
This application note presents a simplified, streamlined, analytical approach to BPA analysis using QuEChERS-based methodology. The QuEChERS extraction and subsequent SPE cleanup protocols provide a faster analysis with less toxic reagents, and similar detection limits compared to other methods.
Bisphenol A (BPA) is a possible endocrine disrupter that can mimic the body’s own hormones, leading to possible adverse health effects. In 2001, concerns about the estrogenic activity of BPA were raised, and several governments released reports questioning the safety of its use in consumer products. There has been a concomitant interest in the development of analytical methods for the determination of BPA in foods intended for human, particularly infant, consumption. BPA can be extracted from foods such as infant formula using methylene chloride, acetonitrile, or other organic solvents. Since infant formula is such a complex matrix, significant and often cumbersome cleanup and enrichment steps are employed following the initial extraction. This application note presents a simplified, streamlined, analytical approach to BPA analysis using a QuEChERS-based methodology.
QuEChERS methodology is quicker, easier, cheaper, safer than, and as rugged as alternative methods of sample preparation. The methodology is now commonly used worldwide for sample preparation of fruits and vegetables, prior to GC-MS or LC-MS determination of pesticide residues. This extraction and cleanup technology can be applied to other types of analytes and sample matrices as an alternative to more cumbersome methods of sample preparation. Requirements for the sample matrix include the following: can be readily homogenized, and is suitable for extraction with acetonitrile. The matrix should contain 75% or more water by weight, which is essential for the subsequent liquid partition cleanup. If sufficient water is not present in the sample matrix, it should be added to the homogenized sample prior to QuEChERS extraction. In order for a compound to be efficiently extracted using QuEChERS methodology, it must have a suitable partition co-efficient when partitioned between acetonitrile and salt-saturated water. Prepared infant formula meets the matrix criteria for QuEChERS extraction. BPA meets the analyte liquid/liquid partition criteria for QuEChERS extraction and cleanup.
This application note presents a rapid QuEChERS-based method for the determination of BPA in infant formula at low ng/g (ppb) levels. The method employs a DisQuE pouch product for QuEChERS-based sample preparation. The resulting extract is first cleaned-up using dispersive SPE (dSPE), and is further cleaned-up and concentrated using an Oasis HLB cartridge. The QuEChERS extraction and subsequent SPE cleanup protocols provide a faster analysis with less toxic reagents, and similar detection limits when compared to other methods.
|
System: |
ACQUITY UPLC H-Class |
|
Column: |
ACQUITY BEH C18 3.0 x 100 mm, 1.7 μm (p/n 186005301) |
|
Injection volume: |
30 μL |
|
Temp.: |
40 °C |
|
Mobile phase A: |
0.1% NH4OH in water |
|
Mobile phase B: |
0.1% NH4OH in MeOH |
|
Gradient: |
50% B initial (hold for 0.2 min), linear gradient to 95% B for 3.0 min, hold for 5.0 min, back to 50% B for 5.1 min. Hold and re-equilibrate for 7.0 min. |
|
Flow rate: |
0.6 mL/min |
|
Vial: |
Total Recovery (p/n 186005663CV) |
|
Mass spectrometer: |
Waters ACQUITY TQD |
The mass spectrometer was operated in electrospray negative (ESI-) mode. The MRM transitions, cone voltages, and collision cell energies used for BPA analysis were as follows, with primary transition used for quantification appearing bold:
|
MRM Transition Cone Collision (m/z) |
(V) |
(eV) |
|---|---|---|
|
227.3 > 133.0 |
40 |
23 |
|
227.3 > 212.1 |
40 |
17 |
Place 10 g liquid infant formula into a 50-mL centrifuge tube. Add 10 mL acetonitrile (ACN), and shake the tube vigorously for 1 min. Add contents of DisQuE pouch for CEN QuEChERS (p/n 186006813), and shake vigorously for 1 min. Centrifuge for 3 min at 4000 rpm, and take a 5-mL aliquot of the supernatant (top layer) for dSPE cleanup.
Transfer the 6-mL aliquot of supernatant to a 15-mL dSPE cleanup tube that contains 900 mg of magnesium sulfate, 150 mg PSA sorbent, and 150 mg of C18 sorbent (p/n 186004834), and shake vigorously for 1 min. Centrifuge for 3 min at 4000 rpm, and take a 1.5-mL aliquot of the supernatant for Oasis HLB SPE enrichment and cleanup.
Transfer the 1.5-mL aliquot from dSPE to a suitable container, dilute to 10.0 mL with reagent water, and mix. Condition an Oasis HLB cartridge 10 mg, 1 cc (p/n 186000383) with 0.5 mL methanol, followed by 0.5 mL of water. Load the diluted extract at a flow rate of 1 to 3 mL/minute. After sample loading is complete, wash the cartridge with 0.3 mL 40:60 methanol/water. Install collection vessel, and elute with 150 µL methanol. Dilute the eluted sample with 150 µL water prior to UPLC analysis. Transfer diluted sample to the Waters Total Recovery Vial.
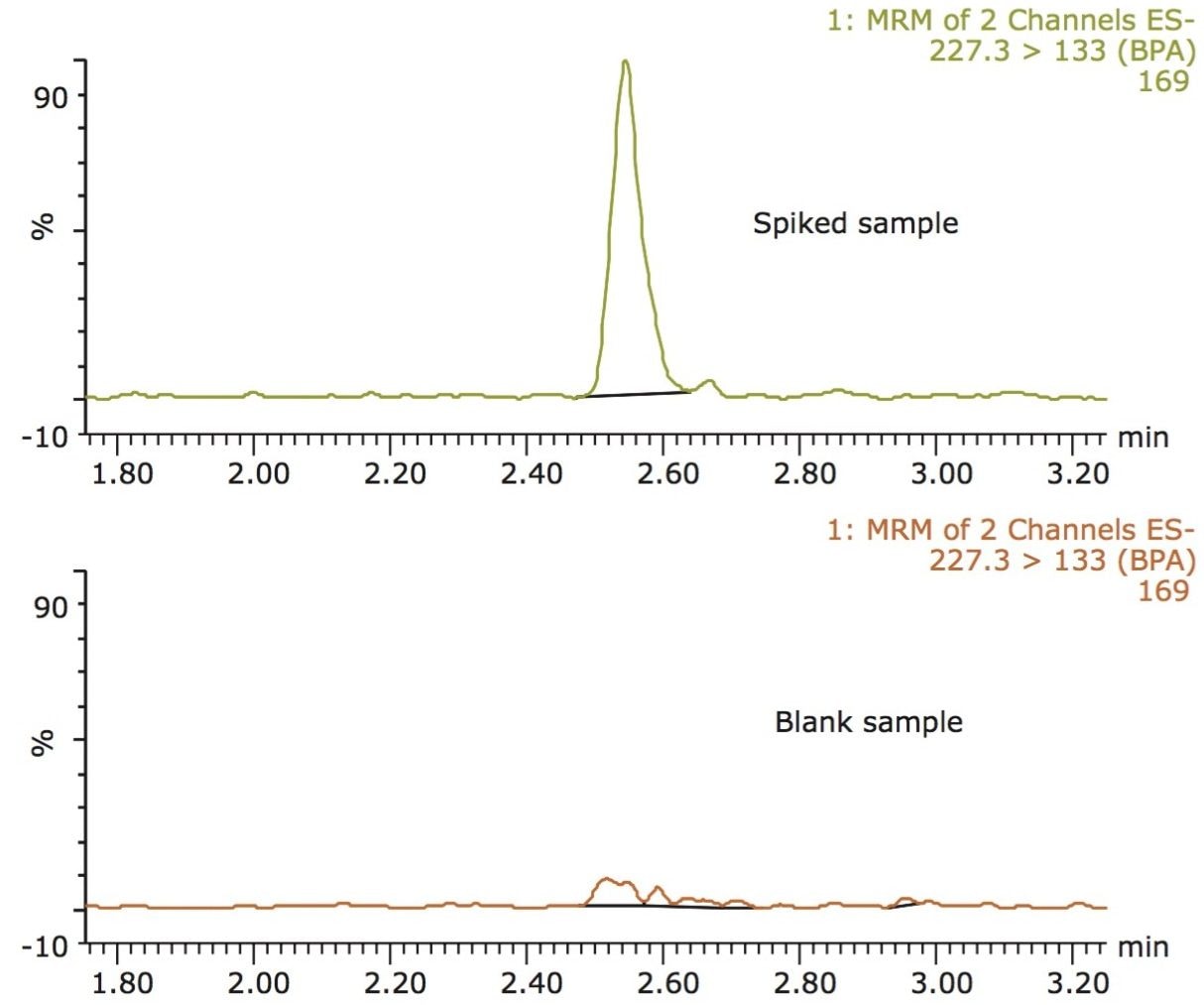
Figure 1 shows a reconstructed UPLC-MS/MS chromatogram obtained from the analysis of an infant formula sample, spiked at 2 ng/g with BPA. BPA recovery was measured at 2 and 20 ng/g (n = 5 for each level). Recovery at 2 ng/g was 88% with 6% RSD. Recovery at 20 ng/g was 98% with 11% RSD. Matrix effects (ion-suppression or enhancement) were under 10%. Recovery was calculated by comparing the MRM peak area for samples spiked prior to QuEChERS extraction (pre-spiked samples) with the MRM peak area for samples spiked after QuEChERS extraction (post-spiked samples). Matrix effects were calculated by comparing the MRM peak area of post-spiked samples with the MRM peak area for equivalent standards, prepared in 50:50 methanol/water.
720004455, September 2012