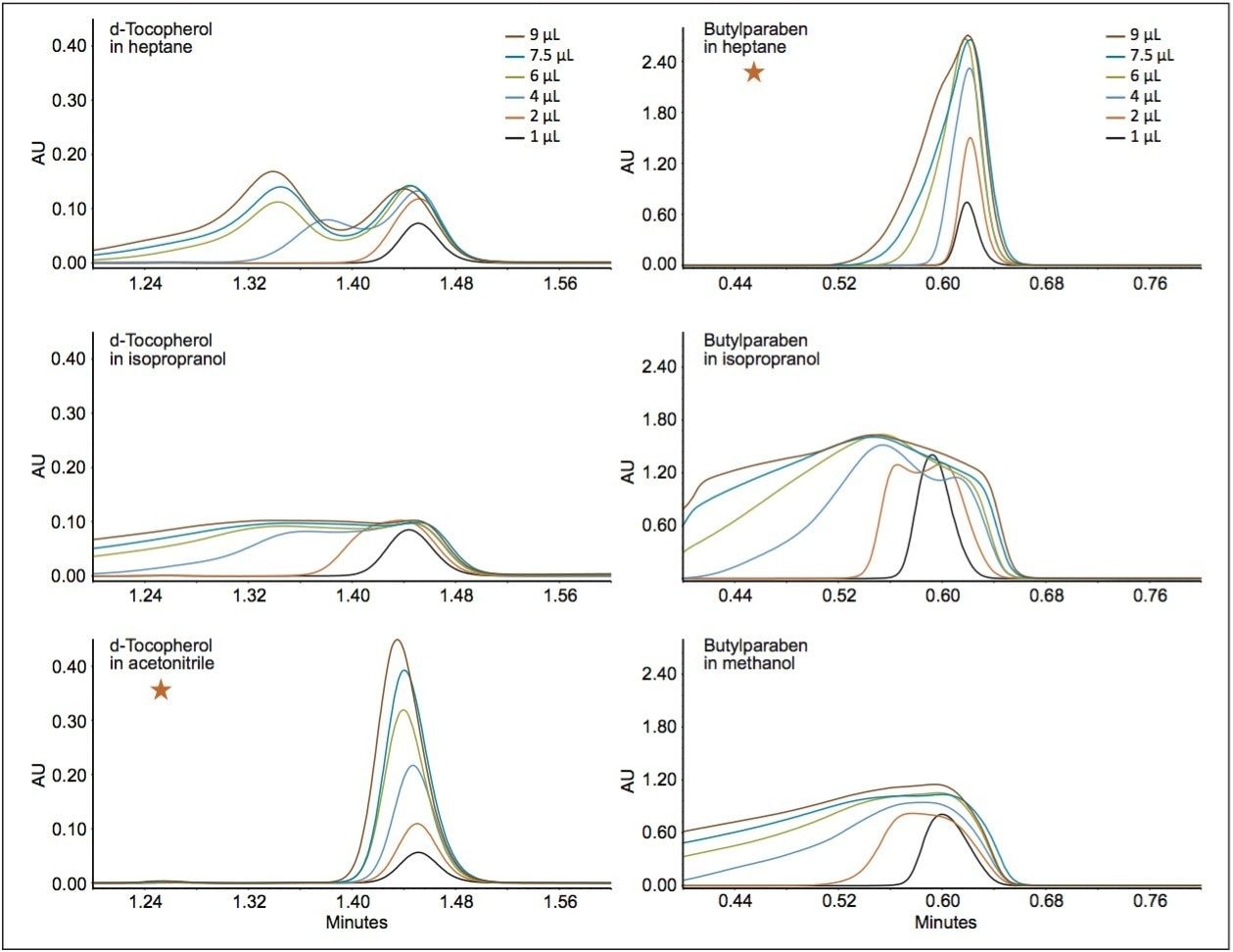
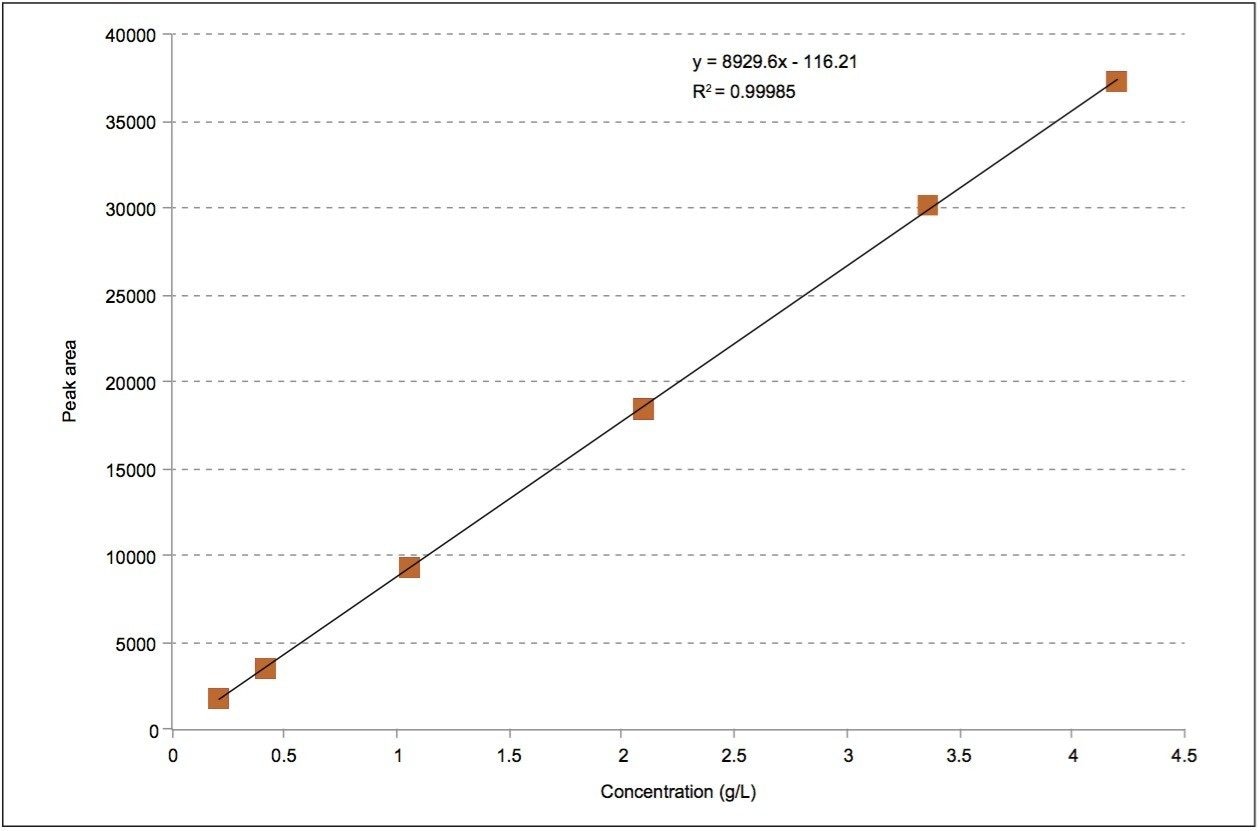
From the injections of butylparaben and d-tocopherol, it can be concluded that polar compounds are separated by the polar substituents of the stationary phases. These data suggest that polar compounds should be dissolved/ diluted in nonpolar solvents for UPC2 separations. For nonpolar compounds (i.e., d-tocopherol), separated on a non-polar stationary phase, a polar injection solvent (i.e., acetonitrile or methanol) should be used. We considered the maximum solubility of d-tocopherol and found it to be ~4.2 g/L in pure acetonitrile with copious sonication. Figure 3 demonstrates a linear response for peak area to d-tocopherol (in acetonitrile) concentration obtained using a 2-μL injection volume. Using solvents of opposite polarity from the solute can be difficult or impractical. When a specific analyte cannot be dissolved in detectable quantities in a very weak solvent, an effective option is to dissolve an analyte in relatively high concentrations in a stronger solvent, such as methanol or isopropanol, then diluting with a weaker solvent, e.g., heptane in Figure 1 or acetonitrile for d-tocopherol in Figure 2.
Choosing the best solvent for a particular sample can be challenging and often results in a pragmatic choice. However, better chromatographic results can be obtained when properly chosen solvents are used. Making a poor selection of sample solvent can lead to very distorted peak shapes and under-informed decisions concerning the chromatographic results. Two basic guidelines are useful for UPC2 separations: 1) Polar analytes should be dissolved in nonpolar solvents when possible, independent of the stationary phase. 2) Based on the stationary phase, nonpolar analytes should be dissolved in solvents of the opposite polarity. Nonpolar solvents should be used for polar stationary phases and polar solvents for nonpolar (or mixed) stationary phases.
720004981, March 2014