This application note demonstrates a rapid and simple analysis of BVO in soft drinks and beverages using UPC2/MS. Convergence Chromatography (CC) coupled with mass spectrometry enables a rapid, simple, and direct analysis of BVO in beverages.
Brominated vegetable oil (BVO) is often used as a weighting agent, or as a solubility-transmitter for citrus oils and other lipophilic compounds1 in soft drinks and beverages. Since lipophilic compounds are insoluble in water and their densities are lower than 1 g/cm3, they would gradually separate from the aqueous phase. BVO has a much higher density (1.33 g/cm3). By blending BVO with the lipophilic ingredients, the density can be adjusted close enough to 1 g/cm3, and a stable emulsion can be formed. The U.S. FDA has established a BVO limit at 15 ppm in finished beverages, while many countries in Europe, Asia, South America, and Australia, have banned its use in beverages.
BVO is generated by adding bromine to the double bonds of unsaturated triacylglycerides (TAGs) in vegetable oil (VO). The BVO’s chemical structure or even composition is not clearly defined due to the non-uniform composition of the starting material, VO. Figure 1 shows the structure of a BVO compound. Analysis of BVO is rarely reported. Gas chromatography with mass spectrometry (GC-MS) has been proposed recently for the analysis of BVO in soft drinks and cocktail syrups.2,3 This GC-MS method requires tedious derivatization (or saponification) of BVO, and has a long run time (about 50 min).
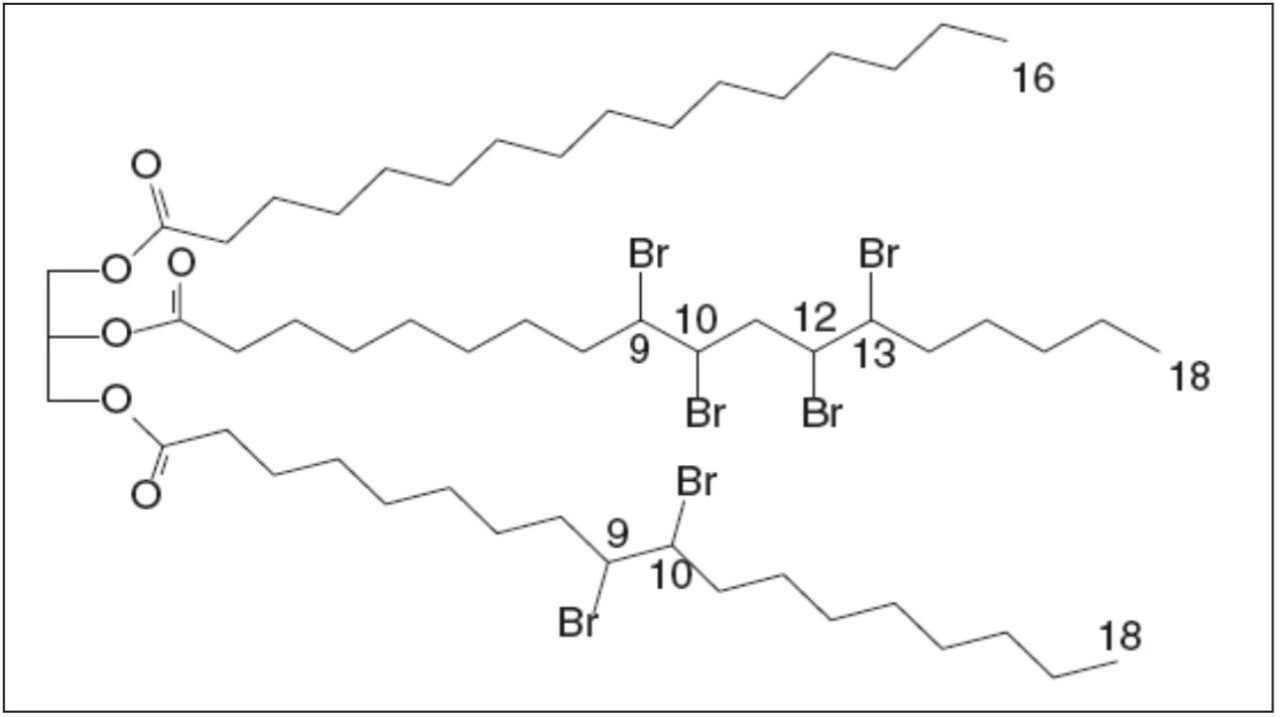
Waters UltraPerformance Convergence Chromatography (UPC2) is a state-of-the-art supercritical fluid chromatography (SFC) technology that provides exceptional efficiency and speed of separation.4 UPC2 has been applied to a wide range of compounds, including VO, and has shown great benefits in selectivity, throughput, and ease-of-use.5 This application note demonstrates a rapid and simple analysis of BVO in soft drinks and beverages using UPC2/MS. BVO was extracted and analyzed directly without any derivatization. The chromatography total run time was 9 min. The analytical method performance (limit of quantitation or LOQ, repeatability, linearity, and recovery), as well as the analysis of BVO in soft drinks and beverages are presented.
10.00 grams of decarbonated soft drinks and beverages that were purchased from a local grocery store were mixed with 10.0 mL (or 6.59 grams) hexanes, vortexed for 30 seconds, and centrifuged at 4000 rpm for 1 min. The upper layer (hexanes) was collected and weighed. The extract was dried under gentle nitrogen stream, and reconstituted with 1000 μL chloroform. A common BVO sample was obtained from a collaborator.
|
UPC2 conditions |
|
|---|---|
|
System: |
ACQUITY UPC2 |
|
Column: |
ACQUITY UPC2 BEH, 3 x 100 mm, 1.7 μm |
|
Column temp.: |
60 °C |
|
Injection volume: |
2 μL |
|
Flow rate: |
1.500 mL/min |
|
Mobile phase A: |
Compressed CO2 |
|
Mobile phase B: |
Isopropanol (IPA) |
|
Gradient: |
1% IPA for 2 min, linear ramp to 70% IPA in 3 min, and hold at 70% IPA for 1 min before returning to the initial condition. Equilibrate for 3 min. |
|
MS System: |
Xevo TQ-S |
|
Ionization mode: |
ES- |
|
Capillary voltage: |
3.00 kV |
|
Cone voltage: |
20.00 V |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
500 °C |
|
Cone gas flow: |
150 L/hr |
|
Desolvation gas flow: |
1000 L/hr |
|
Collision gas flow: |
0.18 mL/min |
|
Nebulizer gas flow: |
7.00 Bar |
|
Software: |
MassLynx |
The main challenge in the analysis of BVO is that it is comprised of a mixture of TAGs that differ in fatty acids chain length, degree of unsaturation (number of double bonds), degree of bromination, and structural and spatial arrangement (isomerism). The different types of BVO compounds could number in the dozens or hundreds. In addition to that, the composition of BVO can differ from batch to batch due to variance in the VO source and the bromination process.
In this study, we employed UPC2 with tandem mass spectrometry to analyze BVO. UPC2 has been applied to the analysis of TAGs, and has shown exceptional efficiency and speed of separation.6,7 Multiple reaction monitoring (MRM) transitions from molecular ions to bromide ions (Br-) can selectively detect brominated TAGs, and avoid potential interference from VO or other non-brominated compounds.
Due to the complex nature of BVO, it is challenging to detect all BVO components in any given sample. So, this study was designed to monitor a small, but representative, number of BVO compounds to quantify. Figure 2 shows the mass spectra of BVO and VO obtained by combining mass spectra across their chromatographic peaks. These were obtained on a UPC2 BEH 1.7 μm, 3.0 x 100 mm Column under the gradient conditions described in the experimental section. 12 molecular ions across the mass range of BVO were selected for quantitation, shown in Figure 2. These ions were confirmed by the parent ion scan of the bromide ion (data not shown), and were absent in the VO mass spectrum. The purpose of selecting multiple ions across a wide range of m/z is to ensure a true representation of the BVO profile, and eliminate possible skewing of results due to batch to batch variation in the VO TAG composition and the bromination process. The extracted ion chromatogram (XIC) generated from these 12 MRM transitions was used for quantitation. Table 1 shows the MRM transition and the data acquisition conditions used in the study. No sample derivatization is needed in this approach.
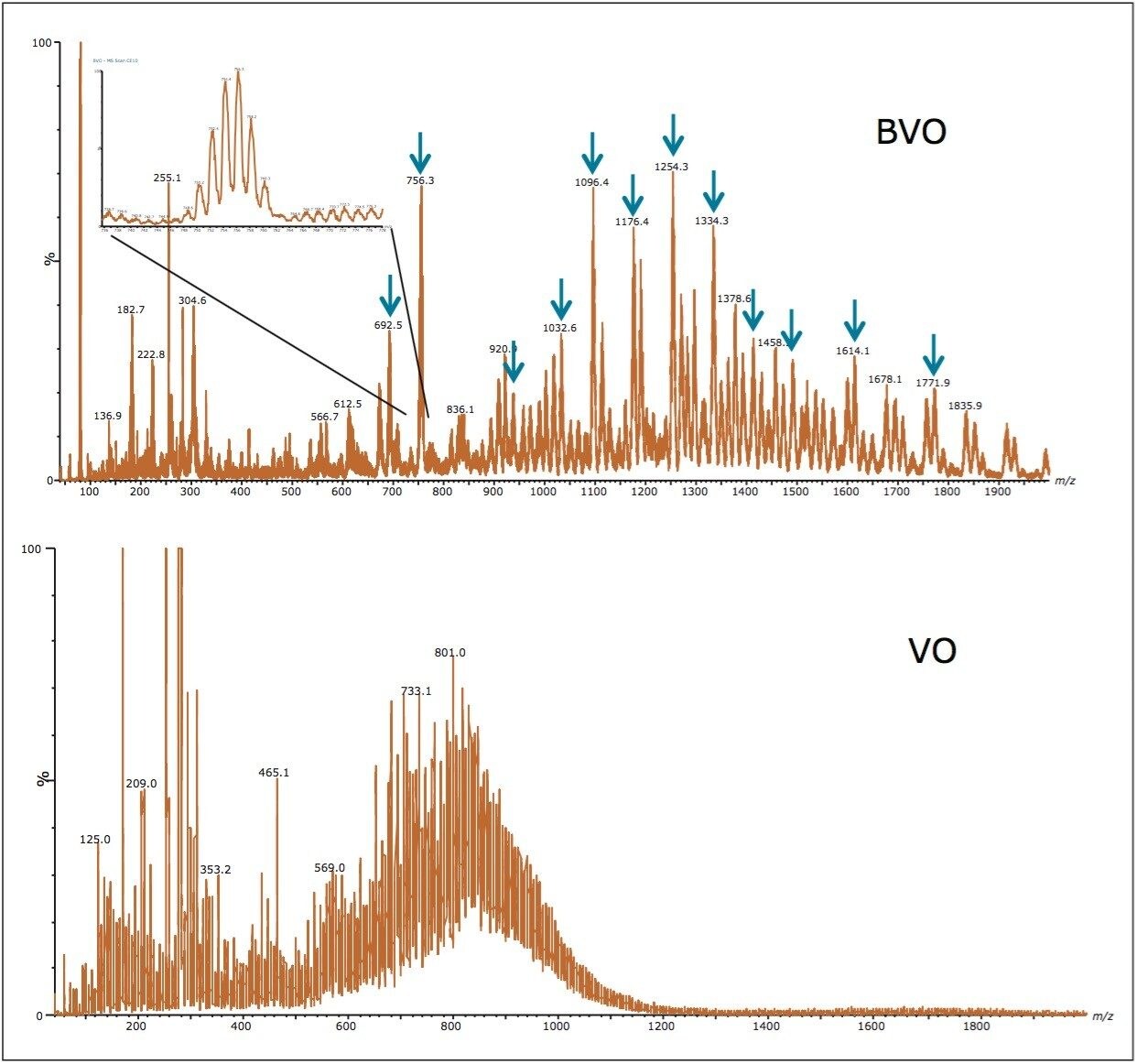
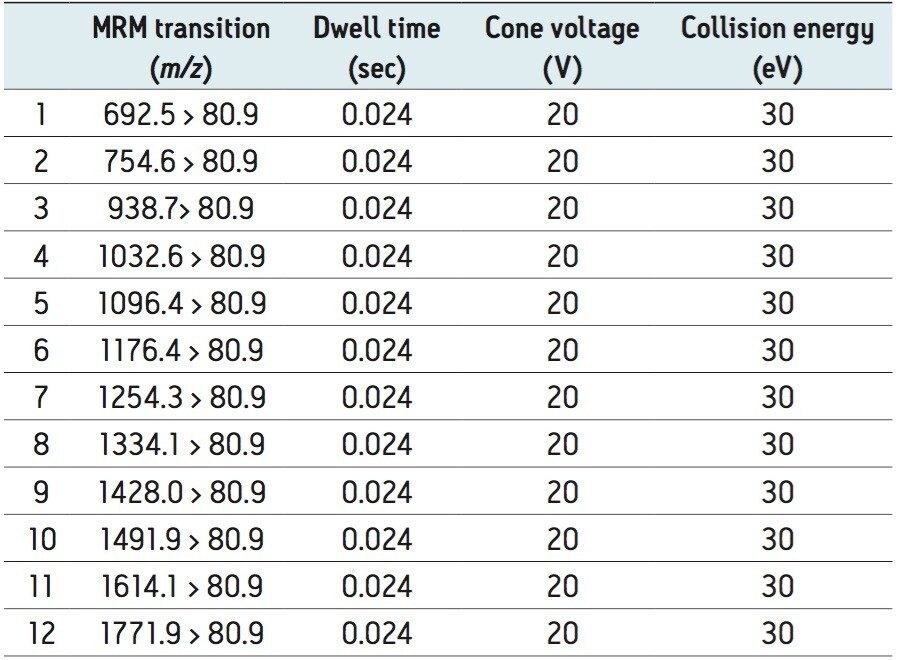
Figure 3 shows an XIC of a BVO solvent standard, and the established calibration curve from concentration 10 to 500 μg/mL. Table 2 shows the corresponding calibration results. A calibration correlation coefficient (R2) of 0.998 was obtained. The LOQ was estimated at 5.7 μg/mL, which corresponds to 0.6 ppm in finished beverages. Repeatability (RSDs in peak area) was <5% at three levels of standard concentrations, shown in Table 3.
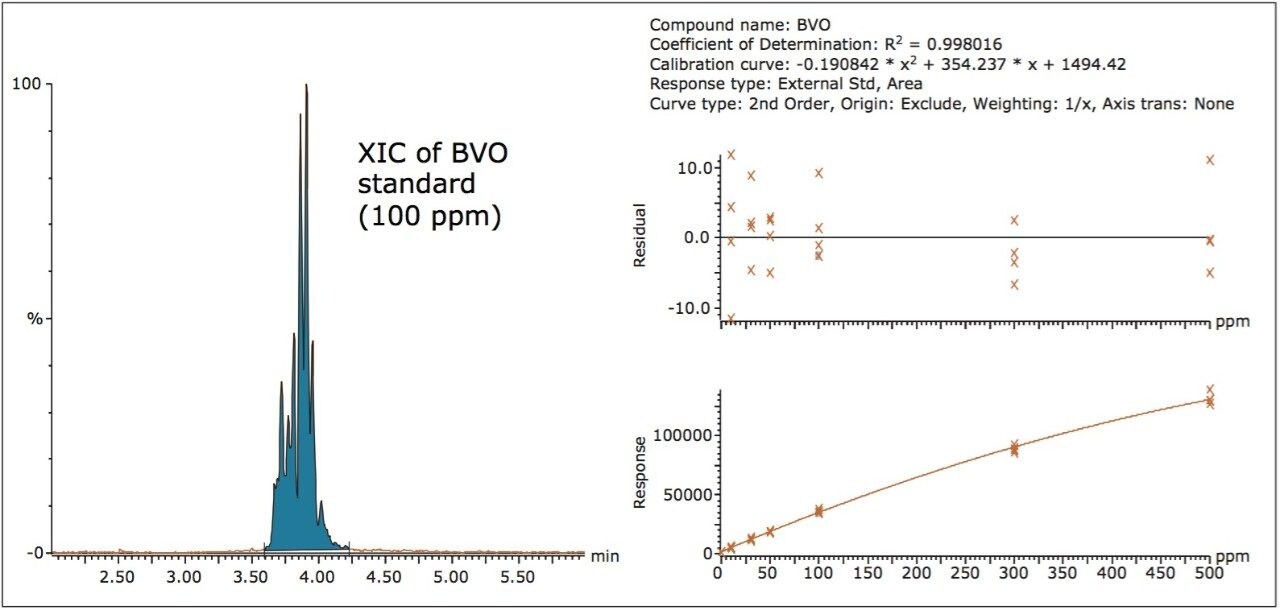
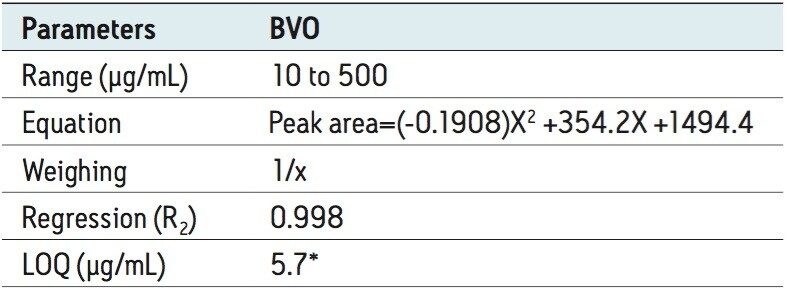
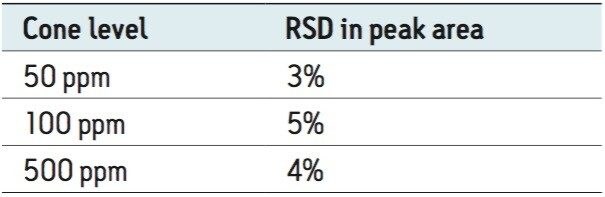
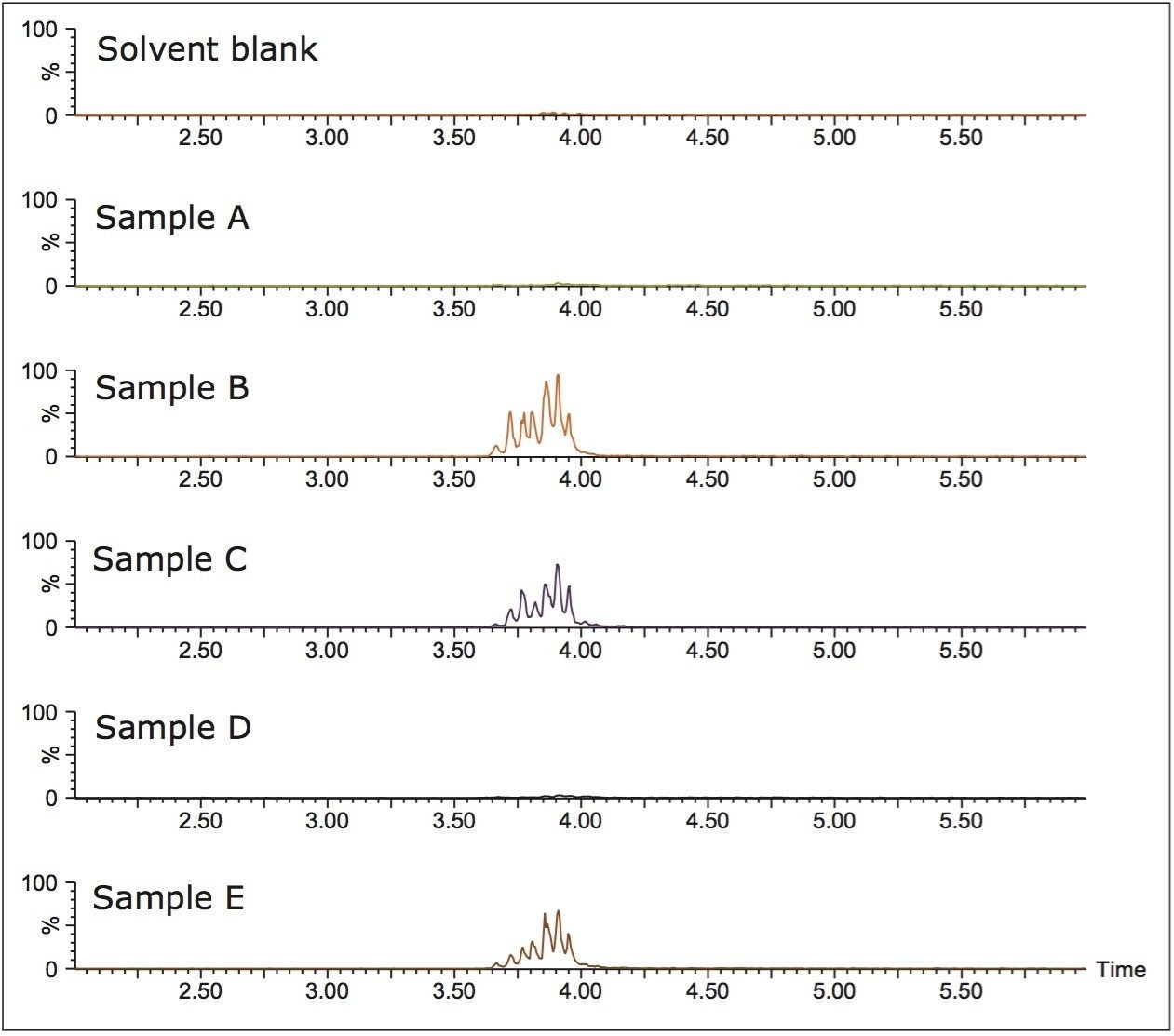
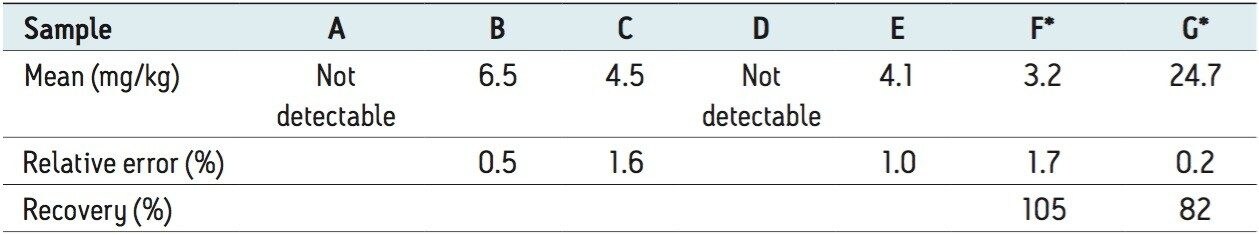
Figure 4 shows the XIC of a blank solvent sample and five soft drink samples. The soft drinks were products from major brands. Samples B, C, and E have BVO in their label claims, whereas samples A and D do not. Table 4 shows the analysis results for these five soft drinks and the recovery study results on spiked soft drink samples. The calibration curve from the solvent standards was used in the analysis and the recovery study. No BVO was detected in samples A and D, which is in agreement with their label claims. Samples B, C, and E contained 4.1 to 6.5 ppm BVO, which is within the U.S. FDA limit for finished beverages. The relative error in the duplicated measurement was <1.6% for the three samples that contained BVO. Sample D was used as a blank sample, into which BVO was spiked at 3 ppm and 30 ppm for recovery study. Recoveries of 105% and 82% were obtained at 3 ppm and 30 ppm, respectively.
Convergence Chromatography (CC) coupled with mass spectrometry enables a rapid, simple, and direct analysis of BVO in beverages. Unlike the GC-MS method, there is no derivatization in this UPC2/MS method. After a simple liquid-liquid extract, BVO can be analyzed directly by UPC2/MS. The chromatographic run time is 9 min, which is at least five times faster than the GC-MS method. The UPC2/MS method shows good accuracy and precision in the analysis of BVOs. It can potentially be used in food testing labs for routine BVO determination. For countries where BVO is banned for food additives, this method could be used for screening of BVO existence in beverages.
720005079, June 2014