
This application note demonstrates a fast screening method using a simple ”dilute-and-shoot” approach for multi-residue analysis of pesticides in orange juice.
Pesticides can be detected below legislative limits in fruit juice using a “dilute and shoot” approach with the ACQUITY UPLC I-Class System coupled to the Xevo TQ-S Mass Spectrometer.
Pesticide residues in fruit juices have always been an important food safety issue, especially taking into account the high consumption of juice by children. A recent report concerning the detection of the fungicide carbendazim in orange juice has drawn widespread public attention. Since carbendazim is not licensed for use on citrus fruits in the United States, in January 2012, the Food and Drug Administration (FDA) began testing all shipments of orange juice imported into the U.S.
Many published methods are capable of analyzing pesticides in fruit juice for regulatory purposes. However, sample preparation is required for these methods in order to minimize matrix interferences. With advances in LC-MS/MS technologies, namely UPLC separation and ultra-sensitive MS detection, a fast screening method using a simple ”dilute-and-shoot” approach was evaluated for multi-residue analysis of pesticides in orange juice.
|
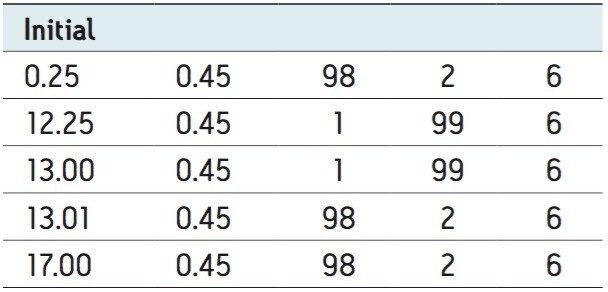
LC system: |
ACQUITY UPLC I-Class |
|
Column: |
ACQUITY UPLC BEH C18 2.1 x 100 mm, 1.7 μm |
|
Column temp.: |
45 °C |
|
Injection volume: |
10 μL |
|
Flow rate: |
0.45 mL/min |
|
Mobile phase A: |
10 mM ammonium acetate (pH 5) in water |
|
Mobile phase B: |
10 mM ammonium acetate (pH 5) in methanol |
|
Weak needle wash: |
Water |
|
Strong needle wash: |
Methanol |
|
Seal wash: |
90/10 water/methanol |
|
MS system: |
Xevo TQ-S |
|
Ionization mode: |
ESI Positive |
|
Capillary voltage: |
3 kV |
|
Desolvation temp.: |
500 °C |
|
Desolvation gas flow: |
1050 L/Hr |
|
Source temp.: |
150 °C |
A multi-residue MS method for the acquisition of two MRM transitions for each of 375 pesticides was created using Waters Quanpedia Database. Quanpedia is a compound database for quantitative LC-MS methods. By selecting “Run Samples”, Quanpedia automatically creates an LC method, data acquisition method (MRM), and the associated data processing method (quantification method) that are required to perform the analysis. The Auto Dwell and time scheduling windows provide at least 10 data points across the peak for accurate, reproducible quantification.
All pesticide standards were purchased from Sigma-Aldrich. A mix of 80 pesticides at 1000 ng/mL was prepared in acetonitrile and stored at 4 °C.
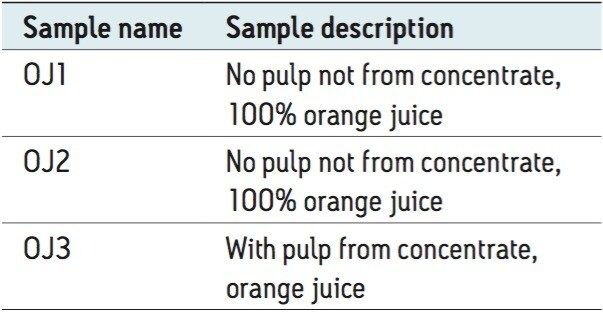
Three samples of retail orange juice were purchased to assess the detection and quantification of pesticide residues at trace levels using the “dilute and shoot” protocol. The three orange juice samples were diluted 100 times with water and filtered with 0.45-μm PTFE membrane syringe filters prior to analysis. Atrazine d5 and carbendazim d3 were used as internal standards and were spiked at 50 ng/mL into each sample. A description of the samples is provided in Table 2.
For spiked samples, a standard mix of 80 pesticides was prepared and spiked into the orange juice to give various concentrations ranging from 5 to 200 ng/mL. Following fortification the samples were diluted (100-fold) with HPLC-grade water. The lowest concentration injected was equivalent to 0.05 ng/mL (50 partsper- trillion). The diluted samples were filtered and analyzed using LC-MS/MS.
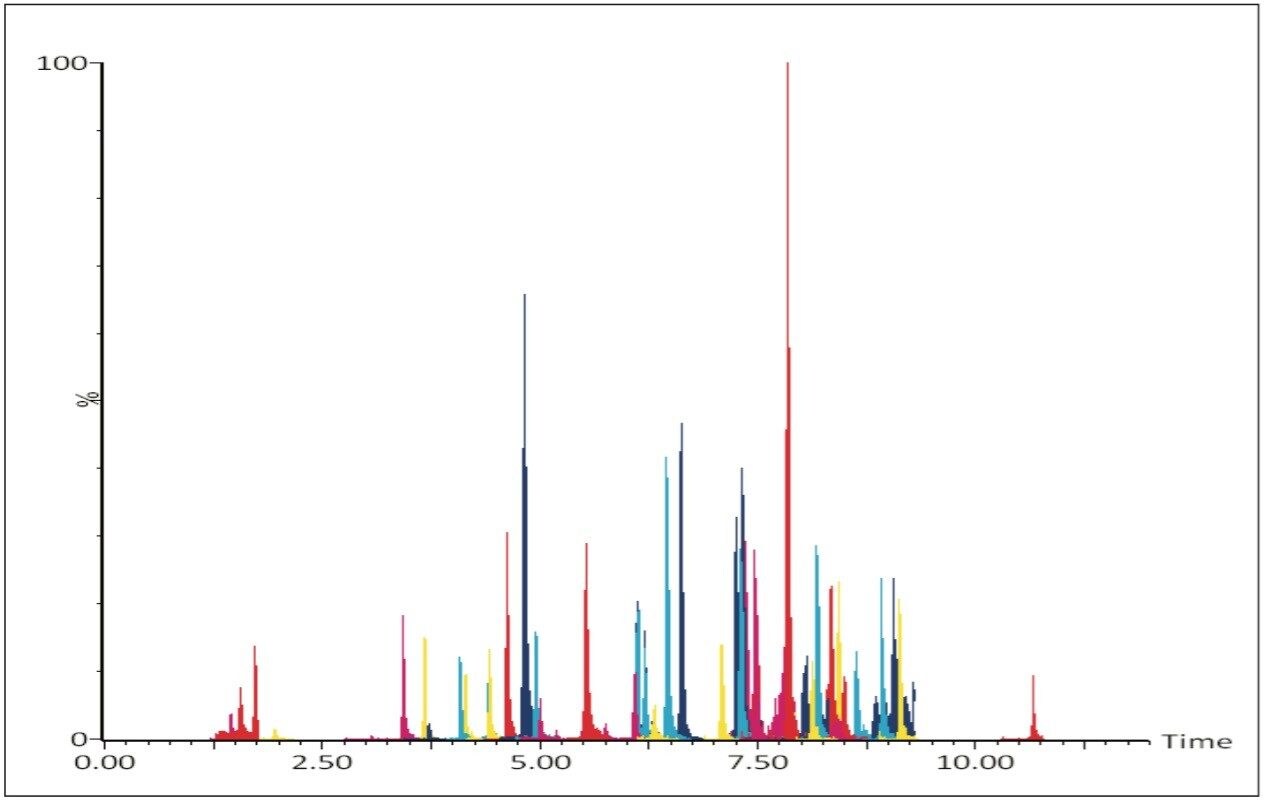
Figure 2 shows an overlay of the MRM chromatograms of the 80 pesticides spiked into sample OJ1 at 10 ng/mL.
The majority of the pesticides were detected at 5 ng/mL in orange juice without the requirement for further sample preparation. Figure 3 shows the total number of pesticides detected at the different concentrations using this simple dilute and shoot approach.
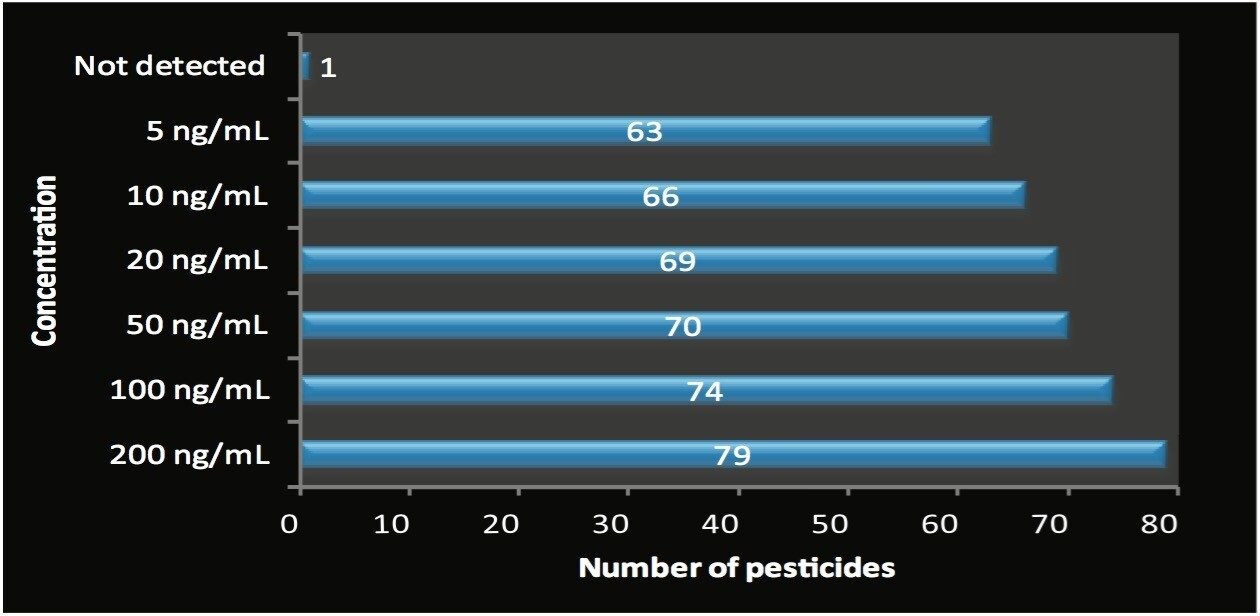
Out of the 80 spiked pesticides, 63 pesticides were detected at 5 ng/mL. At 10 ng/mL, a total of 66 pesticides were detected. All but one of the 80 pesticides were detected at 200 ng/mL.
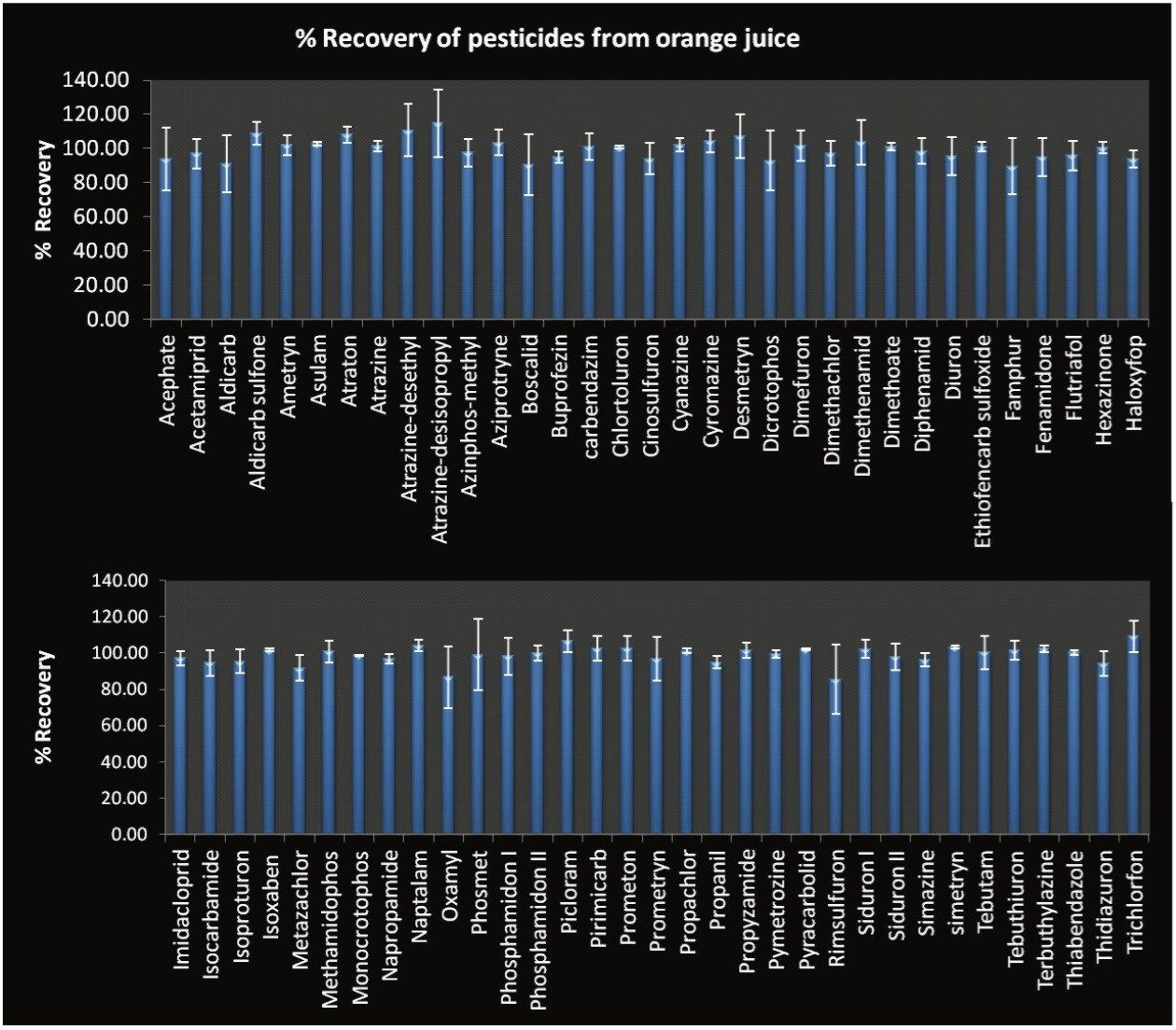
Method accuracy and precision were analyzed using recovery studies of pesticides in three samples (OJ1, OJ2, and OJ3). All samples were spiked at concentrations ranging from 5 to 320 ng/mL to generate matrix-matched calibration curves. Recoveries values expressed as percentages were calculated from samples at 10 ng/mL. Results for each matrix were obtained from samples prepared in triplicate, and each of the triplicate preparations were analyzed twice (total = 18 injections). For all of the compounds detected at 10 ng/mL in orange juice samples the average recovery ranged from 87% to 115% in orange juice samples. These recoveries fall within the performance criteria for analyte recovery specified within the SANCO 12495/2011 guidelines.1 The Relative Standard Deviations (RSDs) of the inter-sample recovery ranged between 0.9% and 19.5%. The majority showed RSDs less than 10% with only nine pesticides showing RSDs above 10%.
Figure 4 shows the average recoveries of the pesticides detected at 10 ng/mL in the three orange juice samples with error bars indicating standard deviation plus and minus the mean value.
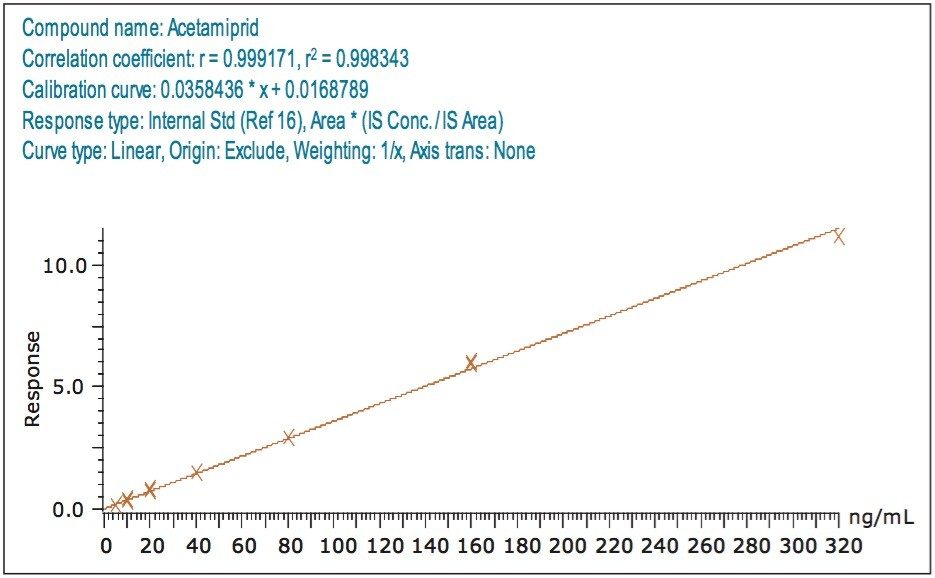
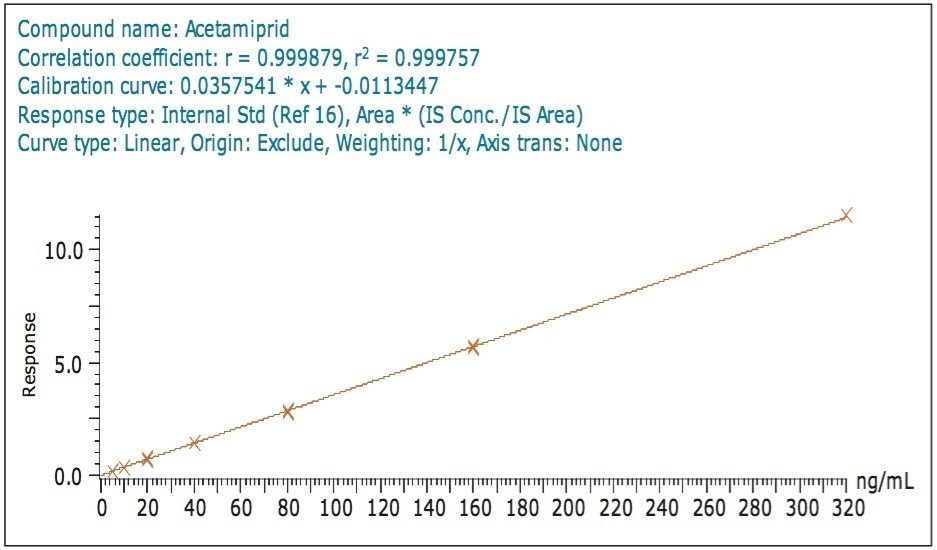
For those compounds detected at 10 ng/mL, the correlation coefficient (r2) for both the solvent standard and matrix-matched standard calibration curves ranged from 0.991 to 0.999 indicating excellent linearity. Figures 5 and 6 show calibration curves for a representative analyte, acetamiprid in solvent, and in matrix extract, respectively.
Developing analytical methods for detection of pesticides in food commodities is often challenging due to the complexity of the matrices. The presence of matrix components may cause an ion enhancement or ion suppression effect associated with the analyte signal. Matrix effects can significantly affect quantification of analytes, reproducibility, and accuracy of the overall method. It is therefore necessary to characterize and often desirable to reduce matrix interferences. This is typically achieved through sample preparation steps that are often time-consuming and laborious.
With the introduction of ultra-sensitive tandem quadruple technology, simple sample dilution is now a potential option to overcome matrix effects associated with the analysis of fruit juices.
Matrix effects were studied for all three samples by comparing the slope of calibration curve obtained in both the solvent and in the presence of matrix. An increase in the gradient of the matrix curve compared to the solvent curve indicated ion enhancement, while a decrease in the gradient of the matrix curve indicated ion suppression. A percentage variation within +20% was considered as no observable matrix effect, as this variation is close to the repeatability values. Values between +20 to +50% were considered as a medium matrix effect, and a strong matrix effect was deemed to be values above 50% and below -50%.1,2 Figure 7 shows the percentage of pesticides exhibiting matrix effects in the three different retail brands of orange juice tested.
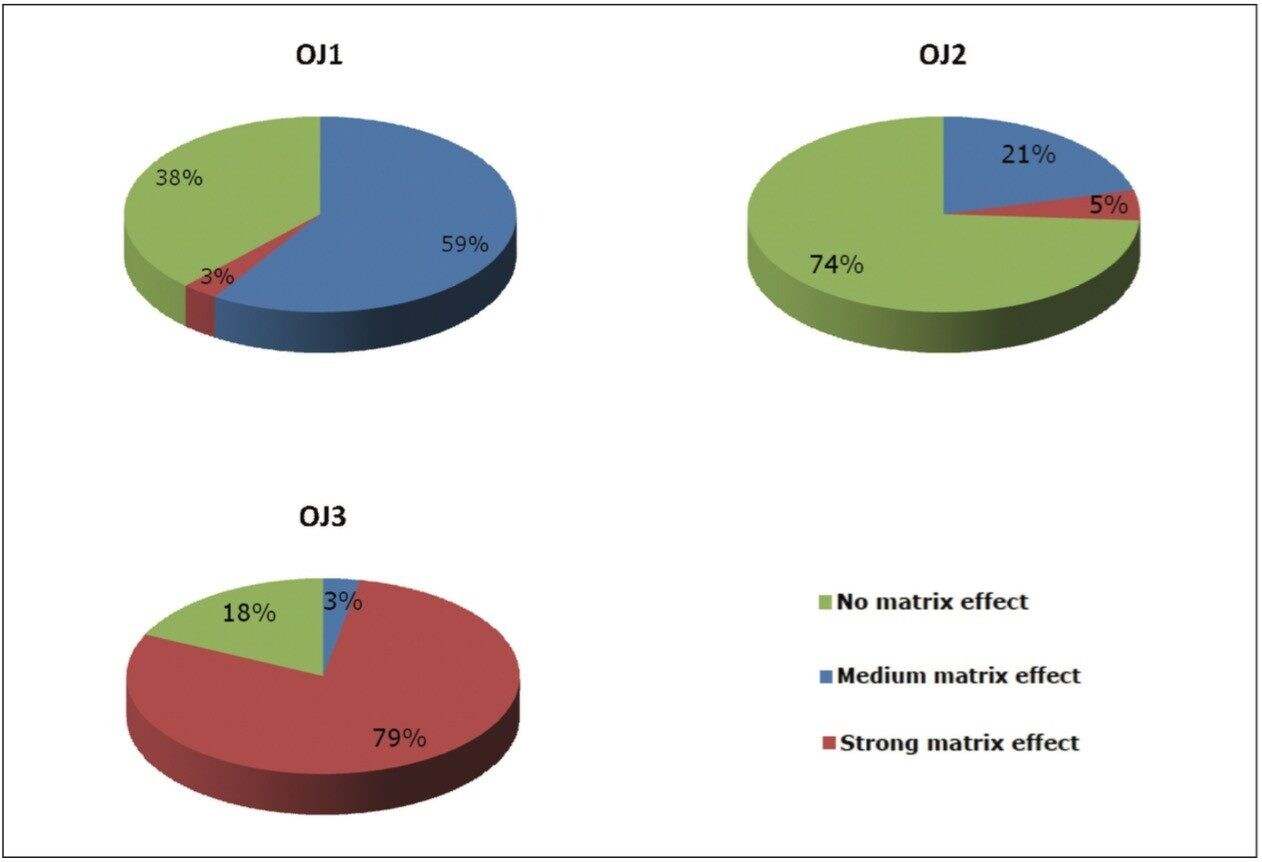
Figure 7 shows that OJ1 and OJ2 had limited matrix effects for the majority of the pesticides that were included in this study. For sample OJ2, 74% of the 66 pesticides showed less than 20% ion suppression or ion enhancement in matrix as compared to that in solvent. For OJ2 only 5% of the pesticides showed a large matrix effect. In this case quantification of samples against a solvent-based calibration curve can be used to avoid the requirement of matrix-matched calibration curves. For OJ3, however, a strong matrix effect was observed. Almost 80% of the pesticides showed either a significant suppression or enhancement. This effect may potentially be attributed to the presence of pulp in this sample. This observation is consistent with the expectation that more complex matrices require further sample cleanup.
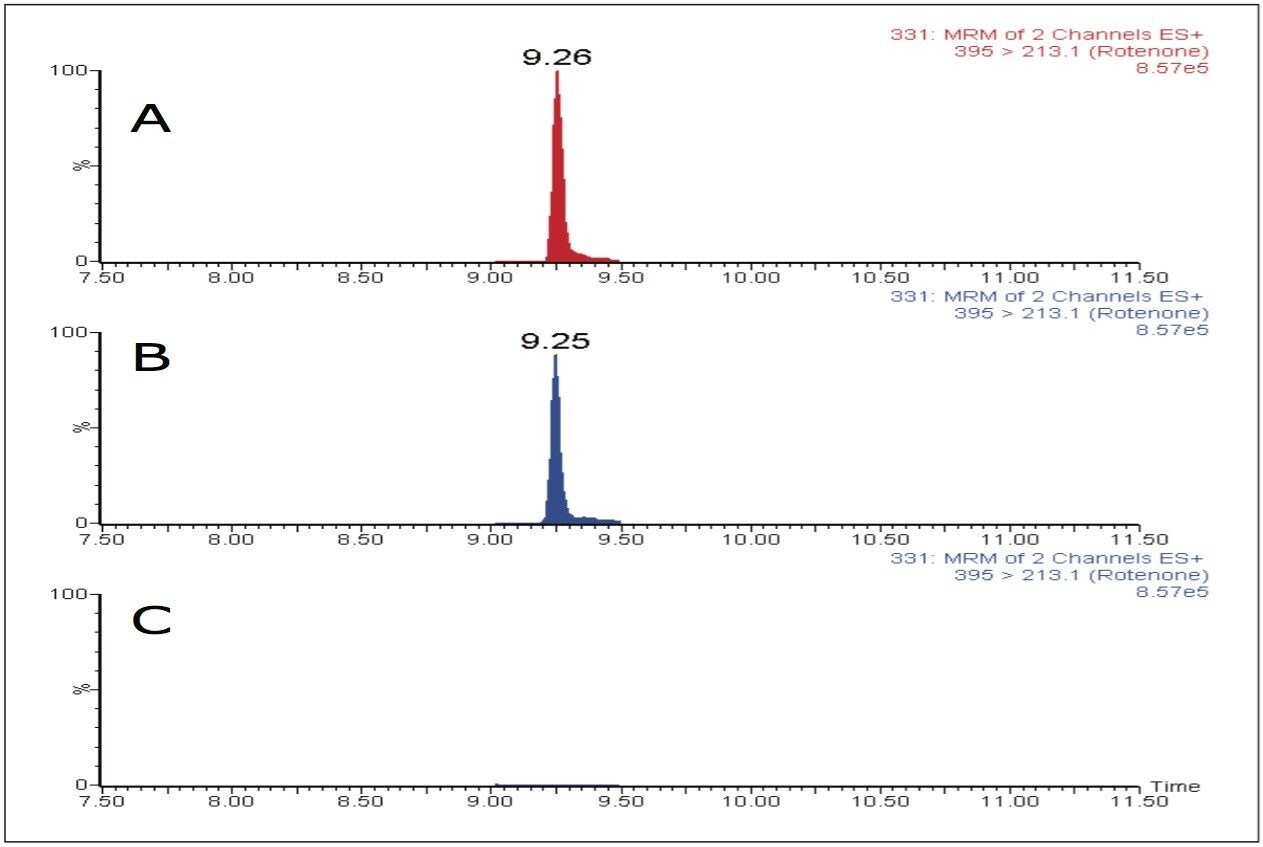
An example for the requirement of additional cleanup is the case of the pesticide rotenone. Rotenone was not detected with the dilute and shoot method due to significant matrix effects at this retention time. To determine whether sample cleanup would rectify this, orange juice (OJ1) samples were subjected to a QuEChERS-based extraction using Waters DisQuE Sample Preparation Kit.3 Figure 8 shows the MRM chromatograms of rotenone spiked at 80 ng/mL in water and diluted 100 times along with the chromatograms of OJ1 sample fortified at 10 ng/mL, prepared with QuEChERS and the dilute and shoot method, respectively. As shown in Figure 8, rotenone was easily detected following the QuEChERS extraction.
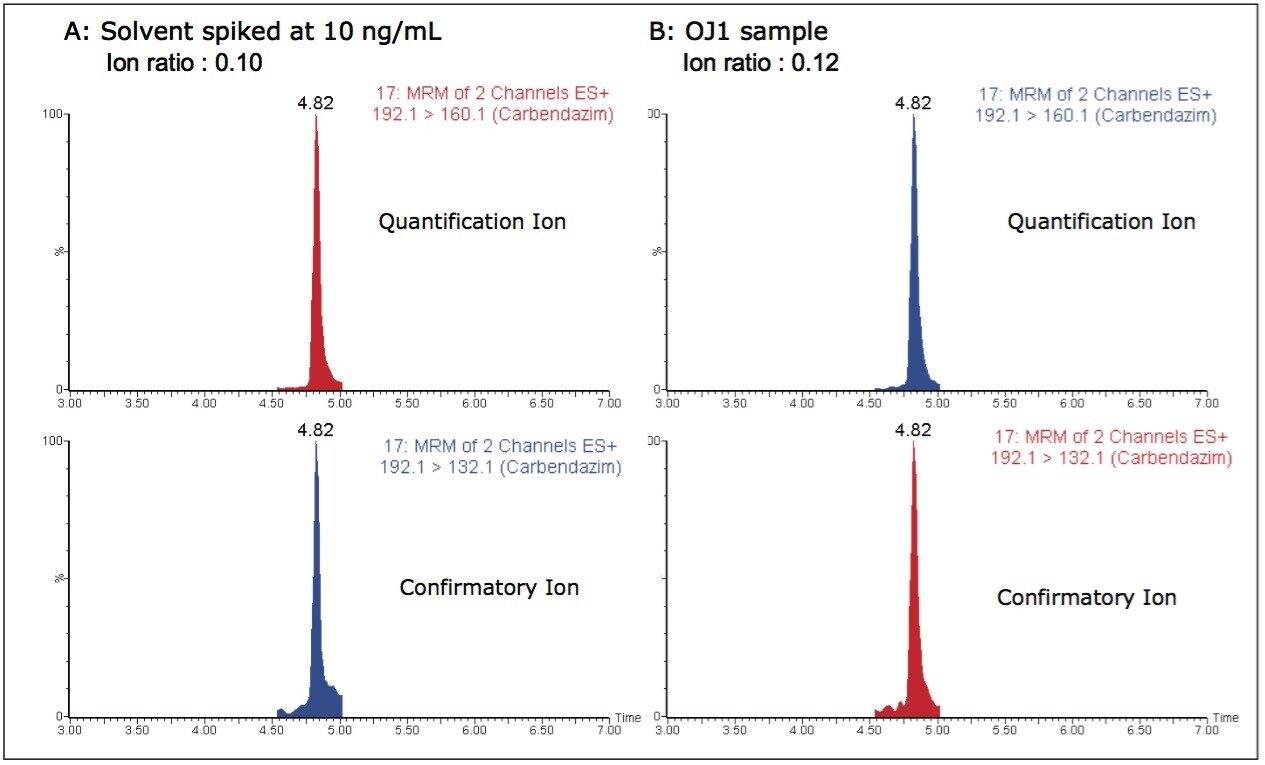
Using the dilute and shoot method, carbendazim was detected in one of the survey orange juice samples at a low concentration. The incurred carbendazim residue concentration was calculated using the standard addition method to ensure the accurate quantification and account for any matrix effects. The calculated concentration of carbendazim was determined to be 1.5 ng/mL. The identification of carbendazim was also confirmed using the expected ion ratio, based on a standard. Figure 9 shows MRM chromatograms of a carbendazim standard in water, equivalent to 10 ng/mL along with the sample of orange juice found to contain carbendazim at 1.5 ng/mL.
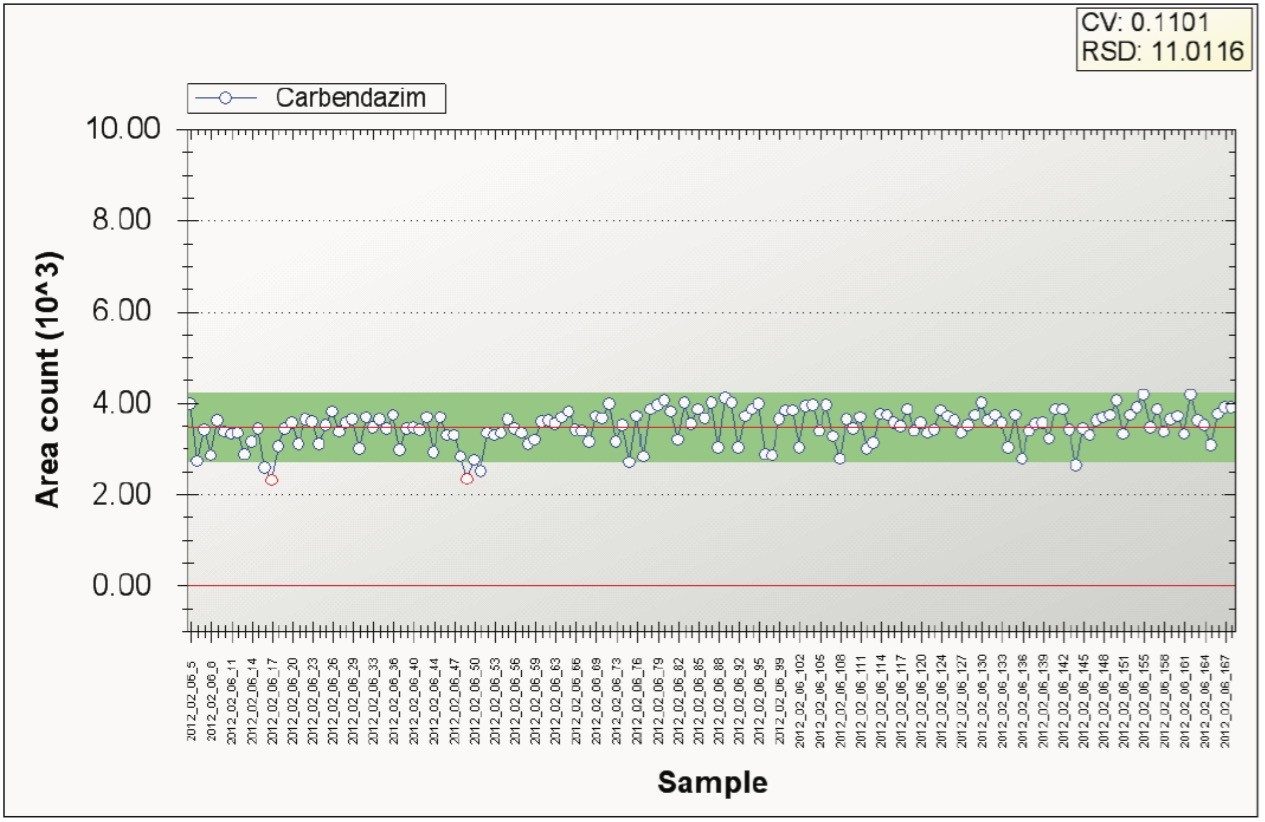
To assess the robustness of the method a study was undertaken to monitor the effects of continuous injections of diluted juices over 44 hours. Figure 10 shows the TrendPlot graph of the repeatability of 155 injections of orange juice spiked with carbendazim at 10 ng/mL concentration. No decrease in performance was observed over the course of this study.
720004403, July 2012