
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the benefits of UltraPerformance Convergence Chromatography (UPC2) as an orthogonal mode of separation to reversed phase liquid chromatography (LC).
In this study contrasting UPC2 and UPLC chromatographic techniques by peak elution order, we highlight the importance of orthogonality, the challenges associated with impurity isolation and purification, and the role of profile fingerprinting.
Chromatographic profiling of pharmaceutical drugs, raw materials, biologics, and even natural products is an important technique for scientists to best understand the characteristics related to their research or final products. Typically, scientists rely on analytical tools that provide orthogonal ways to analyze their solutes and assess their content.
For example, in pharmaceutical companies, LC columns with different selectivity are used to evaluate the raw material purity, refine route synthesis, determine finished drug product stability, and investigate container closure effects. For natural product research, different analytical techniques such as reversed-phase LC-MS, high performance thin layer chromatography (HPTLC), capillary electrophoresis with mass spectrometry (CE-MS), and gas chromatography with MS (GC-MS) are utilized to discover bioactive constituents in multiple sample extraction techniques. For bioanalytics, orthogonal methods of analysis are explored to determine drug safety and efficacy. Purification groups use orthogonal selectivity to better isolate and purify solutes of interest. In commercialization, information from orthogonal analytical tools aids in the final decisions that aim to improve product quality.
In this investigation, pharmaceutical compounds and natural products were analyzed by convergence chromatography (UPC2) and reversed-phase LC, using UPLC. Evaluating the orthogonality between the two chromatographic techniques was accomplished by tracking the peak elution order of the solutes by UV and mass spectrometry. The samples were analyzed using an ACQUITY UPLC System for the reversed-phase LC analysis and the ACQUITY UPC2 for the convergence chromatography analysis. Sub-2-μm particle stationary phases were used for each separation technique. Each example highlights the importance of orthogonality, the challenges associated with impurity isolation and purification, and the role of profile fingerprinting.
An impurity profiling method was developed for metoclopramide using both RPLC and UPC2.1 The example shown in Figure 1 demonstrates the relative retention differences between UPC2 and UPLC. Peaks that are not resolved by UPLC are resolved by UPC2, and vice versa. UPC2 also has the ability to provide longer retention for polar compounds that are difficult to retain by UPLC (e.g., peaks 1 and 2). In addition, UPC2 can be used to resolve critical pairs (i.e., peak 5 from metoclopramide), facilitating the isolation of unknown compounds for subsequent identification and characterization.
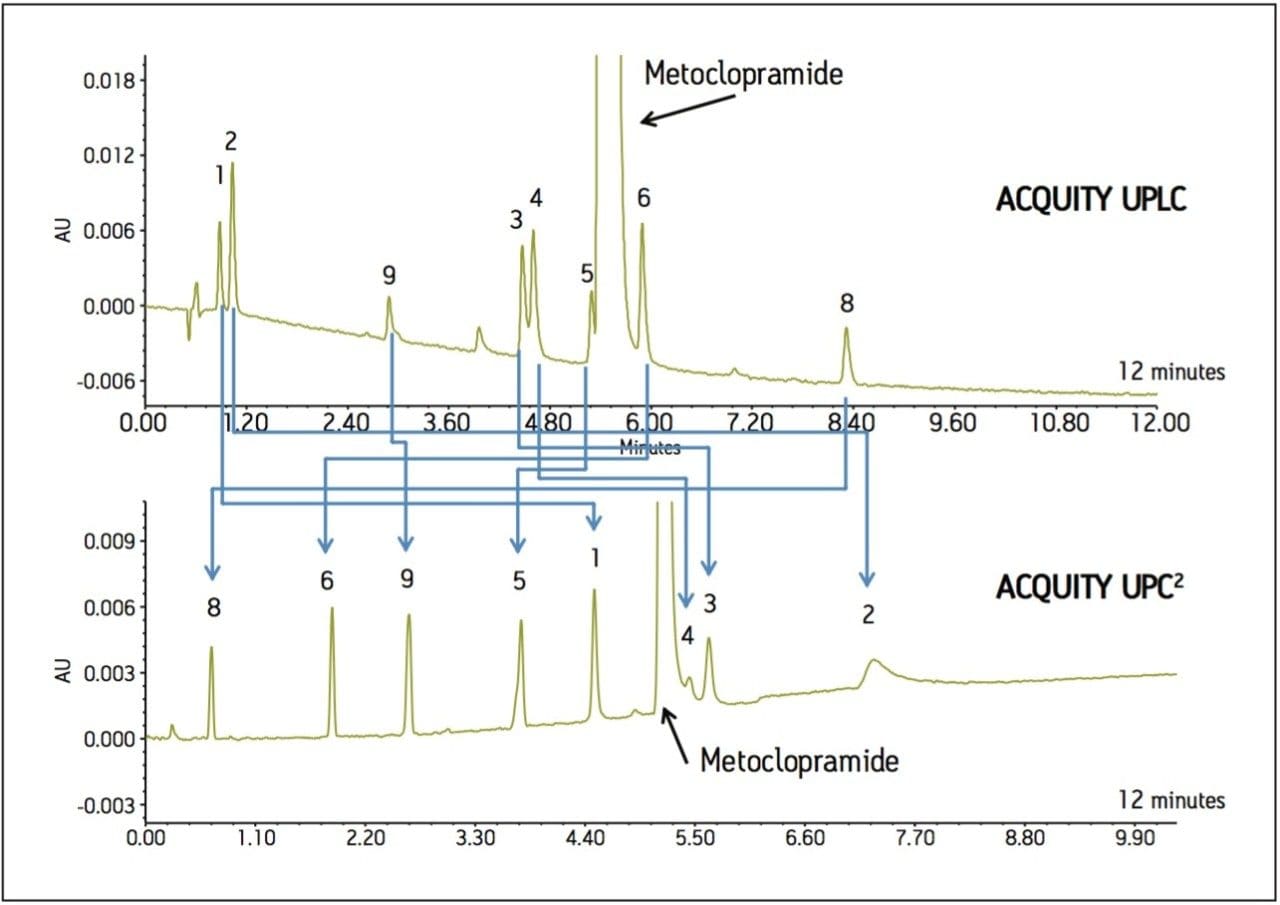
Therefore the use of both analytical tools maximizes the ability to choose an appropriate scale-up approach, effectively aiding the drug development process.
The next example demonstrates the need for orthogonal modes of separation for complex natural product extracts (German and Roman chamomile methanolic extracts). For UPLC and UPC2, peaks were identified using mass spectrometry (ACQUITY SQD System). Each peak was assigned a nominal peak I.D., whereas retention times were recorded for each UPLC and UPC2 result. The data was plotted as retention time versus peak I.D. (not shown) to visualize the orthogonality of the separation techniques. The UPC2 elution profile was observed as a scattered relationship rather than an inversely linear relationship to that of UPLC.
The highly non-polar analytes that eluted last by reversed-phase UPLC were observed to elute earlier by UPC2 (Figure 2). The real importance of an orthogonal method is illustrated with the extracted chromatograms for m/z 475 using both UPLC and UPC2 where the peaks are isobaric (same mass). UPC2 resolves an additional peak that cannot be resolved in LC, and that also cannot be distinguished by MS due to the isobaric nature of the compounds. This makes UPC2 an ideal orthogonal method for characterization of complex mixtures (Figure 3).
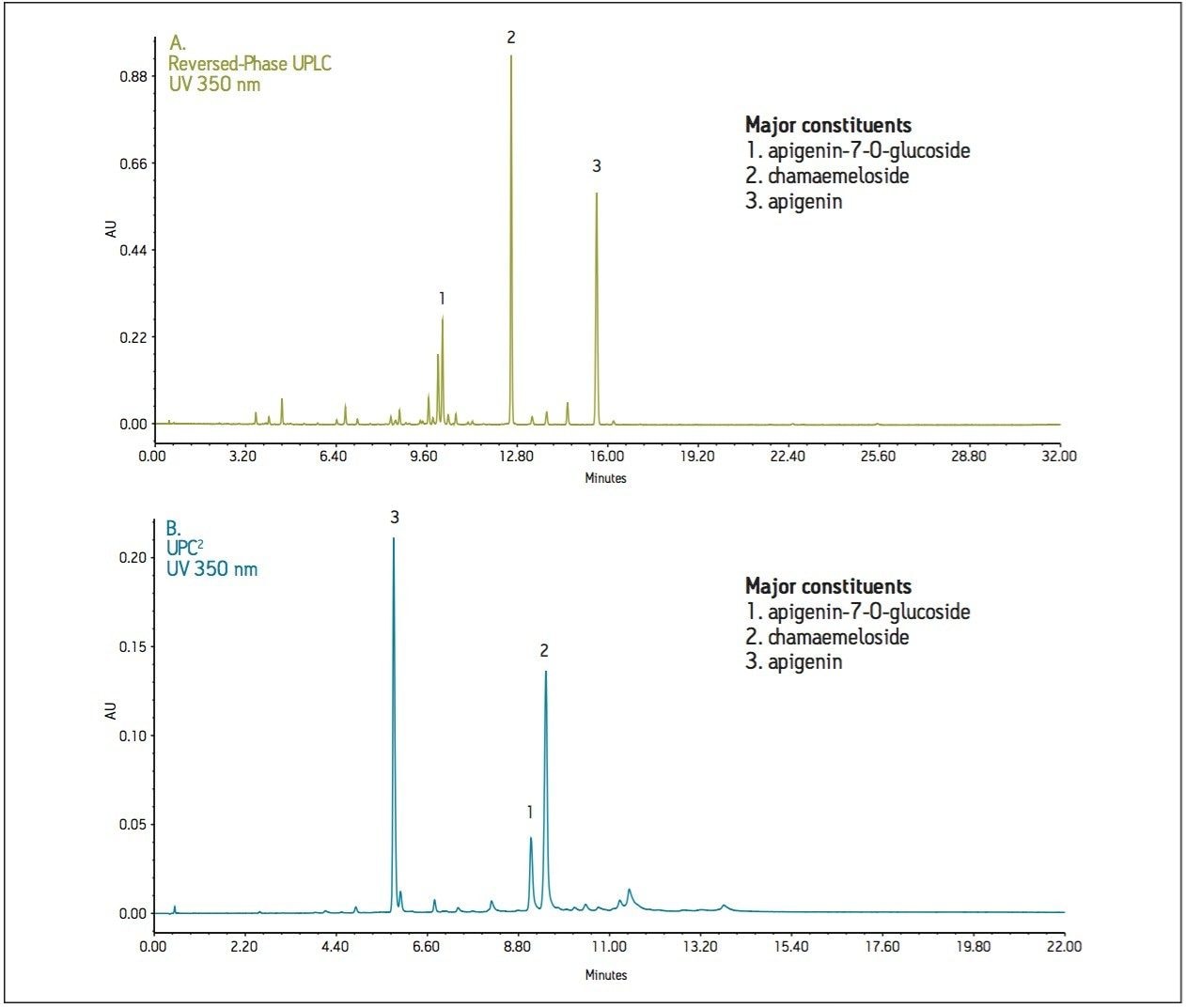
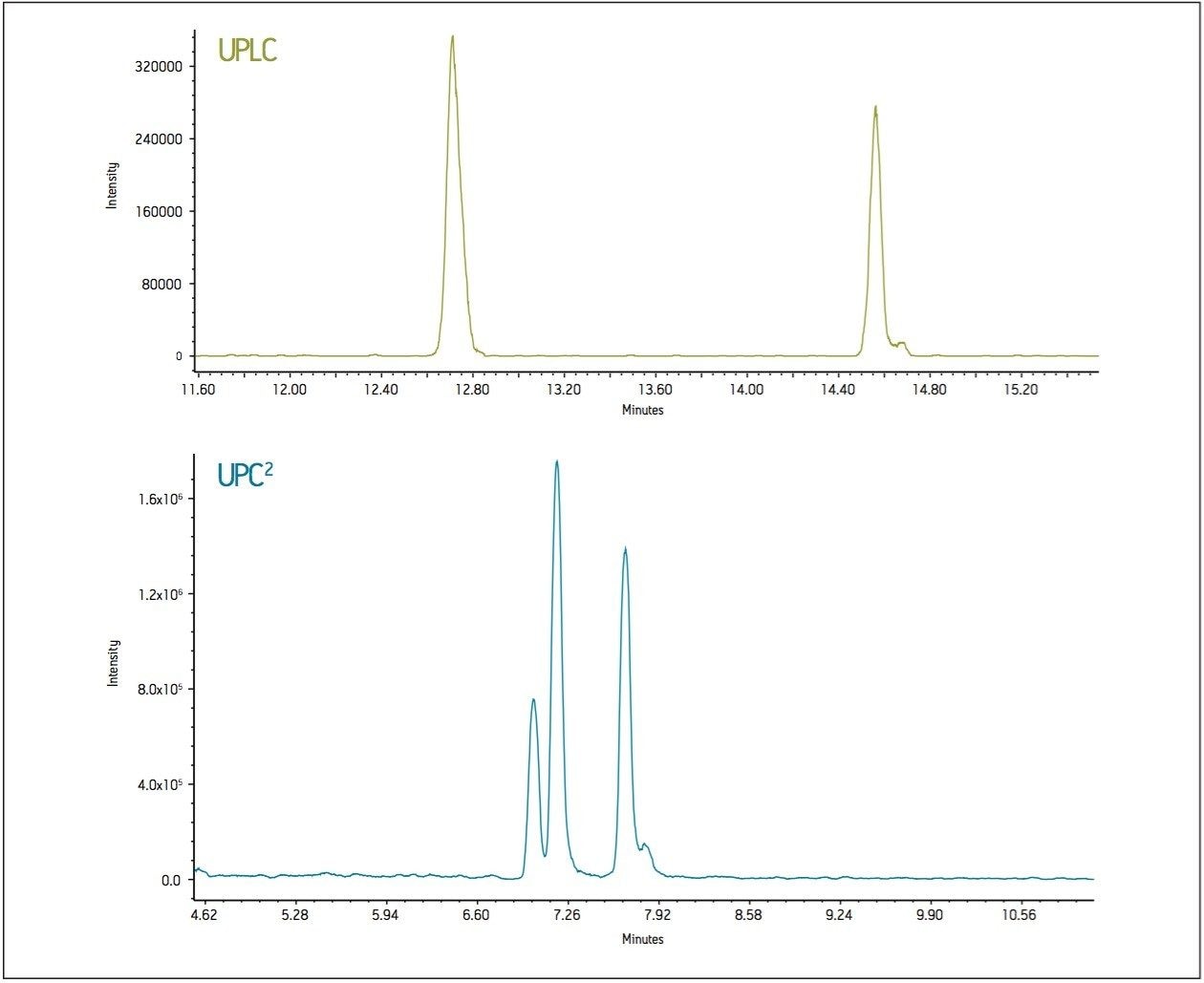
In conclusion, both application examples have proven convergence chromatography to be an effective orthogonal separation technique to commonly used reversed-phase LC. UPC2 provides a different elution order than RPLC, which is critical for resolving all peaks in a sample. From a drug development perspective, the different selectivity allows the scientist to choose a separation technique that will provide an easier path for scale-up and isolation. Unlike normal-phase LC, the separations are compatible with MS detection to give more information on unknown peaks or peak identification. The chamomile methanolic extract, directly injected for UPC2 analysis, demonstrated compatibility with many of the extraction solvents used for complex mixture analysis such as natural products. Additionally, UPC2 can resolve isobaric compounds, which cannot always be separated by other techniques.
720004689, May 2013