For research use only. Not for use in diagnostic procedures.
This application note demonstrates the benefits SYNAPT G2-Si System for the quantification of proteins and peptides in a new High Definition MRM (HD-MRM) mode.
***nanoACQUITY UPLC applications readily transfer to the ACQUITY UPLC M-Class System***
Recently, targeted LC-MS-based methods have been applied to quantitative proteomics, facilitating highly accurate and multiplexed relative and absolute quantification studies. Both triple quadrupole and high-resolution mass spectrometers have been used. In both instances, the protein is not quantified directly but one or more signature peptides that are released in stoichiometric amounts during enzymatic digestion are monitored as surrogates.
To achieve absolute quantification, typically, an accurately determined amount of a standard stable isotope-labeled peptide is added to the sample. The key benefits of triple quadrupole-based SRM/MRM assays are sensitivity, acquisition speed, and linear dynamic range.
However, the specificity of SRM/MRM peptide quantification has been questioned,1,2 prompting the use of high-resolution accurate mass instruments for peptide quantification. Here, the benefits of the SYNAPT G2-Si System are demonstrated for the quantification of proteins and peptides in a new High Definition MRM (HD-MRM) mode, where T-Wave ion mobility separations are used with MS/MS mode on a quadrupole time-of-flight mass spectrometer, to provide a highly sensitive and selective quantitative assay with maximum response for all product ions of a molecular peptide ion in a single acquisition.
The cytosolic content of E.coli, bovine serum albumin (BSA), and alcohol dehydrogenase (ADH) were digested using trypsin. The digest mixtures/lysates were injected onto a nanoACQUITY UPLC System equipped with an ACQUITY UPLC BEH 1.7 μm, 15 cm x 75 μm column coupled to a SYNAPT G2-Si Mass Spectrometer. The BSA on-column amounts ranged from one attomole to one femtomole on-column. Data were processed with TargetLynx and/or Skyline.3
The High Definition Multiple Reaction Monitoring (HD-MRM) acquisition method of the SYNAPT G2-Si High Definition MS System provides sensitivity or selectivity benefits for targeted quantification. In this study, we describe the sensitivity and coverage benefits provided by the use of a Wideband Enhancement acquisition mode on SYNAPT G2-Si. Wideband Enhancement uses ion mobility separations to provide maximum duty cycle for all product ions in a single MS/MS acquisition.
Maximum transmission, as shown in Figure 1, is achieved by the synchronization of the pusher region of the time-of-flight analyzer with target m/z ranges as ion mobility separated ions are released from the Triwave device. The signal intensity of product ions can be increased from five- to ten-fold. Hence, this provides very low limits of detection and quantitation for all potential product ions from a target peptide molecular ion without loss in duty cycle.
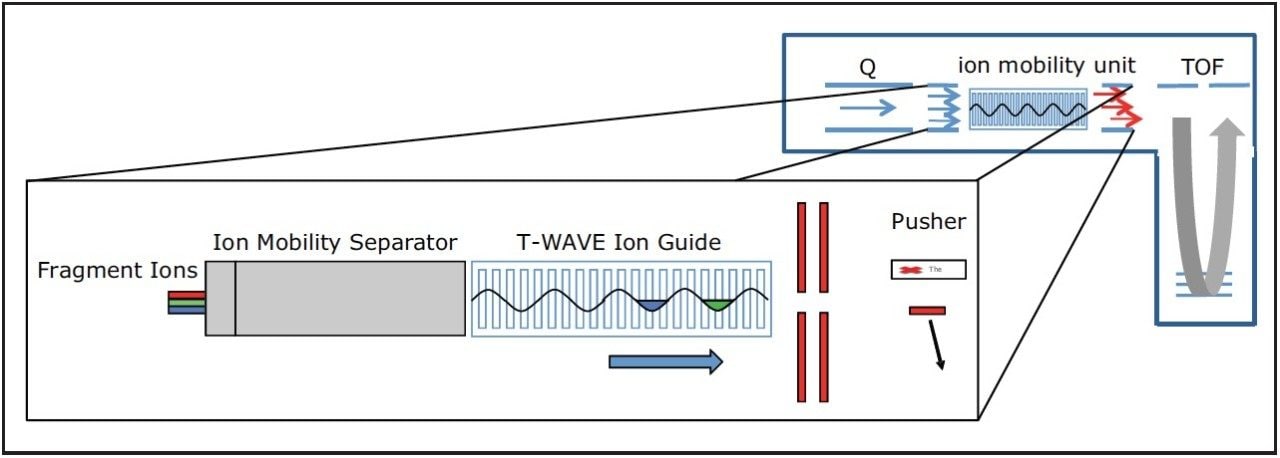
Acquisitions can be conducted in either scheduled1 or non-scheduled mode, with the former providing the ability to measure the transitions of several hundred components during the course of a single LC-MS experiment. Collision energies can be in silico predicted or derived from previous discovery experiments. Data can be processed and quantified with either TargetLynx or Skyline.
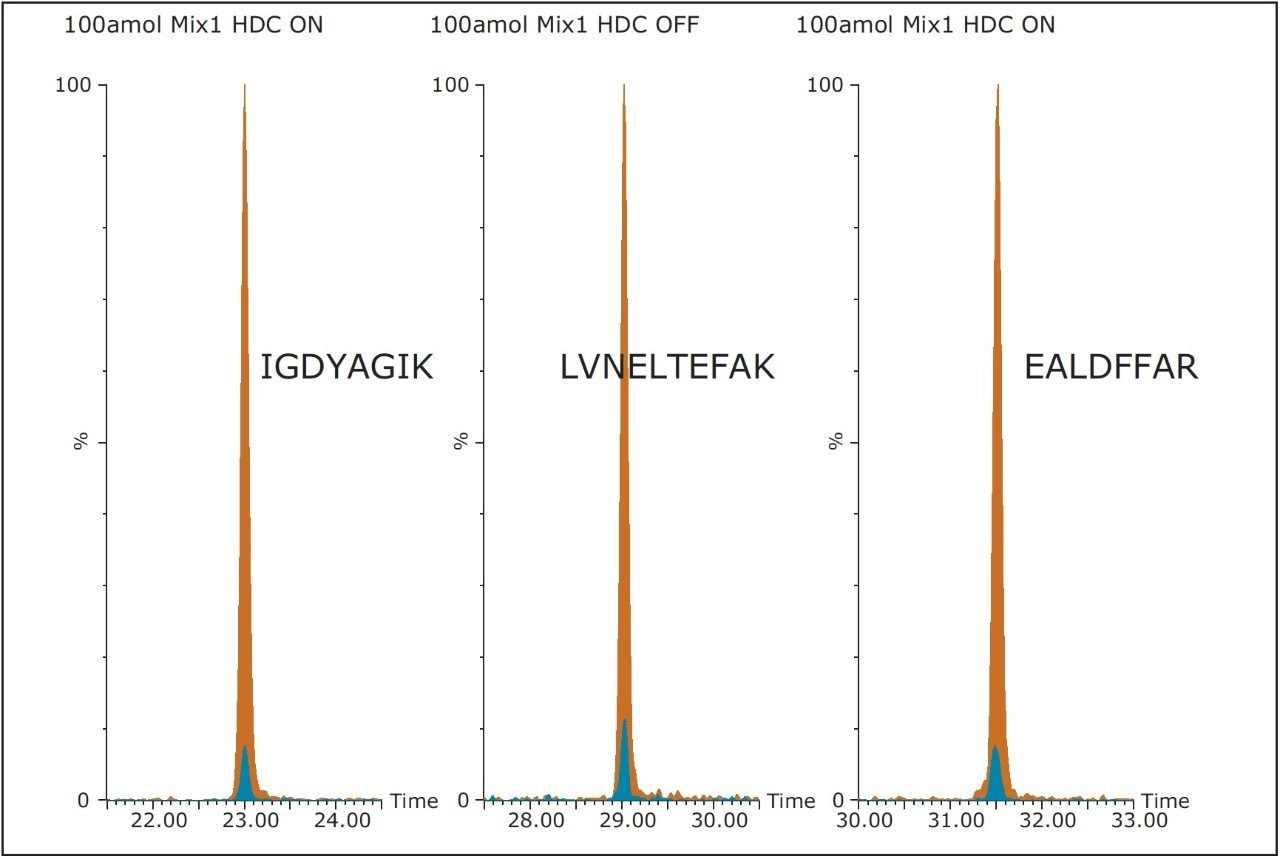
The benefit of Wideband Enhancement is demonstrated in Figure 2. Here, [Glu1]-Fibrinopeptide B solution was infused at a constant flow rate. Data were acquired by normal CID MS/MS and targeted HD-MRM. In both cases, the same number of Tof integrations were combined. For this particular experiment, >10 fold signal increase was observed. As shown in Figure 3, for example BSA and ADH peptides, summation of several product ions gives rise to increased sensitivity.
![.Product ion spectrum of [Glu1]-Fibrinopeptide B](/content/dam/waters/en/app-notes/2013/720004730/720004730en-f2.jpg.82.resize/img.jpg)
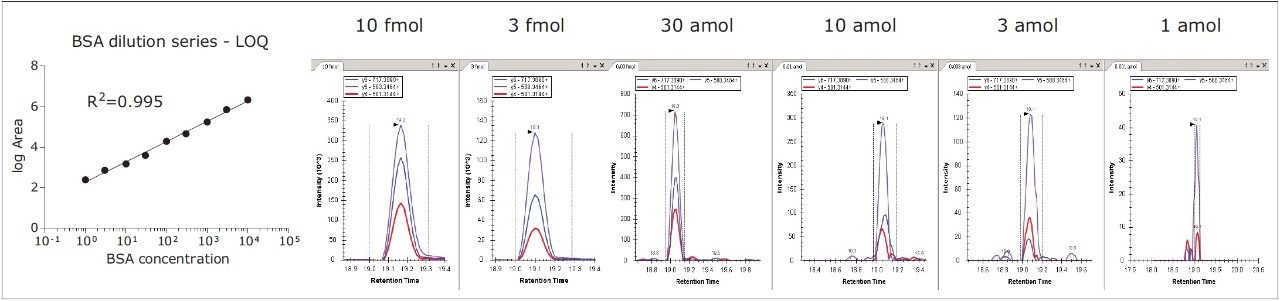
Further benefits are demonstrated by the on-line analysis of selected peptides from a tryptic digest of bovine serum albumin. The peptides were separated by nanoscale LC-MS and quantified by targeted HD-MRM. The results, shown in Figure 4, illustrate the quantification of one of the peptides over at least four orders of dynamic range, and the integration of the signal response by Skyline of three product ions/pseudo transitions.
720004730, June 2013