
Fast separation of hydrophobic vitamins and carotenes results in significantly improved throughput and cost savings, allowing laboratories to meet the increasing demand for regulatory compliance monitoring where a high number of analyses are required.
Fat-soluble vitamins (FSVs) include vitamins A, D, E, K and carotenoids such as β-carotene. The FSVs are involved in complex metabolic reactions related to important biological functions such as vision (vitamin A), calcium absorption (vitamin D), antioxidant protection of cell membrane (vitamin E), and blood coagulation (vitamin K).1 Beta-carotene is a precursor to vitamin A and has 100% vitamin A activity in the human body. Lycopene is not an essential nutrient for humans; however, its potential antioxidation benefit resulted in increased marketing in some dietary supplements along with other ingredients. The chemical structures of some FSVs are shown in Figure 1.
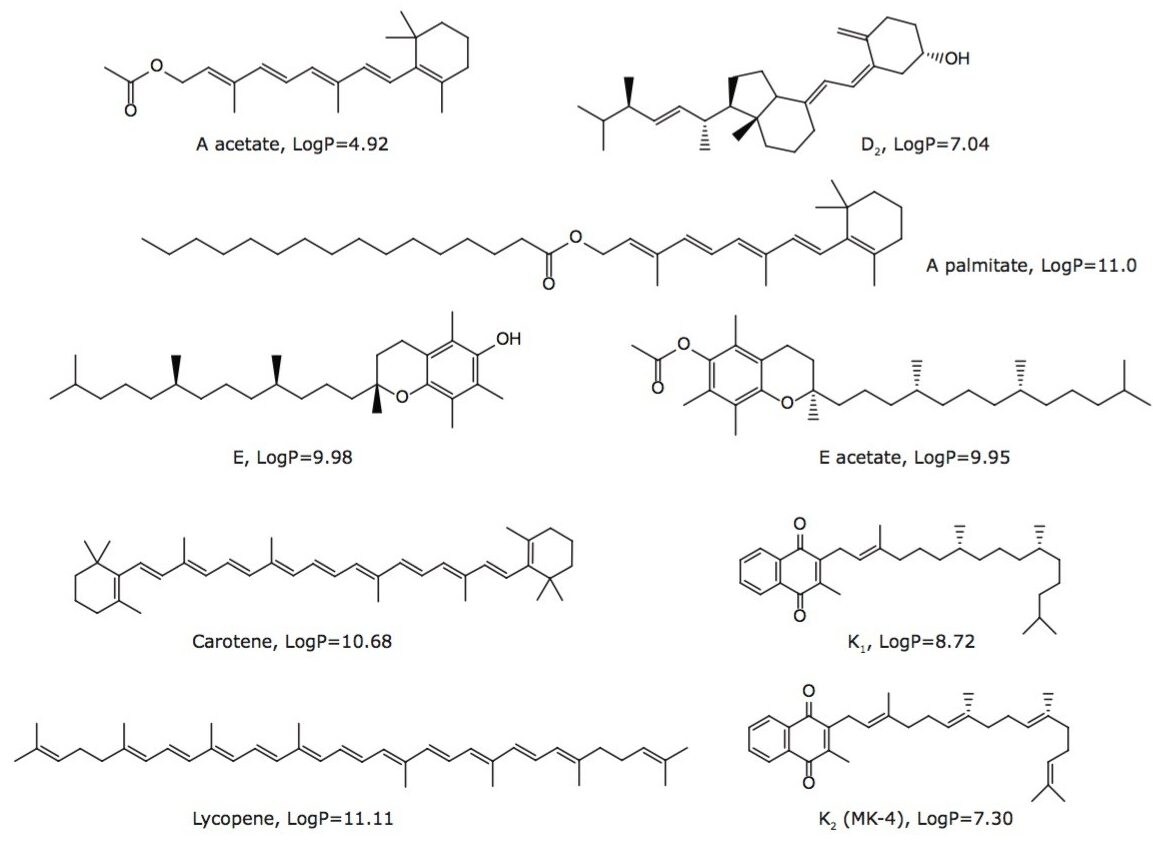
Vitamins and supplements represent a multi-billion-dollar market. It is anticipated that this market will continue to grow over the next five to ten years.2 The growth is primarily driven by worldwide consumers’ pursuit of a better and healthier lifestyle and their increasing awareness of health and food habits. However, there are also growing concerns about the safe consumption of FSVs from supplements and fortified foods, particularly for vitamins A and D, where overdosing could impose serious health risks.3 As more stringent legislation is being introduced or contemplated in many countries to reinforce regulatory compliance of micronutrients in fortified foods and dietary supplements, there is an increasing demand for developing fast, reliable, and fit-for-purpose analytical methods to quantify FSVs in various matrices for quality control, labeling, and regulatory compliance.
Currently, most FSV separations are carried out using liquid chromatography (LC), both reversed phase (RP) and normal phase (NP).1, 3-5 While there are ample AOAC methods for the analysis and quantification of individual FSVs in food and supplement matrices, there is a general lack of methods for simultaneous analysis of FSVs in vitamin pre-mixes, as well as analyzing carotenoids.3
Due to its speed, cost-effectiveness, and sustainable nature, UltraPerformance Convergence Chromatography™ (UPC2) offers a viable alternative by producing fast analysis of formulations that contain many FSVs.6 In this application, we present a single-injection method for the simultaneous separation of nine FSVs using UPC2 in four minutes. The optimized method also shows good reproducibility in both retention time and peak area, and can potentially be adopted for high-throughput quantitative analysis.
Seven fat-soluble vitamins: retinyl acetate (vitamin A acetate), retinyl palmitate (vitamin A palmitate), alpha-tocopherol (vitamin E), alpha-tocopheryl acetate (vitamin E acetate), ergocalciferol (vitamin D2), vitamin K1, vitamin K2 (MK-4); and two carotenes: lycopene and β-carotene were purchased from Sigma Aldrich and used as received. All samples were dissolved in methyl tert-butyl ether (MTBE) at approximately 0.1 mg/mL and transferred to an amber vial prior to analyses.
|
Time (min) |
B% |
|---|---|
|
0.0 |
2.0 |
|
2.0 |
2.0 |
|
2.5 |
20.0 |
|
3.5 |
20.0 |
|
3.75 |
2.0 |
|
4.0 |
2.0 |
There are many publications describing the separation of FSVs and carotenoids, using both NPLC and RPLC.1, 3-5 Since FSV analysis often requires the use of low polarity organic solvents to dissolve the samples, NPLC offers the advantage of compatibility with organic solvents, allowing for direct injection of FSVs or FSV extracts without the need for an evaporation step. RPLC, however, is often superior to NPLC in chromatographic efficiency,7 but can be problematic for FSV analysis for the following reasons: 1) low solubility of analytes in mobile phase, and 2) strong retention for FSVs that results in a lengthy run time. To that end, semi-aqueous mobile phases (mixtures of methanol or acetonitrile and water), or non-aqueous mobile phases are used; the latter is also referred to as non-aqueous reversed phase (NARP) LC.7-8 UPC2, though widely practiced as a normal phase separation technique, is well-suited for the separations of low polarity analytes. The main mobile phase for UPC2, CO2, is not only a good solvent for low polarity analytes (similar polarity to hexane), but is compatible with the organic solvents used to dissolve those analytes during sample preparation, such as MTBE in this study. In addition, the low polarity of CO2 promotes non-polar interactions between analytes and the mobile phase, resulting in shorter retention and faster run time.
A total of six columns, ACQUITY UPLC HSS T3, ACQUITY UPLC HSS C18, ACQUITY UPC2 HSS SB, ACQUITY UPC2 CSH Fluoro-Phenyl, ACQUITY UPC2 2-Ethyl Pyridine, and ACQUITY UPC2 BEH; and a total of four solvents including: isopropanol (IPA), acetonitrile (ACN), ethanol, and an equal mixture of IPA/ACN, were initially screened for method development. The combination of the ACQUITY UPLC HSS C18 Column and ACN was the only one that provided the separation between the critical pair vitamins K1 and K2. A relatively low column temperature of 30 °C was chosen for two reasons; first, to improve selectivity between K1 and K2, and secondly to minimize possible on-column degradation of the carotenes due to their high susceptibility to oxidation.
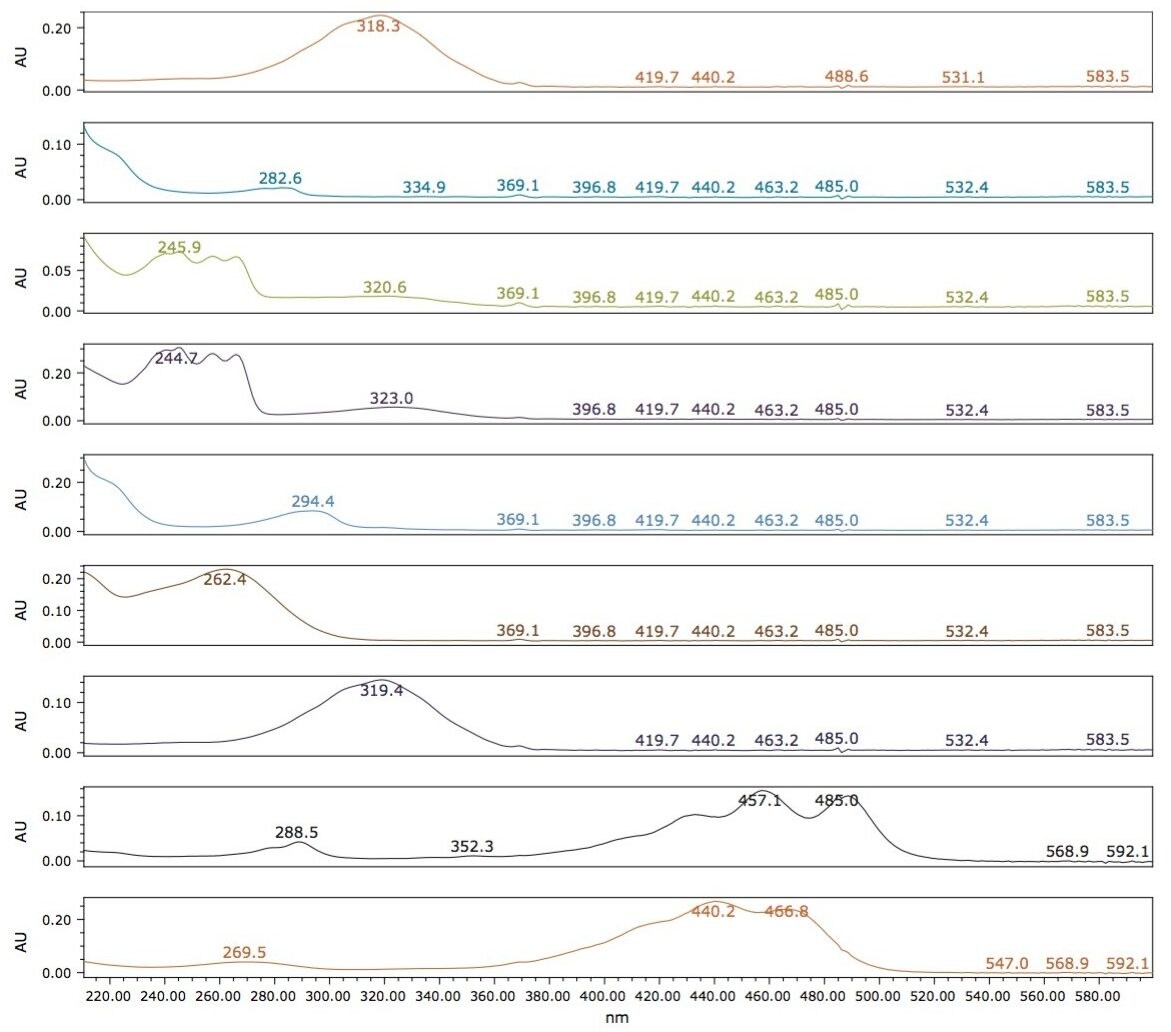
The resulting chromatogram and UV spectra for each component after method optimization are shown in Figures 2 and 3, respectively. Overall, the elution order of the nine FSVs loosely tracks their LogP values. All seven vitamins eluted off with a low percentage of acetonitrile (2%) in the mobile phase, while the two carotenes required a much higher percentage of acetonitrile (20%) for elution from the column. This observation can be rationalized by the analyte structures and their propensity to interact with the mobile/stationary phase. While non-polar interactions between CO2 and the lipophilic moiety of the FSVs applies for all nine analytes, the oxygen atoms in the seven vitamins, in the form of carbonyl or hydroxyl groups, induce additional polar interactions between the analytes and acetonitrile in the mobile phase, thus reducing the retention time. The two carotenes, on the other hand, do not contain any hetero-atoms in the molecules and are more likely to form strong interactions with the non-polar octadecyl carbon chains on the stationary phase, resulting in longer retention time.
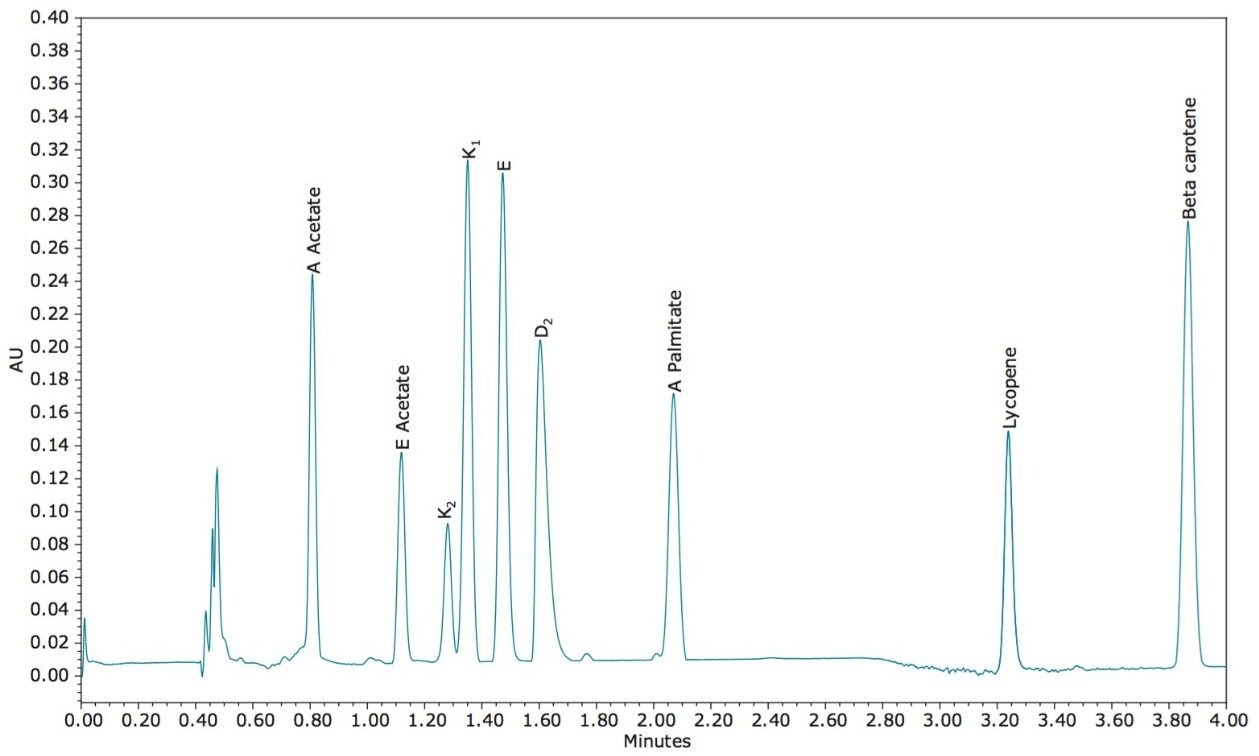
In order to test the reproducibility of the method injection-to-injection, six replicate injections for the nine FSVs were performed, as shown in Figure 4. The reproducibility statistics are summarized in Table 1. The relative standard deviations (RSDs) for retention time are less than 0.25%. For peak area, seven out of nine FSVs have RSDs of less than 1%. For the first peak (vitamin A acetate), the slightly elevated RSD is likely due to the rising baseline associated with the injection sequence. Nevertheless, the reproducibility demonstrated here should satisfy regulatory compliance monitoring where general tolerance for overall process reproducibility is typically around ±20%.3
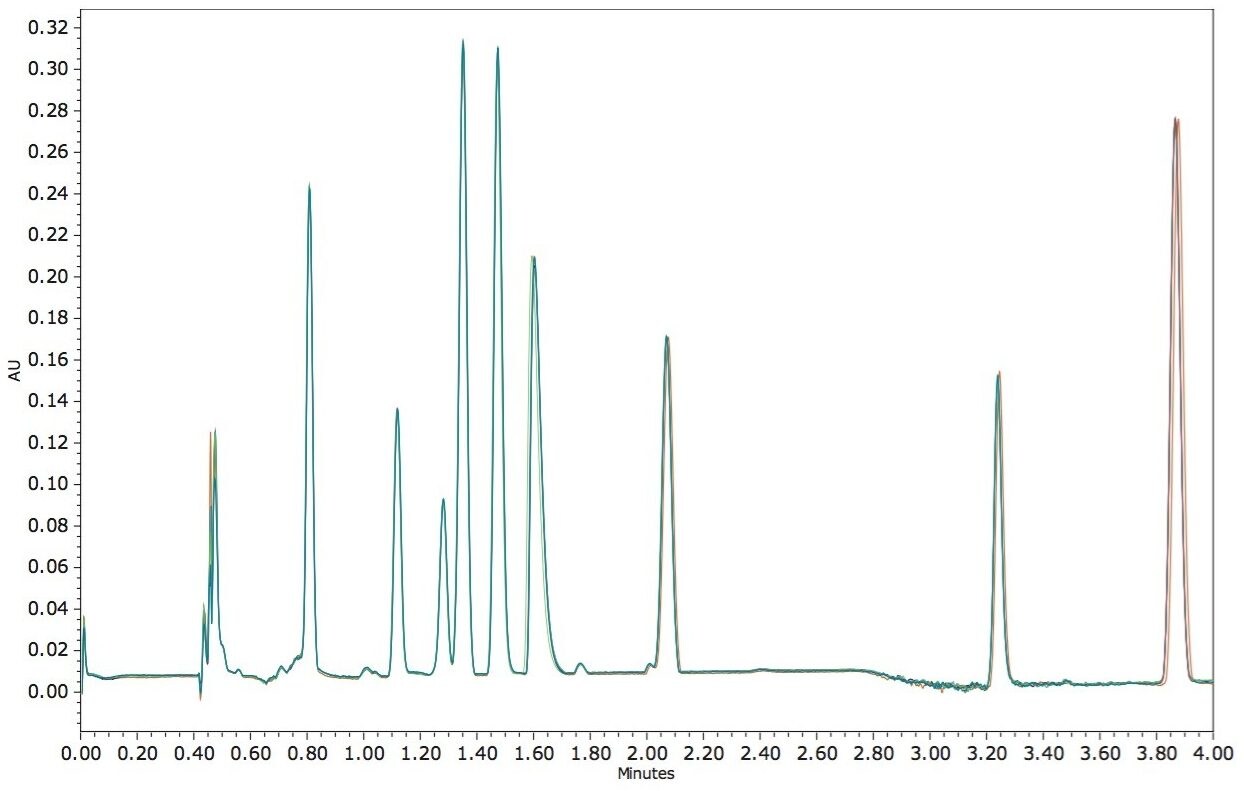

In this application, we have demonstrated the simultaneous separation of nine FSVs in 4 minutes using a single-injection UPC2 method. Based on six replicate injections, the RSDs for retention time of all nine peaks were less than 0.25% for retention time, and less than 3% for peak area (<1% in most cases). The UPC2 methodology eliminates the need for multiple methods often required for analyzing such mixtures using LC, greatly reducing the number of assays for laboratories routinely performing multiple analyses of FSVs. The UPC2 method is 4 to10 times faster than traditional methods of analysis. The high speed of the UPC2 analysis can be routinely used by food/supplement industries for regulatory compliance monitoring, requiring a large number of analyses.
720004551, January 2013