
In this study, we report a systematic investigation on the general applicability of SFC for chiral separations. A total of 176 diverse racemic small molecule compounds were screened; many are commercially available pharmaceutical compounds across several disease areas.
By screening a total of 176 diverse, racemic, small molecule compounds, it was demonstrated that SFC is a highly successful chromatographic technique for the chiral separation of small molecule compounds. SFC should be adopted as the technique of choice for chiral analysis and purification across many industries, including pharmaceutical, agrochemical, and nutraceutical.
Chiral chromatography, including gas chromatography (GC), capillary electrophoresis (CE), high performance liquid chromatography (HPLC), counter-current chromatography (CCC), and supercritical fluid chromatography (SFC), have been used in chiral resolution.1 Among these, HPLC and SFC on chiral stationary phases (CSPs) are the most widely utilized chromatographic techniques in drug discovery for obtaining milligrams to multi-grams of pure enantiomers.2
There has been growing awareness of SFC, largely driven by the pharmaceutical industry.1-3 However, they often deal with proprietary compounds for specific disease areas.4-7 For pharmaceutical compounds, factors such as target disease area (e.g. oncology, inflammation, or psychiatry), route of administration (oral or intravenous), and location of target receptor proteins (cell surface, inside cell, or between cells) pose different requirements on the molecules. Consequently, drug candidates for different disease areas may concentrate in different zones of diversity space.
Herein, we report a systematic investigation on the general applicability of SFC for chiral separations. A total of 176 diverse racemic small molecule compounds were screened; many are commercially available pharmaceutical compounds across several disease areas. The experiments were carried out in a parallel manner under selected chromatographic conditions to maximize the success rate while adhering to the principles of speed and simplicity, often required in drug discovery.
A total of 176 commercially available small molecule racemates were selected for this study. The structures of the compounds were imported to ChemDBsoft Software (TimTec, Inc., Newark, DE, USA) for diversity analysis. All compounds were dissolved in methanol at 1 to 2 mg/mL.
|
System: |
Method Station X5 SFC System (abbreviated as the “X5 SFC System” hereafter) |
|
Sample manager: |
Alias AutoSampler Fluid Delivery Module |
|
Column oven: |
Analytical-2-Prep Column Oven (10-port) Automated Back Pressure Regulator |
|
Detection: |
Four 2489 Tunable UV/Vis (TUV) Detectors, one 2998 Photodiode Array (PDA) Detector |
|
Data management: |
MassLynx Software |
|
Columns: |
AD-H, OD-H, OJ-H, AS-H, and IC (4.6 x 250 mm, 5 μm, Chiral Technologies West Chester, PA, USA) |
|
System: |
Method Station X5 SFC System (abbreviated as the “X5 SFC System” hereafter) |
|
Sample manager: |
Alias AutoSampler Fluid Delivery Module |
|
Column oven: |
Analytical-2-Prep Column Oven (10-port) Automated Back Pressure Regulator |
|
Detection: |
Four 2489 Tunable UV/Vis (TUV) Detectors, one 2998 Photodiode Array (PDA) Detector |
|
Data management: |
MassLynx Software |
|
Columns: |
AD-H, OD-H, OJ-H, AS-H, and IC (4.6 x 250 mm, 5 μm, Chiral Technologies West Chester, PA, USA) |
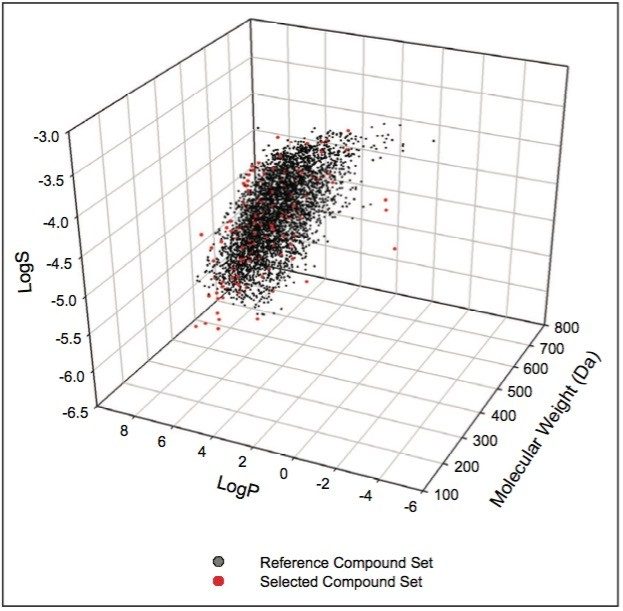
To ensure that our compound selection represented the types of chemical entities typically encountered in pharmaceutical research while maintaining a manageable sample size, we selected compounds with a wide range of diversity in physical property, structure, and functional group. The compound selection included esters, alcohols, acids, amines, and sulfoxides, to name a few (see Appendix). The structures of these compounds were imported to the ChemDBsoft Software, and the diversity was assessed by the following computed values: Lipinsky score, molecular weight, LogP, LogS, rotational bonds, proton donors, and proton acceptors. The results were then compared to those from TimTec’s ApexScreen diverse compound library containing a total of 5,040 compounds.8 Figure 1 shows the distributions of our compound selection and the reference ApexScreen diverse library in the chemical space defined by molecular weight, LogP, and LogS. Clearly, albeit being a much smaller sample set, our compound selection covers a similar spatial span as the diverse ApexScreen set. Furthermore, based on the proprietary algorithm of ChemDBsoft for diversity analysis, our compound selection has a numeric diversity score of 0.84; whereas the ApexScreen library has a score of 0.90. Overall, the diversity analysis indicates that our compound selection is sufficiently diverse for the purpose of this evaluation.
When developing a chiral SFC separation, users often start with the screening of multiple columns and multiple mobile phases to determine the optimal chromatographic condition. Currently, there are over 200 commercially available CSPs. However, it has been the experience of many users that derivatized polysaccharides-based CSPs are extremely successful in SFC.2, 4-7 The AD-H, OD-H, OJ-H, AS-H, and IC columns were chosen in this study.
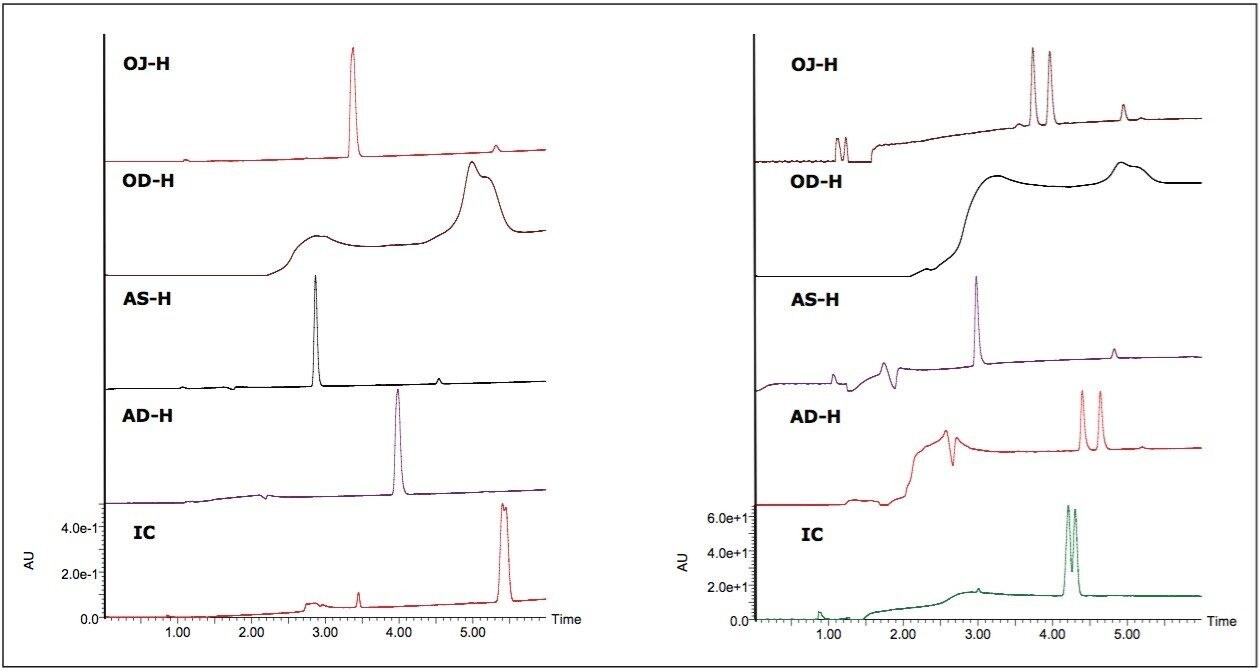
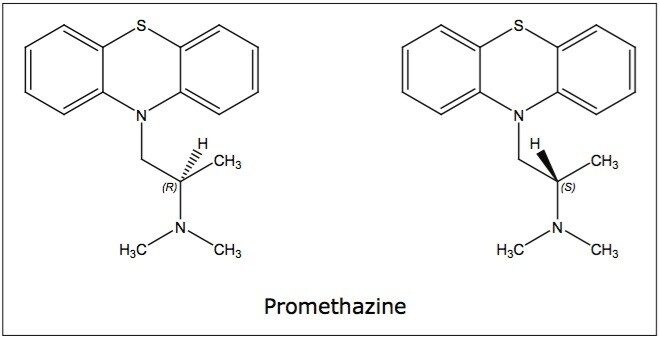
In terms of co-solvent selection, methanol and isopropanol were first chosen, as they often induce vastly different selectivity in chiral chromatography. Since derivatized CSPs typically do not need acidic additives when used in SFC, the two alcohols spiked with 0.2% DEA were also included for basic compounds. An example of the co-solvent effect is shown in Figure 2. With methanol as the co-solvent, there was no separation for promethazine. When isopropanol with 0.2% DEA was used, baseline resolution was achieved on the OJ-H and AD-H columns, and partial separation was achieved on the IC column.
At the drug discovery stage, minimizing the time required to obtain pure enantiomers is of paramount importance.2, 9 It should be noted that the primary objective of this study is to gauge the general applicability of SFC for chiral resolution through a “first-pass” or “tier-1” screening,6 rather than an exhaustive search for the optimal conditions for each compound. Thus, the experiments were designed to reflect simplicity and speed, which are often required in such analyses at the drug discovery stage. A 6-min generic gradient (5% to 45% in 5 min and 45% to 5% in 1 min) followed by a 2-min re-equilibration (an 8-min injection cycle) was used in this study. However, to run a total of 176 compounds sequentially, with the selected five columns and four co-solvents, it would take a formidable total of 469 hrs (176 x 8 x 5 x 4 = 28,160 min) to finish all the experiments. To that end, the X5 SFC System lends itself as a facile solution to expedite the screening process in an automated fashion.10 In the parallel mode, an injected sample is carried by the mobile phase and divided into five columns. As a result, five columns are screened simultaneously. Two examples are demonstrated in Figure 2. Each injection resulted in five chromatograms. In this study, using the X5 SFC System, all experiments were finished in less than 100 hrs, representing an 80% reduction in the total analysis time.
All chromatograms were manually inspected. Based on the best chromatogram for each compound, the compounds were categorized as baseline resolved (Rs > 1.5), partially resolved (0.3 < Rs < 1.5), and not resolved. The results were tabulated in a Microsoft Excel Spreadsheet. In the cases where separations were observed on multiple columns, the one generating the highest resolution was recorded as the “hit” column.
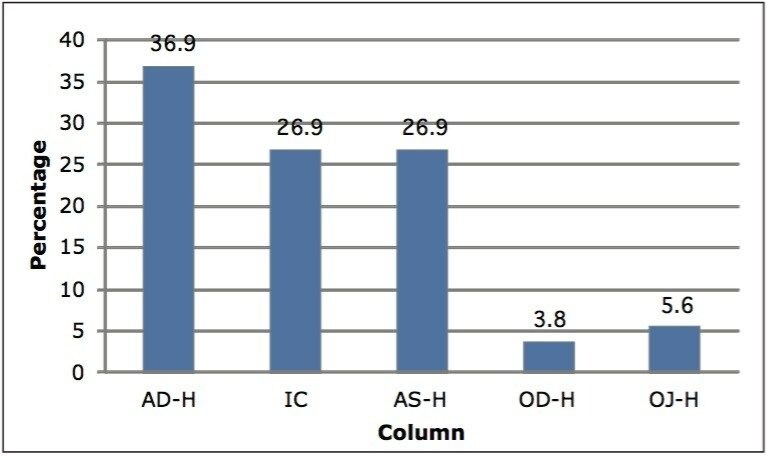
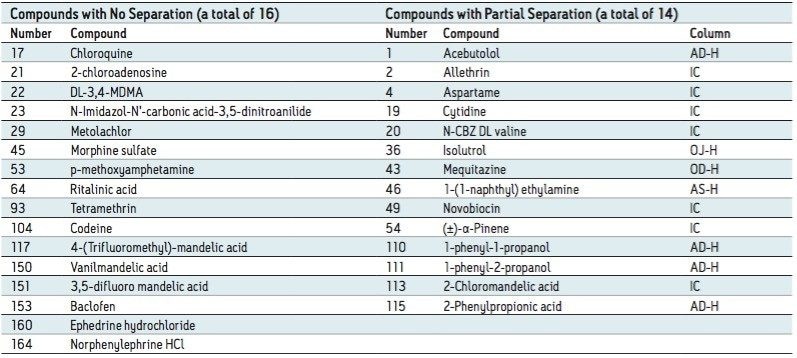
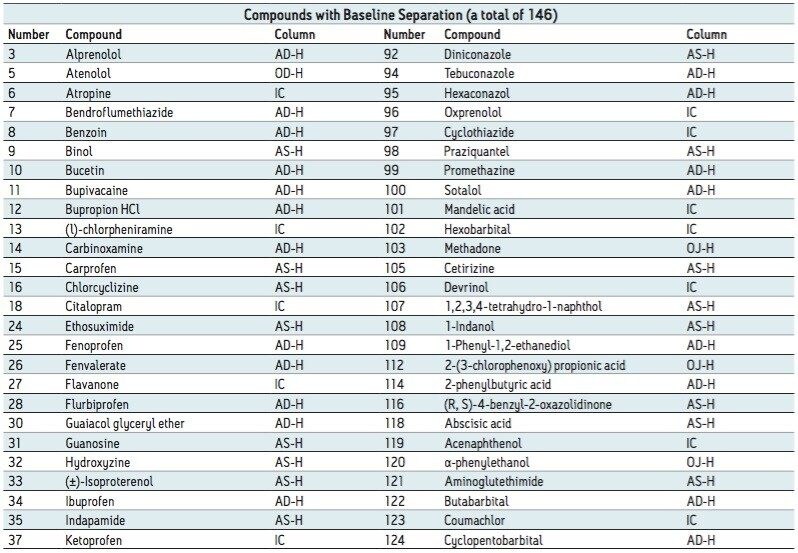
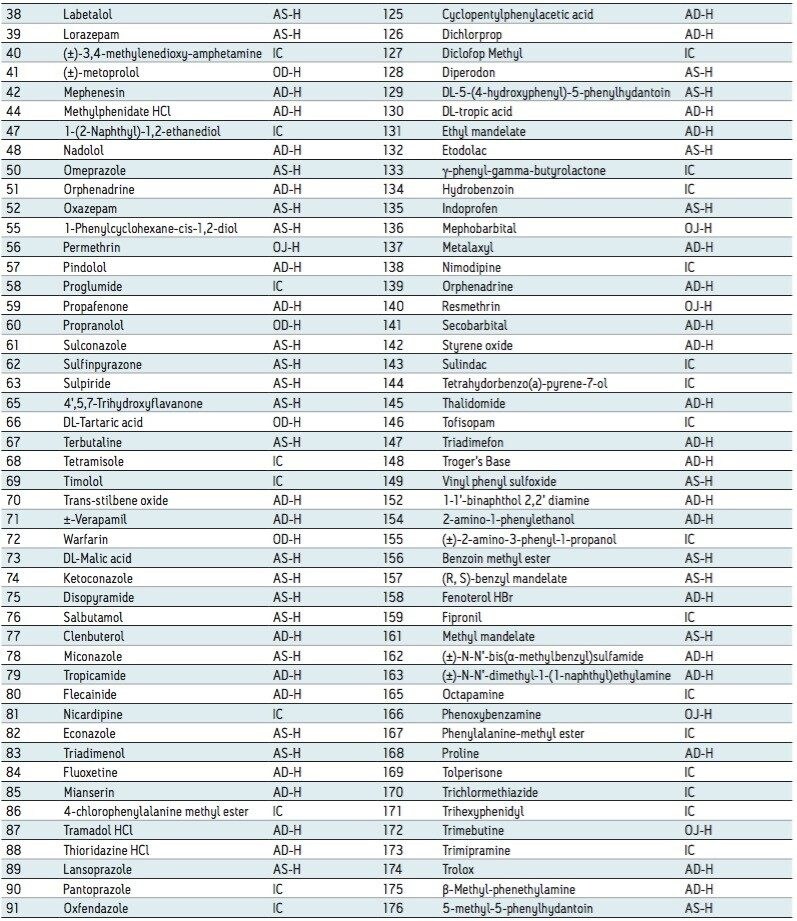
Among the 176 compounds selected, 146 compounds were baseline resolved, 14 compounds were partially resolved, and 16 compounds were not resolved. The results are summarized in Figure 4. Overall, partial or baseline separations were observed for 91.5% of the compounds; of those, 83.5% of the compounds were baseline resolved. Our observation is similar to the study by Riley et al.6 In their survey, a total of 120 proprietary compounds were screened using the same five columns used in our study with different co-solvents. An overall 95.6% success rate was reported. It should be noted that this observation was within the boundaries of the selected experimental conditions. In practice, for those partially or not resolved compounds from the “first-pass” screening, additional screening with different columns and/or co-solvents can further improve the success rate.
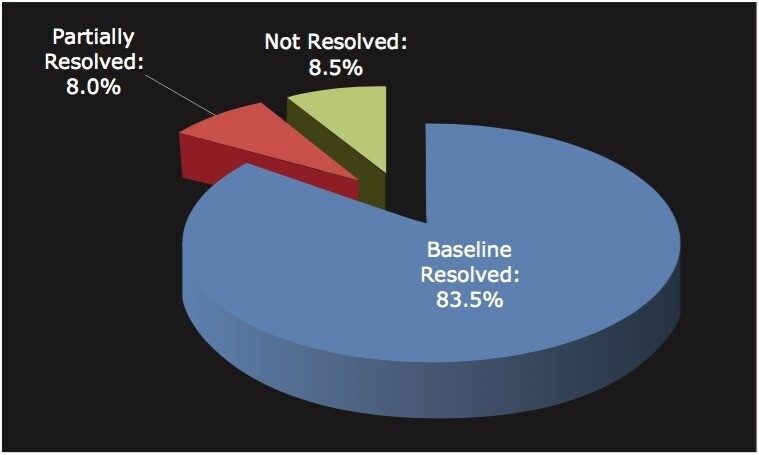
Figure 5 shows the distribution of the column types for the 160 compounds that were baseline or partially resolved. The AD-H column yielded the highest number of “hits”, while the IC and AS-H columns shared the second place. The AS-H and OD-H columns were the distant fourth and fifth, respectively. These results support the perception that no single column is overwhelmingly “universal.” Even with the AD-H column, the success rate was only 37%. These observations also agree with other reports4-7 that AD-H is the most successful derivatized polysaccharides-based CSP in terms of general applicability. However, it is noteworthy that the order of columns in terms of success rate varies from study to study. Barnhart et al.4 reported that for 40 commercial compounds, the order was AD-H > OD-H > AS-H > OJ-H > P-CAP; however, for 100 proprietary Amgen compounds under the same experimental conditions, that order became OD-H > AD-H > AS-H > OJ-H >P-CAP. In the study reported by Riley et al.,6 the order was AD-H > OD-H > OJ-H > IC-H > AS-H for 120 proprietary Pfizer compounds. There are several reasons that could contribute to the observed discrepancy. First, the order of columns is highly compounddependent, as demonstrated by Barnhart et al.4 Two different orders were generated for two different sets of compounds, while other conditions remained same. As a result, caution should be exercised to ensure sample diversity when a general conclusion is drawn. Secondly, the makeup of the selected columns could also change the “hit” distribution. An ideal column selection for screening should include a minimal number of columns with complementary selectivity to maximize the success rate. And finally, the selection of co-solvents also contributes to the “hit” column distribution. This is evidenced by the comparison between our results and the one reported by Riley et al.6 From a chromatography perspective, the main difference lies in the selection of co-solvents. Yet, the OD-H column ranked second in their study and ranked last in our study.
In this study, we have screened a total of 176 commercially available racemic small molecule compounds, including many pharmaceutical compounds, to evaluate the general applicability of SFC for chiral separations. The selected compounds cover a sufficiently wide range of diversity in structure, functional group, and physical property. Under selected conditions, separations for 91.5% of the selected compounds were observed; among those, 83.5% of the compounds were baseline resolved. In addition, the use of a parallel Method Station X5 SFC System expedited the screening process and improved the throughput by five-fold. The results indisputably indicate that SFC is a highly successful chromatographic technique for the chiral separation of small molecule compounds. This, along with the environmental sustainability and cost-effectiveness, should stimulate a wider adoption of SFC for chiral analysis and purification across many industries, including pharmaceutical, agrochemical, and nutraceutical.
720004180, January 2012