This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the excellent carryover performance of the ACQUITY UPLC H-Class System with compounds that are generally difficult to eliminate from a chromatographic system
Whether analyzing in the mg/mL or ng/mL range, sample carryover is easily managed with the ACQUITY UPLC H-Class System. The flow-through-needle injector design and single wash solvent configuration simplify method optimization, even for difficult-to-eliminate analytes.
For methods requiring quantification across multiple orders of magnitude, it is important to have a sample delivery design that can accurately, cleanly, and reproducibly deliver sample to the column. One of the contributors to unsatisfactory chromatographic peak area precision is sample carryover from previous injections. The injector design must therefore minimize the susceptibility to carryover. Severe sample carryover can lead to system contamination, which requires the entire system to be taken off-line for cleaning or even replacement of the contaminated surfaces. Managing carryover becomes even more challenging as detection techniques become more sensitive. Often carryover is not a linear relationship to the mass injected on column as it approaches low or sub-ng levels. This can severely limit the range of quantitation for many applications. The simpler the injector design and the better the carryover management, the easier it is for users to optimize their method.
The ACQUITY UPLC H-Class System was designed to be flexible for routine analysis while providing outstanding performance for both UPLC and HPLC applications. The sample delivery system is based on a flow-through-needle design (SM-FTN). With this type of injector, the interior of the needle is washed by the mobile phase (gradient) during the analysis. The SM-FTN uses a single solvent to clean the exterior of the needle in the injection port and was designed so that this wash solvent does not come in contact with the sample or mobile phase. Washing the needle and the seal together at the sealing surface reduces the chance for contamination. The duration of the wash is programmable in the method and can be configured for both pre-injection and post-injection wash. Selection of wash solvent composition is sample dependent and should easily solubilize the analyte.
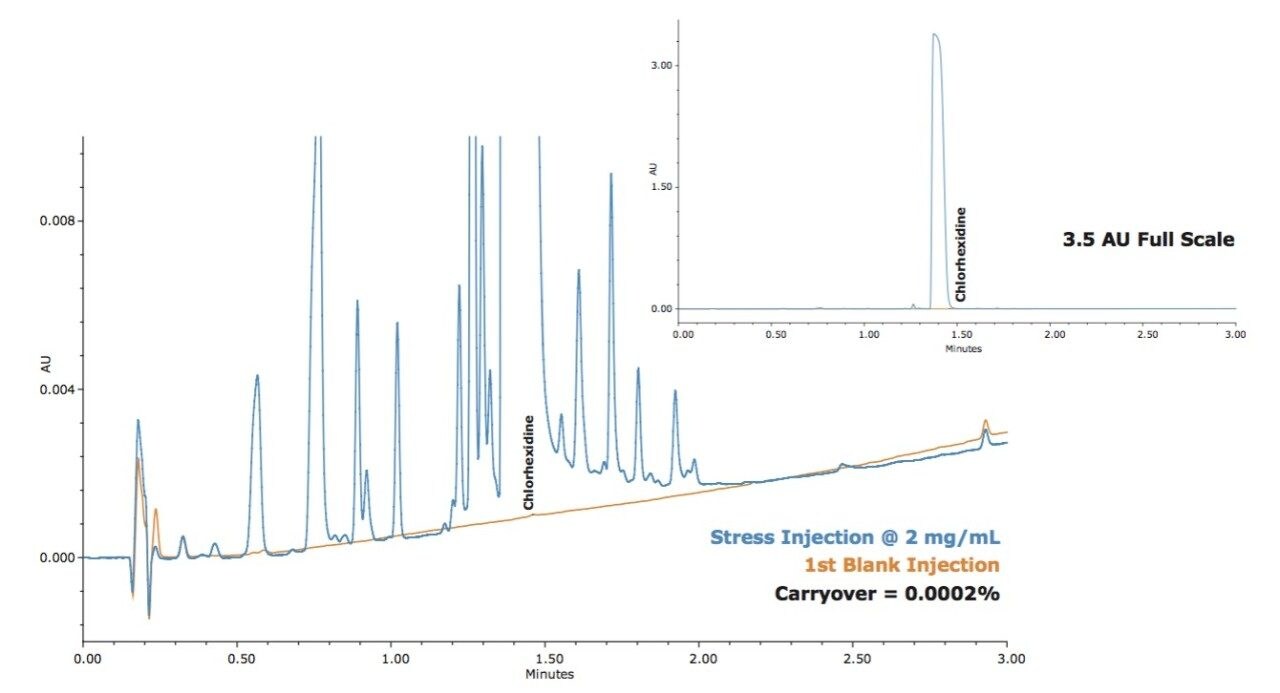
The performance of this carryover management design is demonstrated by assessing carryover with chlorhexidine, a typically sticky compound that can be difficult to remove from an injector or system. Figure 1 shows the carryover observed for the first blank injection after injecting a sample at 2 mg/mL. Even with this difficult-to-remove compound, the ACQUITY UPLC H-Class reduces carryover to below 0.004% measured by gradient analysis (the most challenging way to assess system carryover).
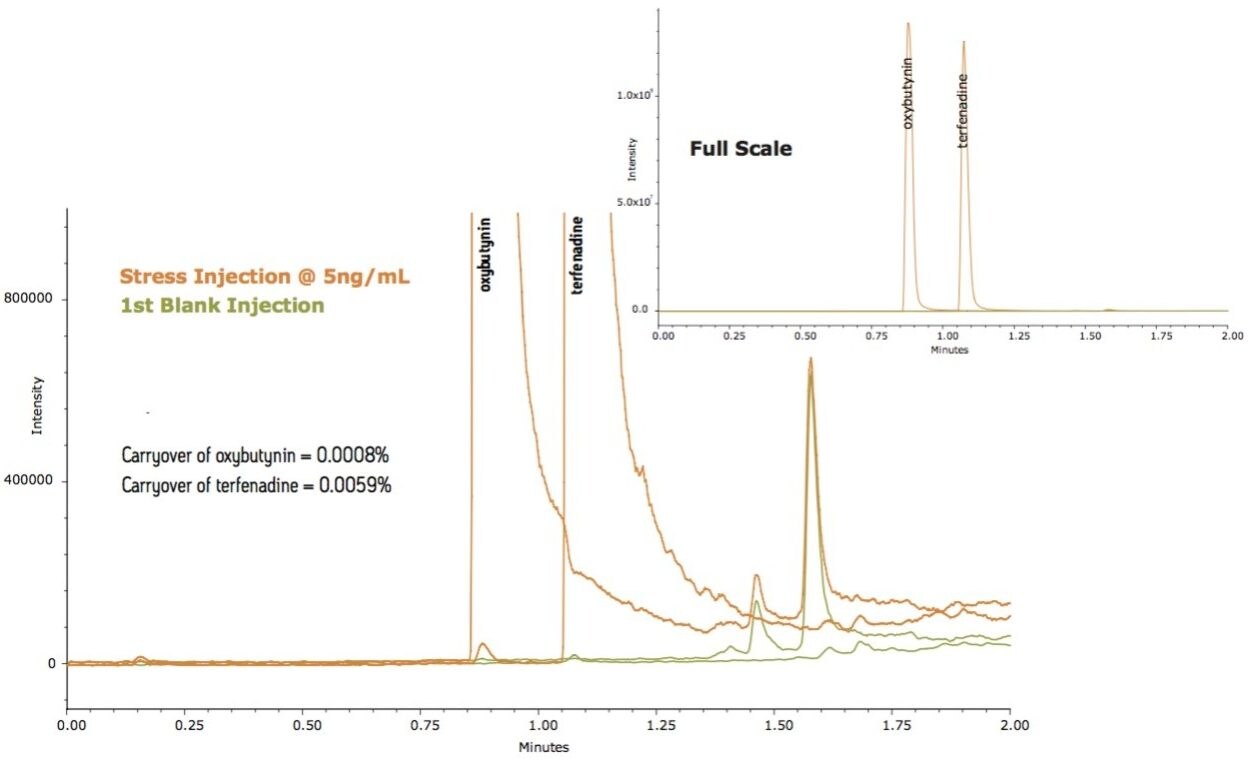
To test carryover for applications that require a more sensitive detection technique such as MS, a sample containing oxybutynin and terfenadine was injected at 5 ng/mL (Figure 2). The carryover measured using a TQ Detector was very low, below 0.001% for oxybutynin and below 0.006% for terfenadine (a sticky analyte). Managing carryover to these levels demonstrates a system solution that is capable of delivering low-level quantification across multiple orders of magnitude.
Dealing with sample carryover is a fact of life in the separation sciences. This is especially true when considering the high sensitivity, high efficiency analyses required by UPLC separations. The ACQUITY UPLC H-Class System easily manages carryover for samples that are typically difficult to remove, whether injecting high concentrations for UV-related compound assays or creating wide calibration ranges at sub-ng levels with MS detection for bioanalysis.
720003616, June 2010