
This application note shows how the availability of the HSS Cyano phase in 5-, 3.5-, 2.5-, and 1.7-μm particle sizes facilitates scaling between different instrument platforms (HPLC and UPLC ). Method conditions can be easily scaled for different column configurations using the ACQUITY UPLC Columns Calculator. Scaling methods to utilize the XSelect HSS Cyano XP 2.5 μm Columns maximizes productivity with existing HPLC instrumentation. Additional method transfer to UPLC conditions offers the greatest savings in time and resources.
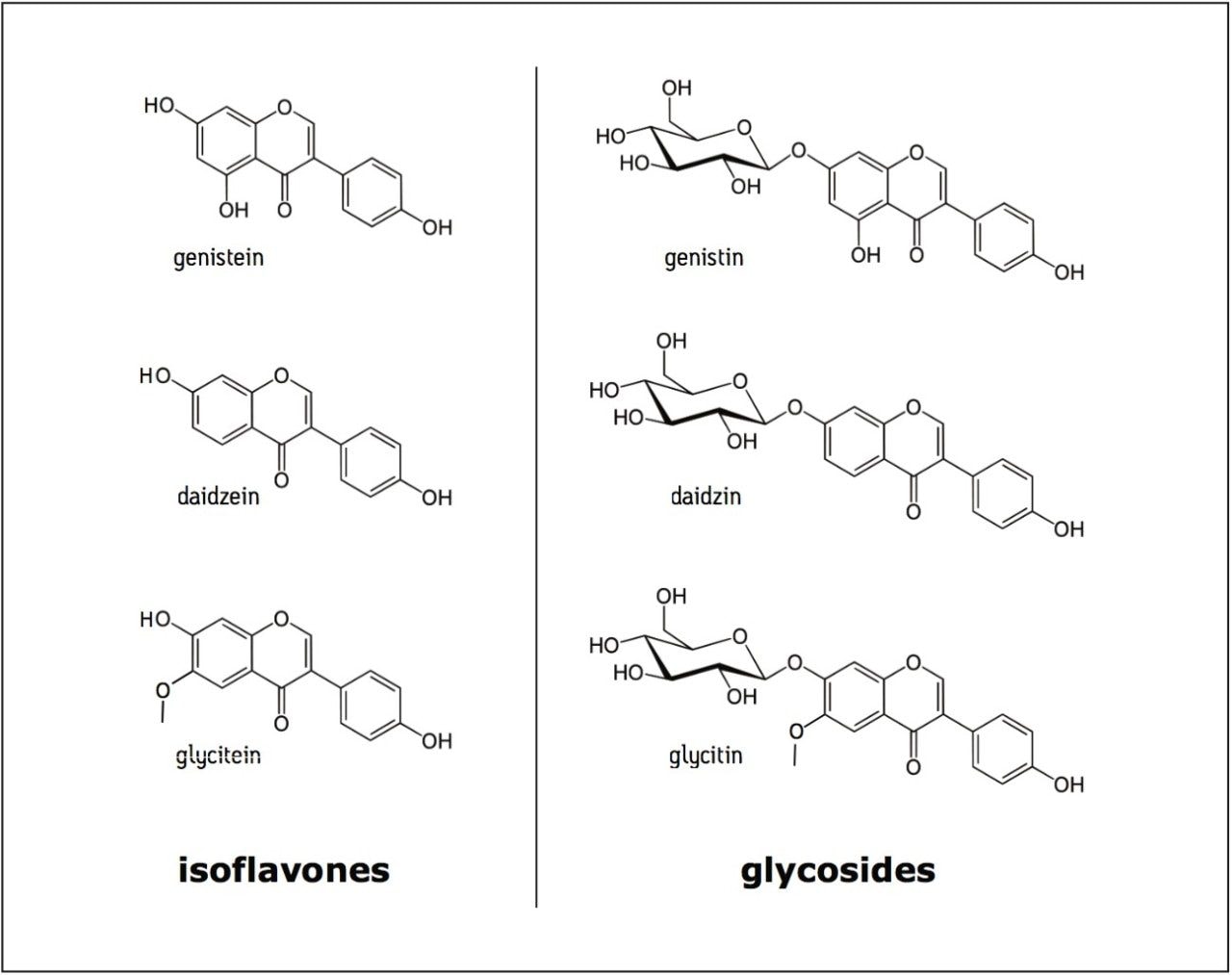
Isoflavones are a class of plant-derived compounds, produced almost exclusively by members of the Fabaceae family, that have been shown to have estrogenic activity in mammals. The major source of isoflavones in the human diet comes from soybeans, in which genistein and daidzein are the predominant components. Isoflavones remain the subject of many scientific studies with accepted methods established by organizations such as AOAC1 and USP2, utilizing traditional reversed-phase C18 columns. Recently, the National Institute of Standards and Technology (NIST) has developed a suite of soy-based candidate Standard Reference Materials (cSRMs) for the certification of soy isoflavones in foods and dietary supplements. For the analysis of those standards, they have developed a method utilizing a 60-minute gradient on a 5 μm cyano column which enables the separation of the soy isoflavones from other components not resolved using the standard C18 methods.3 Using these conditions, they have demonstrated resolution of the three main soy isoflavones and their glycosides (structures shown in Figure 1), as well as the acetyl and malonyl conjugates of the glycosides. Since the conjugates are somewhat unstable in solution, they have added an additional sample preparation step to hydrolyze these conjugates to their associated glycoside, providing a more accurate, reproducible determination of total isoflavone content. Optimization of this method utilizing the XSelect HSS Cyano XP 2.5 μm Column provides a significant reduction in analysis time by traditional HPLC. Further optimization of this method to UPLC with the ACQUITY UPLC HSS Cyano 1.8 μm Column provides additional savings in time, while maintaining the resolution for accurate determination of soy isoflavones.
|
System: |
ACQUITY SQD with PDA detector |
|
Column: |
ACQUITY UPLC HSS Cyano, 2.1 x 50 mm, 1.8 μm, part number 186005986 |
|
Mobile Phase A: |
0.1% formic acid in water |
|
Mobile Phase B: |
0.1% formic acid in acetonitrile |
|
Column Temp.: |
30 °C |
|
Gradient: |
10% (B) for 0.36 min, 10-30% (B) in 3.6 min, hold at 30% (B) for 0.36 min, re-equilibrate at 10% (B) for 1.8 min between injections |
|
Flow Rate: |
0.58 mL/min |
|
Detection: |
UV at 260 nm |
|
Injection Volume: |
3 μL |
|
Strong Needle Wash: |
50/50 acetonitrile/water |
|
Weak Needle Wash: |
10/90 acetonitrile/water |
These UPLC conditions were scaled directly from the 5 μm HPLC method using the ACQUITY UPLC Columns Calculator. The calculator can be used to scale these conditions back to the HPLC conditions, for both the 5 μm and 2.5 μm materials.
|
MS System: |
Waters SQD |
|
Ionization Mode: |
ESI positive |
|
Acquisition Range: |
Single Ion Recording (SIR) |
|
Capillary Voltage: |
3.19 kV |
|
Cone Voltage: |
50 V |
|
Desolvation Gas: |
600 L/hr |
|
Cone Gas: |
0 L/hr |
|
Source Temp.: |
100 °C |
|
Desolvation Temp.: |
350 °C |
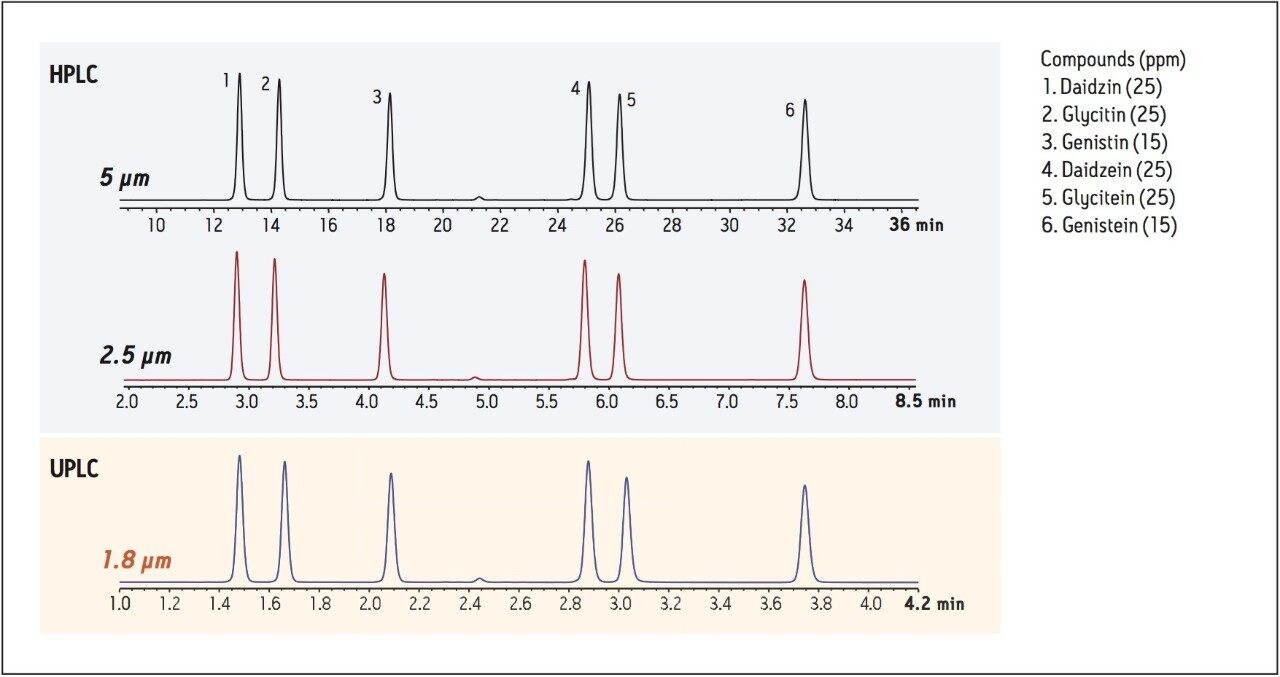
Standard Solution: Prepared from daidzin (25 ppm), glycitin (25 ppm), genistin (15 ppm), daidzein (25 ppm), glycitein (25 ppm), and genistein (15 ppm) using 10/90 acetonitrile/water diluent.
Candidate Standard Reference Materials were obtained from the National Institute of Standards and Technology. Each sample was weighed into 12 ml centrifuge tubes (Table 1). For each tube, 4 mL of 80/20 methanol/water was added followed by sonication for 1 hour. Tubes were centrifuged for 2 minutes at 3000 rpm. A 2 mL aliquot of supernatant was collected from each tube and filtered using a 0.45 μm GHP syringe filter prior to analysis. The remainder of the sample in each tube was hydrolyzed using 150 μL of 2N sodium hydroxide. After mixing for 10 minutes, the solutions were neutralized with 50 μL of glacial acetic acid. The sample was again centrifuged for 5 minutes at 3000 rpm, with the collected supernatant filtered using a 0.45 μm GHP syringe filter prior to analysis.
Based on the method presented by NIST, we developed a method using the soy standard solution with an XSelect HSS Cyano, 4.6 x 150 mm, 5 μm Column, resulting in chromatography with excellent resolution of the soy isoflavones and glycosides. To maximize productivity of the HPLC system, this method was transferred, using the ACQUITY UPLC Columns Calculator, to an XSelect HSS Cyano XP, 4.6 x 75 mm, 2.5 μm Column. This resulted in an HPLC method with a 76% reduction in run time, relative to the 5 μm column. To achieve the greatest benefit, the method was then transferred to UPLC using an ACQUITY UPLC HSS Cyano, 2.1 x 50 mm, 1.8 μm Column. Figure 2 shows the chromatography of the isoflavone standards under HPLC and UPLC conditions. These chromatograms demonstrate the ability to scale between different column configurations by maintaining the same ratio of column length to particle size (L/dp), resulting in similar chromatography but with a significant decrease in analysis time (~88% decrease in run time for the UPLC column relative to the 5 μm HPLC column).
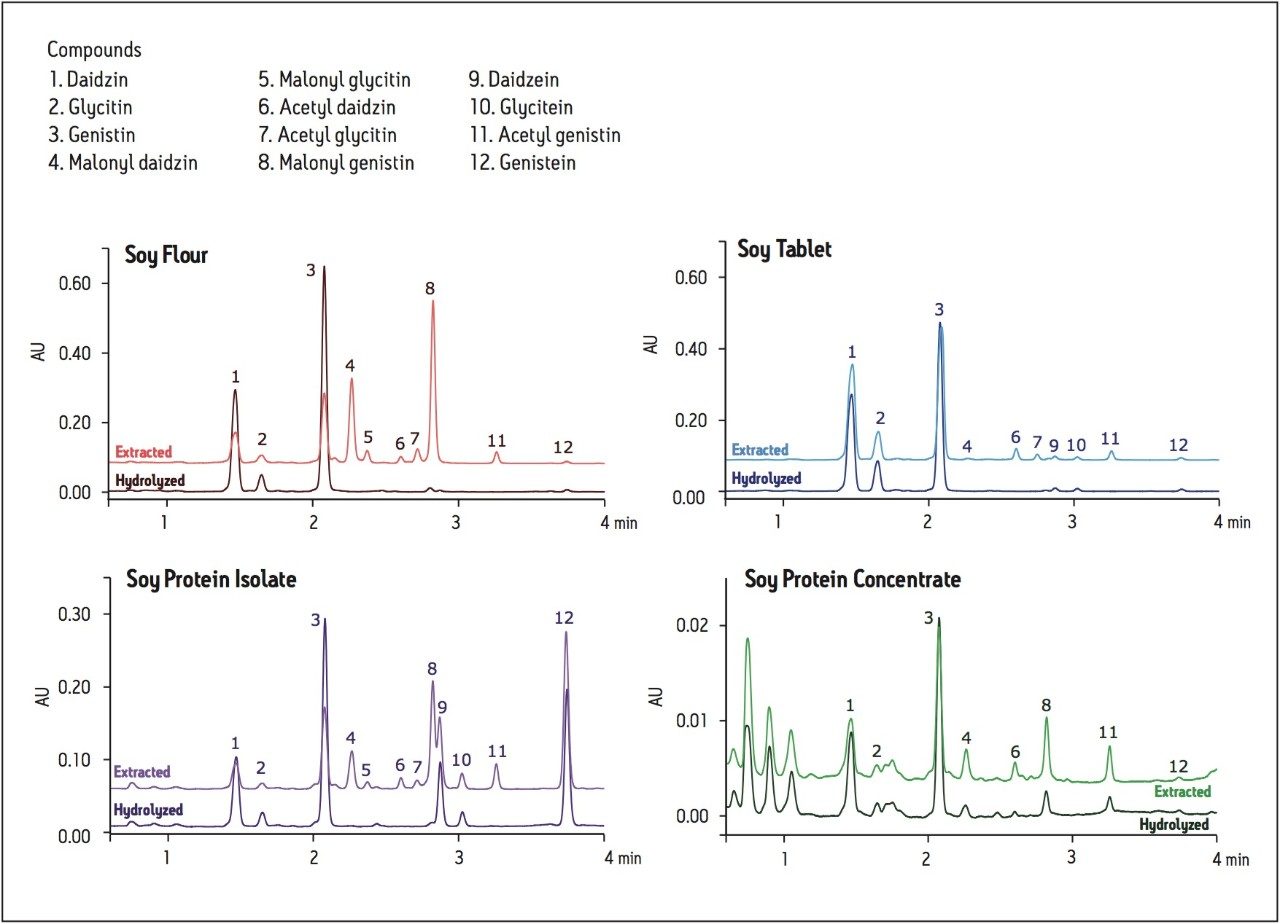
The UPLC method was used to analyze each of the candidate SRM extracts, both before and after hydrolysis of the glycoside conjugates (Figure 3). The identity of the peaks in the UV chromatograms were assigned based on retention times and m/z values from the LC-MS analyses (Soy Flour cSRM example shown in Figure 4). The disappearance of the glycoside conjugate peaks after sample treatment with sodium hydroxide confirms the hydrolysis of these conjugates was successful and complete, which allows for simplified determination of the total isoflavone content.
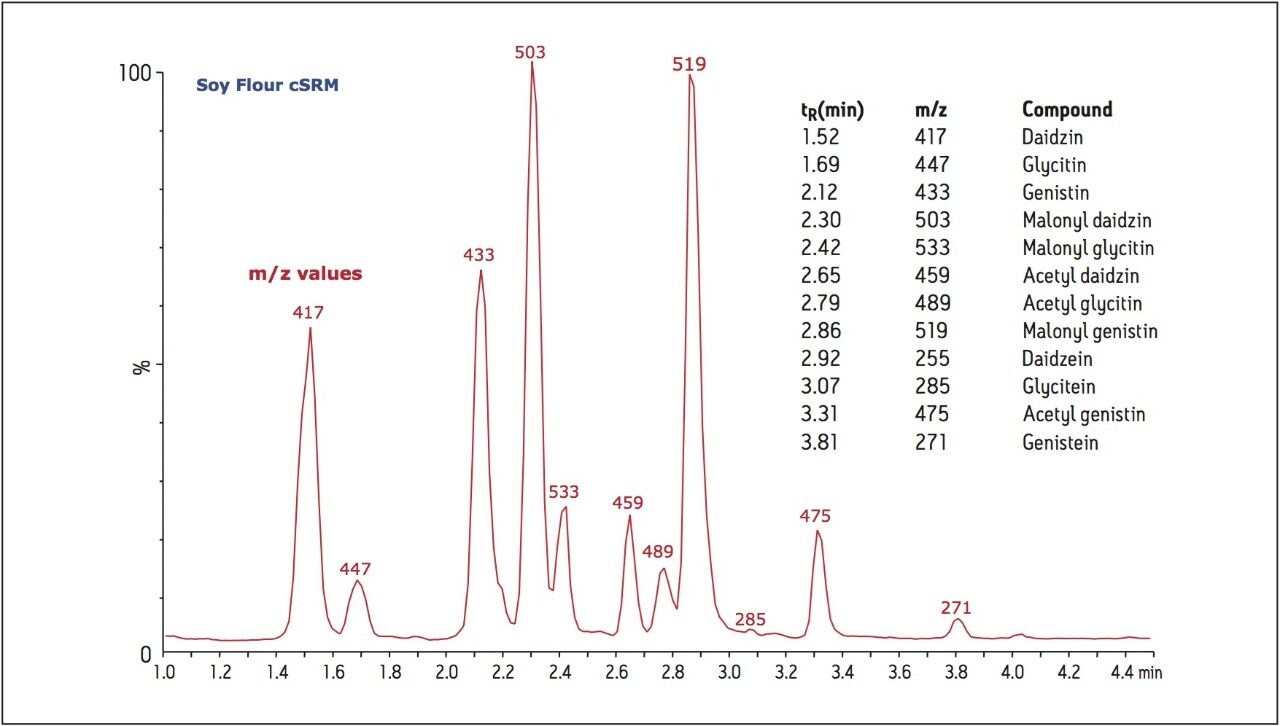
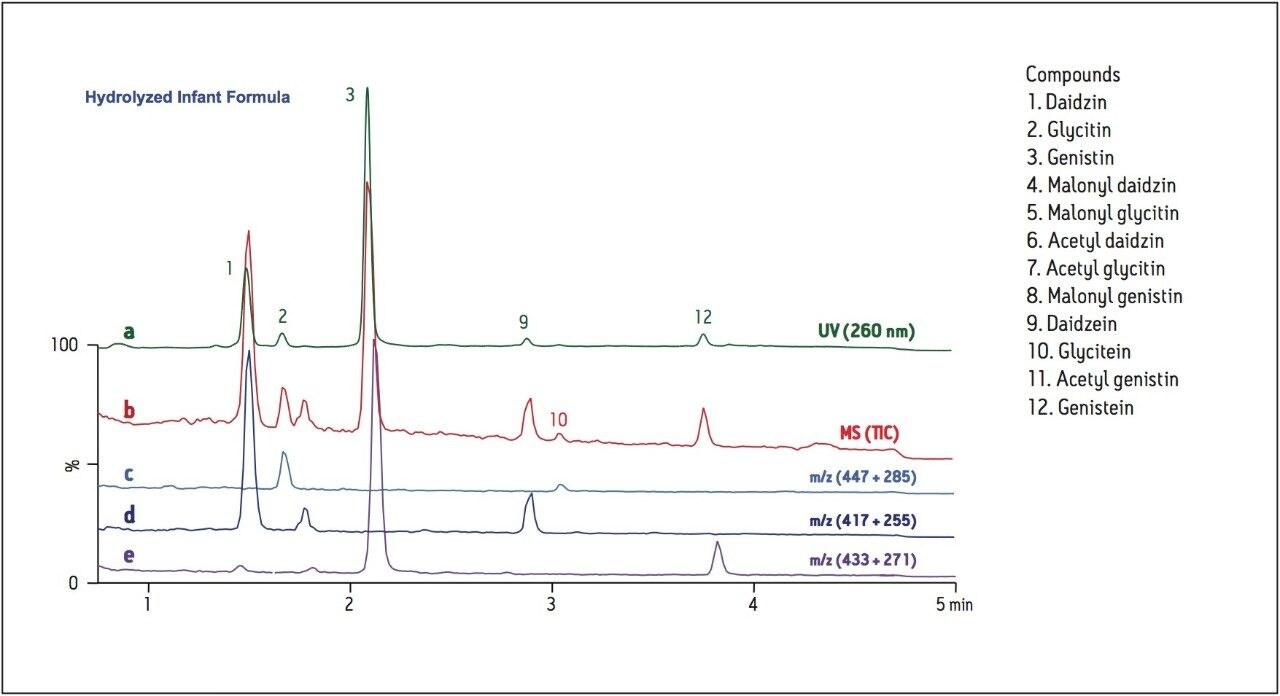
Because of their biological activity and the pervasiveness of soy-based food products, isoflavones have been the focus of many nutritional studies. Some studies advocate the nutritional benefit of isoflavones, asserting that consumption of soy products leads to decreased menopausal symptoms and lowers the risk of certain cancers.4 Other researchers have reached alternate conclusions, with one such study of the isoflavone genistin on mice resulting in significant thymic and immune abnormalities, raising concerns for the use of soy-based infant formulas.5 Because of these concerns, understanding the isoflavone profiles of food products is of critical interest. Using the current method, a sample of commercially-available soy-based infant formula was prepared and analyzed by both LC/UV and LC-MS. Analysis of the extracted sample revealed, in addition to the isoflavones and their glycosides, only a minor concentration of the malonyl genistin conjugate. After hydrolysis, all evidence of the conjugate is gone, revealing only the isoflavones and their glycosides (Figure 5). Using this method, the concentrations of soy isoflavones can be evaluated and monitored, facilitating fundamental research, as well as the quality control of food and nutraceutical consumer products.
Analysis of soy isoflavones using the HSS Cyano stationary phase offers complimentary selectivity to traditional C18 based methods. Hydrolysis of the glycoside conjugates simplifies the resulting chromatography and the determination of total isoflavone content of a sample. The availability of the HSS Cyano phase in 5-, 3.5-, 2.5-, and 1.7-μm particle sizes facilitates scaling between different instrument platforms (HPLC and UPLC ). Method conditions can be easily scaled for different column configurations using the ACQUITY UPLC Columns Calculator. Scaling methods to utilize the XSelect HSS Cyano XP 2.5 μm Columns maximizes productivity with existing HPLC instrumentation. Additional method transfer to UPLC conditions offers the greatest savings in time and resources.
720004253, August 2012