This application brief provides an in-depth mass spectral analysis of complex samples with direct sample introduction utilizing Atmospheric Solids Analysis Probe (ASAP) and High Definition Mass Spectrometry (HDMS).
ASAP-IMS-MS shows the potential to fingerprint crude oil samples, and offers a route to the analysis of involatiles, which cannot be achieved using GC-MS.
The chemical characterization of complex mixtures like crude oil remains an extremely challenging problem. In the case of crude oils, the rarefaction of these natural resources results in the use of heavier products, which need to be characterized. Techniques like FTMS or 2D-GC-MS are commonly used, but there are limitations in the MS separation of isomers. In the case of GC, chromatography is limited to low volatility compounds. The separation of isomers using ion mobility has been explored for some time using experimental instruments, but is now available commercially. Most of the initial applications have been published in the domain of natural polymers, such as proteins. However, other studies have been conducted using ion mobility on experimental instruments in the domain of crude oil1. In this technology brief we explore the potential of a commercial instrument, the Waters SYNAPT G2 HDMS, for the characterization of industrial products, including crude oil.
Direct sample introduction of a crude oil sample and ionization was performed using the ASAP technique with the SYNAPT G2 HDMS Mass Spectrometer. The conventional Tof-MS spectrum obtained is extremely complex, as shown in Figure 1.
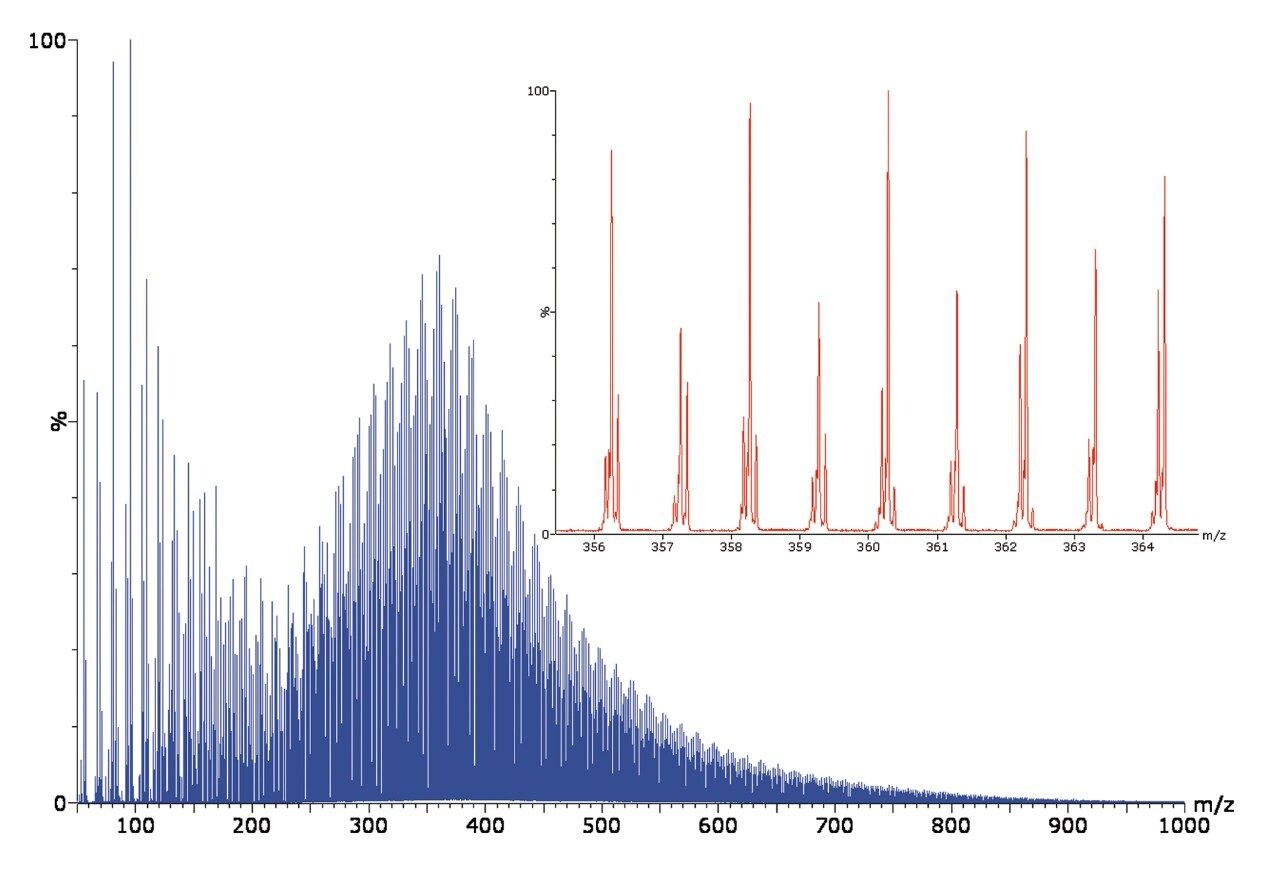
When using ion mobility separation, bands separated by 14 Da (CH2) are visualized, as shown in Figure 2A. By selecting the bands shown in Figure 2B, it is possible to extract the ion mobility mass spectrum and export it into MassLynx Software for further interpretation, as shown in Figure 3.
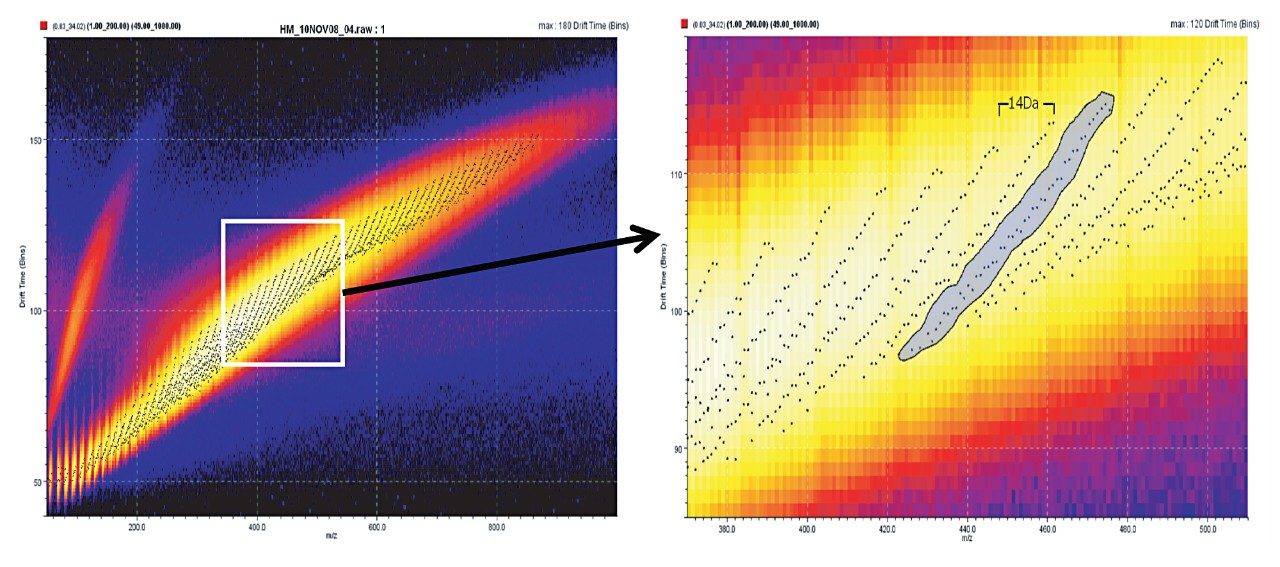
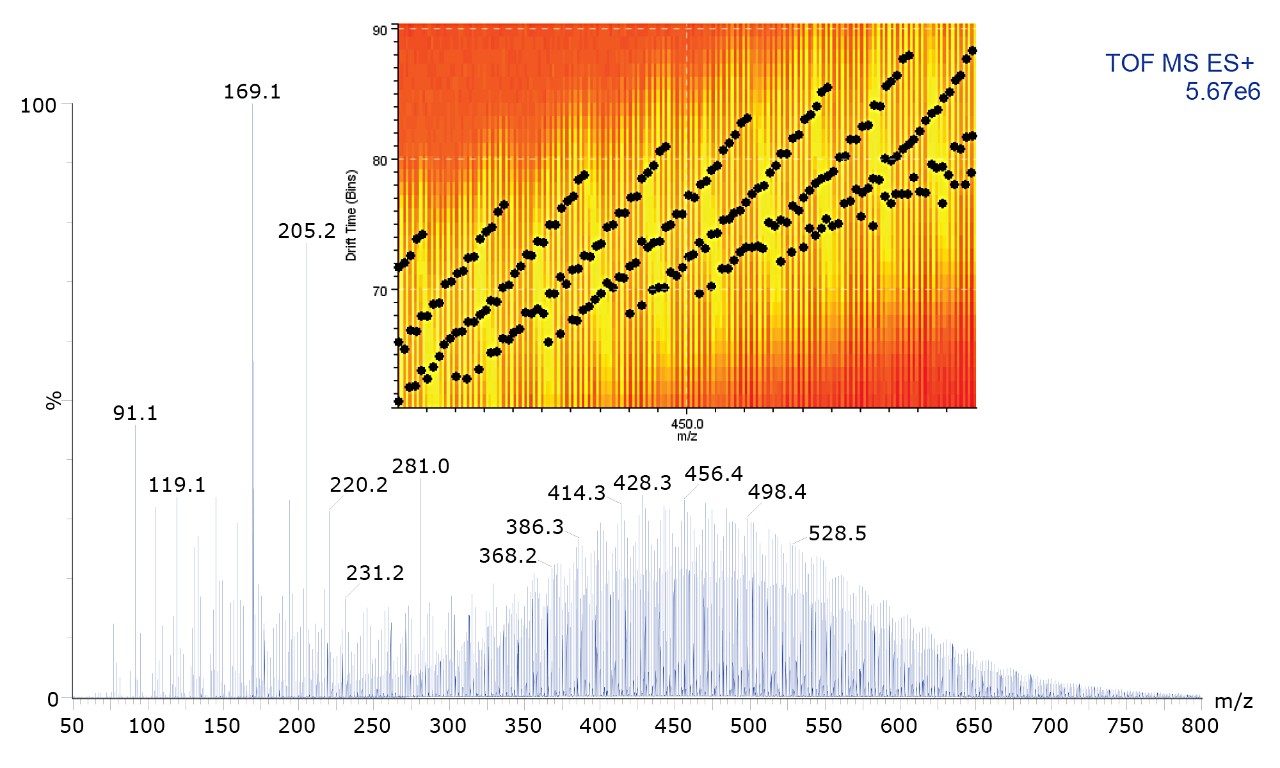
Ion mobility separation (IMS) combined with direct ionization using the ASAP has previously been illustrated.2 The orthogonality of IMS acts as an enabling technology where crude oil sample analysis can be performed with no prior chromatographic separation or sample preparation. The combination of ASAP-IMS-MS shows the potential for this technique to fingerprint crude oil samples, and offers a route to the analysis of involatiles, which cannot be achieved using GC-MS. Useful information was readily extracted from complex data using DriftScope Mobility Environment Software v.2.1.
720004023, September 2011