This work demonstrates the benefits of combining a highly specific solid phase extraction methodology, UPLCchromatography, and high-sensitivity tandem quadrupole MS for the development of a high-sensitivity method for the quantification of estradiol in plasma that also produces high selectivity and high throughput.
Demonstrates the benefits of combining a highly specific solid phase extraction methodology, UPLC chromatography, and high-sensitivity tandem quadrupole MS.
Estradiol is a form of estrogen, a female sex hormone produced by the ovaries, Figure 1. Estradiol is used to treat symptoms of menopause such as hot flashes and vaginal dryness, burning, and irritation. Other uses include prevention of osteoporosis in postmenopausal women, and replacement of estrogen in women with ovarian failure or other conditions that cause a lack of natural estrogen in the body. Estradiol is sometimes used as part of cancer treatment in women and men. Estradiol is also present in males, being produced as an active metabolic product of testosterone.
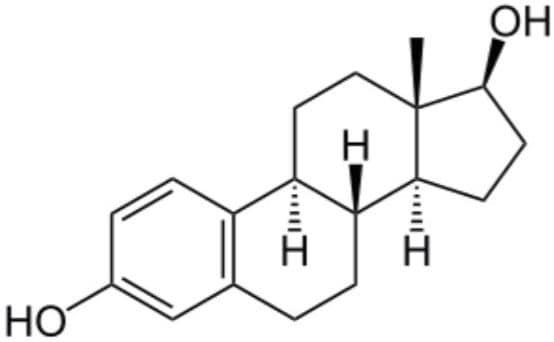
Estradiol is typically administered orally, via tablet or capsule, with dosages ranging from 0.5 to 2 mg. Estradiol is heavily protein bound (98%) mainly to sex hormone-binding globulin, but also to albumin with only 0.04% being biologically active.
The metabolism distribution of exogenous estrogens is similar to that of endogenous estrogens. The major site of metabolism is in the liver where estradiol undergoes reversible metabolism to esterone. Both estradiol and esterone can be converted to estriol, which is the primary metabolite being eliminated in the urine. Estradiol is also metabolized via sulphate and glucuronide conjugation, which in turn undergoes enterohepatic recirculation in the liver. These metabolic conjugates are subject to biliary secretion into the intestine where they undergo gut hydrolysis and reabsorbtion thus increasing the exposure of the drug.
The half life of estradiol in health females is 13hrs. The high protein binding and extensive metabolism results in extremely low circulating levels in the 14 to 50 pg/mL range. Thus in order to determine the pharmacokinetics of estradiol, a high-sensitivity method is required.
The samples were isolated using solid phase extraction employing a Waters Oasis MCX (1 mL, 30 mg) cartridge. A 500 µL aliquot of plasma was diluted with an aqueous solution and loaded onto the SPE cartridge previously conditioned with organic solvent and water. The plasma solution was then washed with an organo-aqueous solution followed by water then eluted in solvent. The eluted samples was evaporated to dryness and reconstituted in buffered solvent then derivatized with dansyl chloride, heated to 60 °C, followed by analysis by LC-MS/MS. The extracted samples were analyzed by reversed-phase isocratic chromatography employing an acidic aqueous buffer and acetonitrile as the organic modifier.
|
LC system: |
ACQUITY UPLC System equipped with a Binary Solvent Manager, Column Manager, and Sample Manager. |
|
LC column: |
ACQUITY UPLC HSS T3 C18 1.8-μm, 2.1 x 100 mm |
|
Elution: |
85% organic solvent 15% acidic aqueous buffer over 6 minutes followed by a high concentration organic wash. |
|
Column temp.: |
45 °C. |
|
MS system: |
Xevo TQ-S |
|
MS mode: |
Positive ion electrospray MS/MS |
|
MS transition : |
506 ⇒ 171 |
The chromatographic method obtained using ACQUITY UPLC and an ACQUITY UPLC HSS 1.8-µm column provided excellent resolution for the estradiol analyte from the endogenous components in the samples. The estradiol compound eluted at 3.3 minutes with a peak width of 12 seconds at the base. The data shown below (Figure 2) illustrates the signal obtained from the lower limit of quantification (LLOQ) for estradiol in plasma.
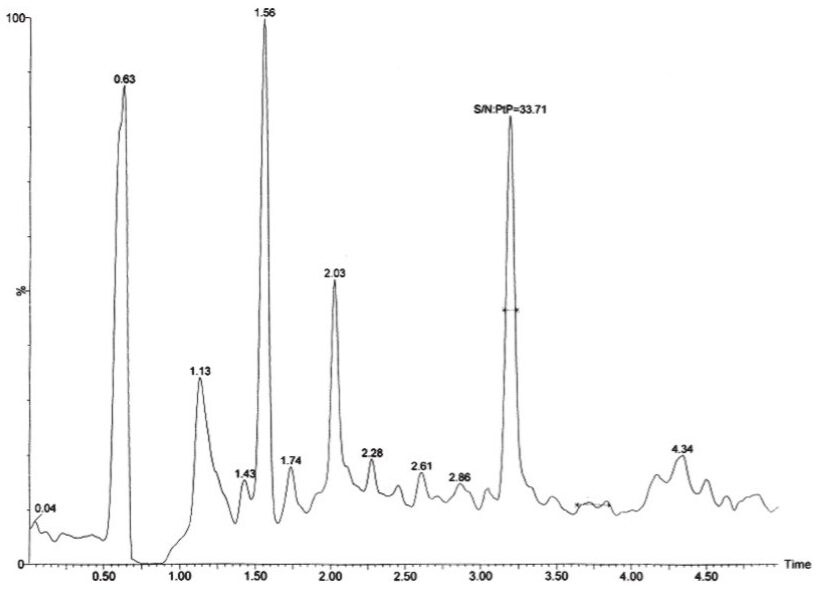
The Xevo TQ-S mass spectrometer is equipped with a novel StepWave ion guide that enables improved ion sampling in the source and also better ion transfer efficiency. This ion guide optics, when combined with the high-resolution chromatography produced by the ACQUITY UPLC System, results in a LLOQ of 2 pg/mL. In this example, we can see that the 2 pg/mL standard extracted from plasma results in an analyte peak with a signal-to-noise response of 33:1.
The high sensitivity achieved for this assay was due to the combination of selective extraction, high-resolution chromatography provided by ACQUITY UPLC, and the sensitivity of the Xevo TQ-S. The new Q0 region in the Xevo TQ-S fitted with the unique StepWave Technology allows a significantly greater amount of the ion flux to be sampled without contaminating the instrument source region.
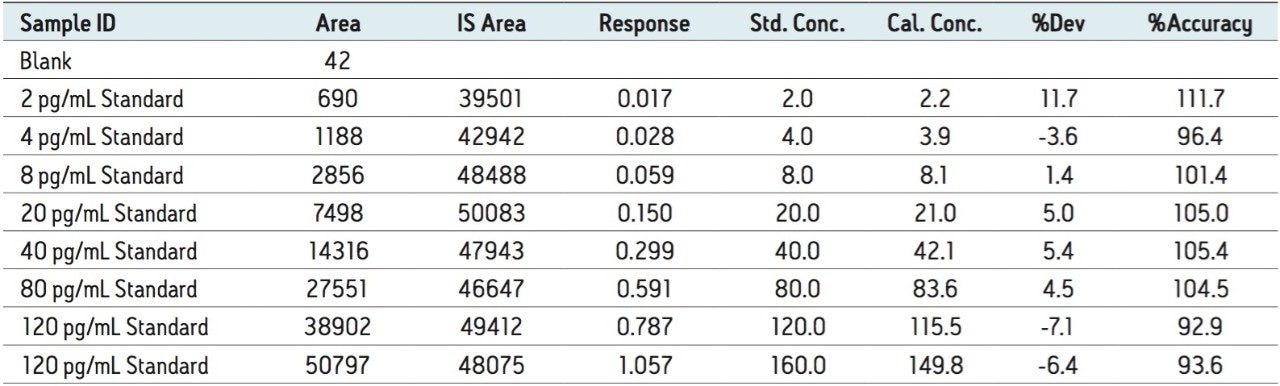
The assay showed linear calibration response over the range of 2 to 160 pg/mL with an r2 value of 0.992, with the back-calculated concentration of the standard being within ± 12% of the nominal concentration (Table 1). Despite the need for high sensitivity and specificity, this assay was performed within a 6-minute inject-to-inject time scale allowing for high throughput analysis with high precision.
The lower and upper limits of serum estradiol in an adult male ranges between 50 and 200 pmol/L and between 70 and 500 pmol/L in an adult female. Therefore, the ability to detect estradiol in human plasma in very low concentration is extremely important. The combined solution offered by sample preparation chemistry, ACQUITY UPLC, and Xevo TQ-S enable us to readily achieve the LLOQ level of estradiol at 2 pg/mL.
Estradiol is a low-dose, low-exposure compound that is rapidly cleared by metabolism in the liver, resulting in circulating levels in the range of 10 to 60 pg/mL. Hence a high-sensitivity assay is required to determine the pharmacokinetics of the compounds. The application of a highly specific solid phase extraction methodology using Oasis MCX, a high-resolution UPLC separation employing ACQUITY UPLC, and the intrinsic sensitivity of the Xevo TQ-S allows for the development of a high-sensitivity method for the quantification of estradiol in plasma. The method delivered a LLOQ of 2 pg/mL with excellent precision and robustness.
720004057, August 2011