
This is an Application Brief and does not contain a detailed Experimental section.
This application brief illustrates how a sample is efficiently taken through a three-step purification process utilizing the AutoPurify capabilities within the Waters FractionLynx Application Manager for MassLynx Software, and the AutoPurification System for MS-directed analysis.
A standard requirement for drug discovery screening of synthetic libraries is that the test compounds must have a minimum purity. Purity is based on the area percent of an LC chromatogram from a detector such as UV, evaporative light scattering (ELS), MS with a total ion chromatogram (TIC), or a combination of multiple detectors. If the screening compounds do not meet this standard, purification is required. Managing the flow of samples, subsequent fractions, and all the associated data through this process can often be difficult and time consuming.
This application brief illustrates how a sample is efficiently taken through a three-step purification process utilizing the AutoPurify capabilities within the Waters FractionLynx Application Manager for MassLynx Software, and the AutoPurification System for MS-directed analysis. This comprehensive informatics solution enables automation from the initial evaluation, through the purification, to analysis of the collected fraction.
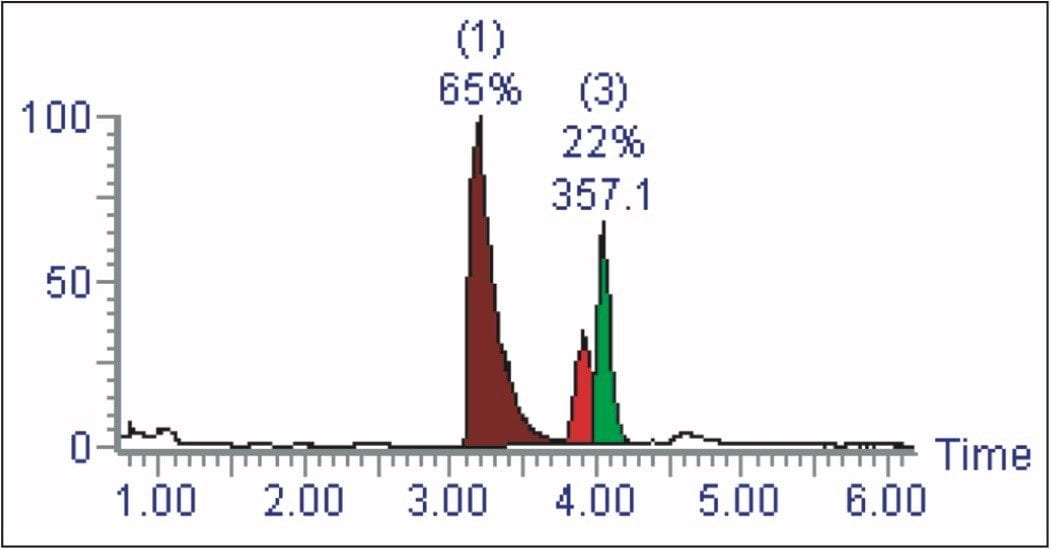
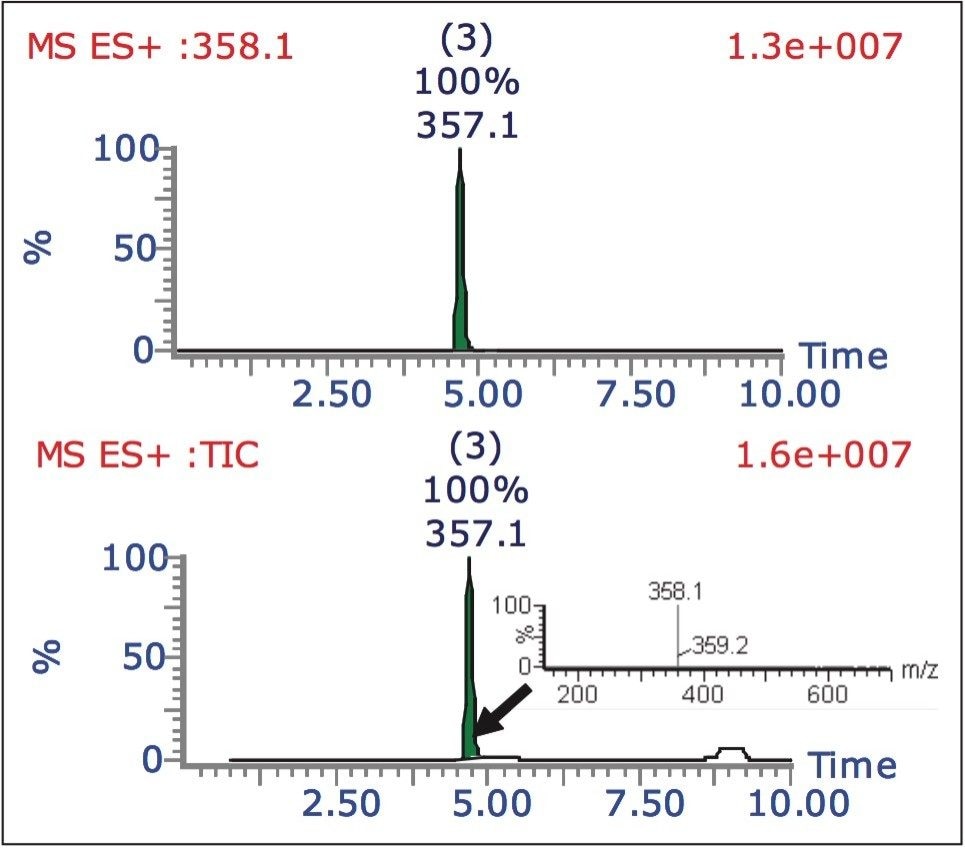
The AutoPurify functionality uses the results of the analytical analysis to determine the purification process. By performing an analytical evaluation of the sample, the presence of the target compound is confirmed and its purity measured (Figure 1).
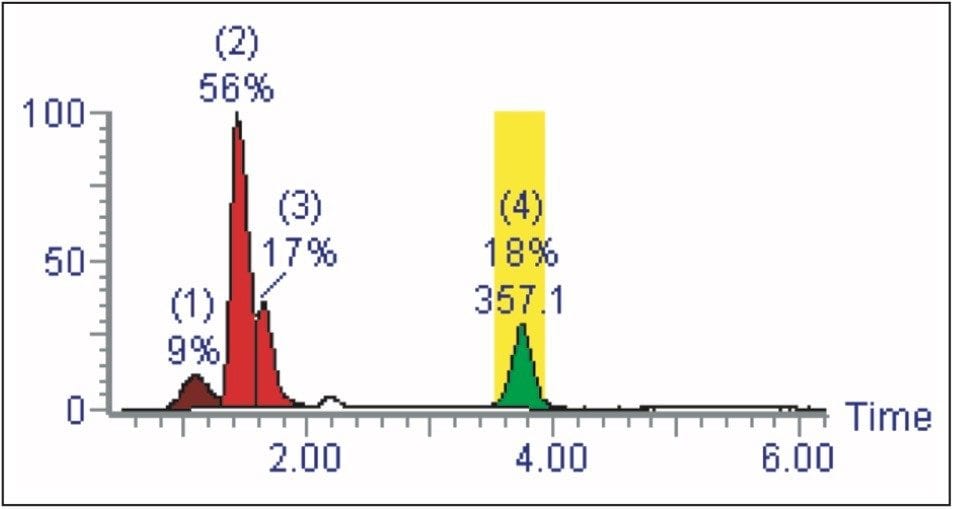
The software will decide which shallow gradient should be used to perform the purification (Figure 2).
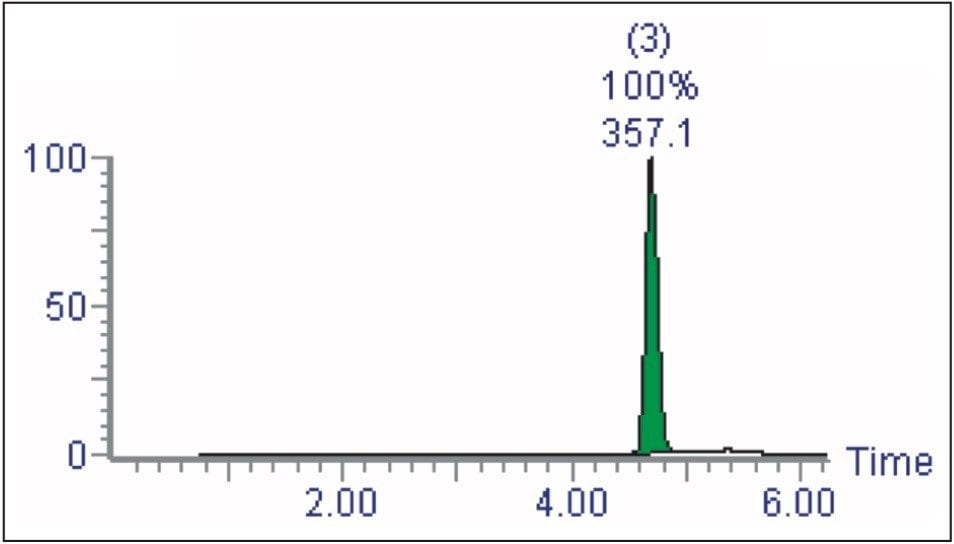
Then, it automatically performs analysis of the collected fractions (Figure 3).
Information determined from analysis of the fractions can be used to help with post-purification handling such as fraction pooling and transfer to an evaporator. A report can be exported in different file formats such as .xml, .csv, and .tab, to easily interface with other sample handling software packages.
In the first of the three-step process, the purity of the target mass is identified by integrating the chromatogram. In the example shown in Figure 4, the area percent of the target determined from the TIC (22%) is then used to calculate the sample purity.
The area percent can also be determined by total absorbance current, wavelength, or analog signal. The purity of the target is then classified as “pass,” “tentative,” or “fail,” based on user-defined limits. In this example, less than 10% pure means purification will not occur, 10% to 80% purity requires purification, greater than 80% is pure enough, and does not require further purification.
In a manual process, the analyst would evaluate the separation, and adjust the gradient to achieve the best results. However, in an open access environment or where large numbers of samples are being handled, automation is necessary.
In the second step of the process, purification occurs. The software will determine the purification method best suited to improving the separation by choosing one of six different shallow gradients. Using the analytical retention time of the target, the appropriate shallow gradient-based method will be chosen.
Shallow gradients, also referred to as narrow gradients, allow for optimal target separation from closely eluting impurities, thus improving the purity of the resulting fraction. Each narrow gradient, whose time window is indicated by the colored lines (Figure 5), is created to cover a different timed section of the analytical gradient.
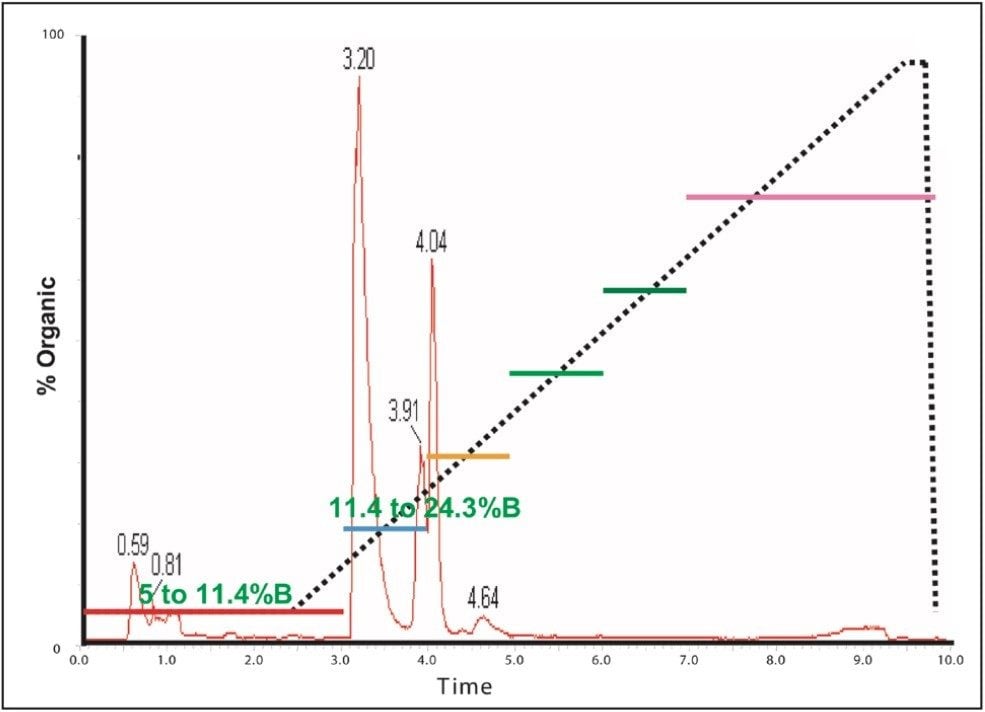
The analytical gradient is indicated by the dotted black line, and shows the solvent change over the course of the gradient to be from 5% to 95% B. With the relationship between the analytical retention time and the elution organic composition known, the software can choose which of the narrow gradients will be used to automatically purify the samples during the purification stage of the process.
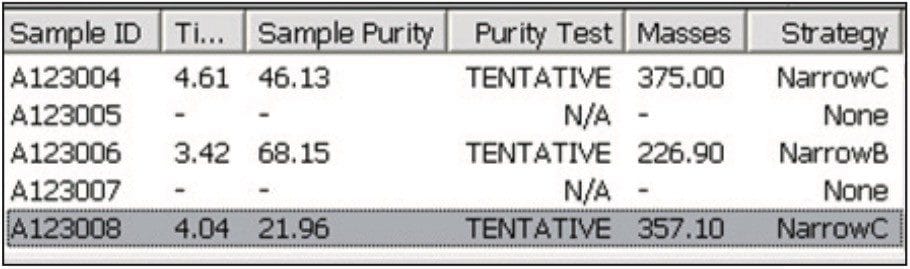
When the software evaluates the analytical sample, it creates a browser report defining the recommended strategy. The user has the opportunity to change the strategy if necessary. The part of the report that refers to the strategy is the results pane (Figure 6). In this example, there are several other samples analyzed, but the one that is of interest is that last one on the list, A123008.
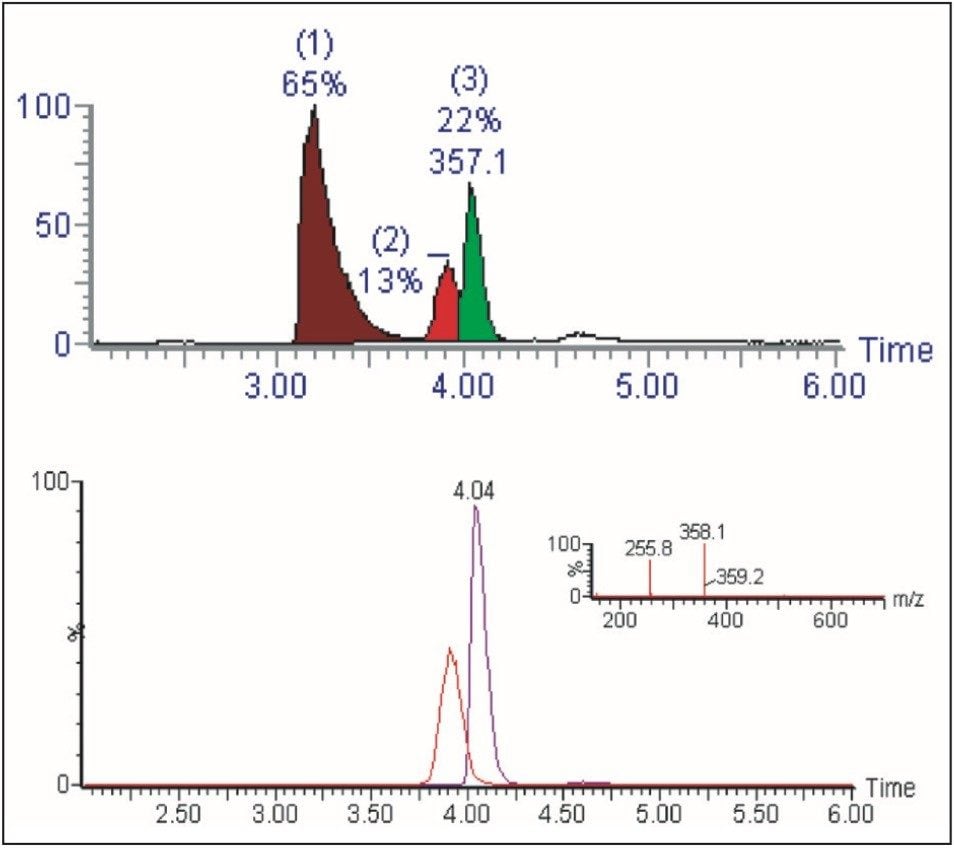
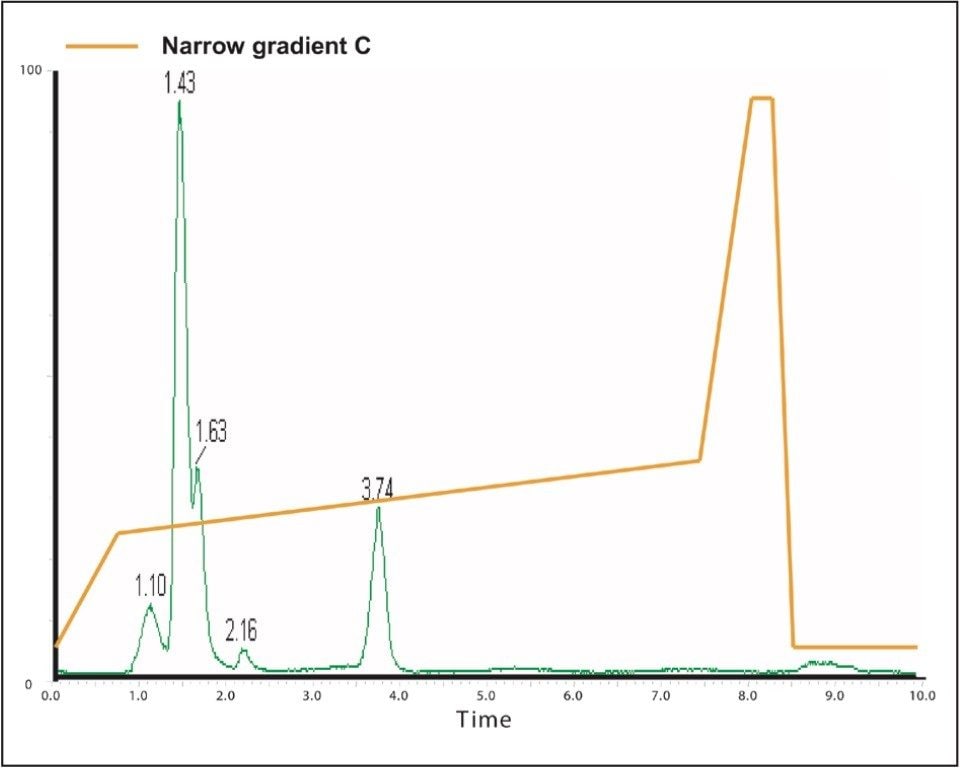
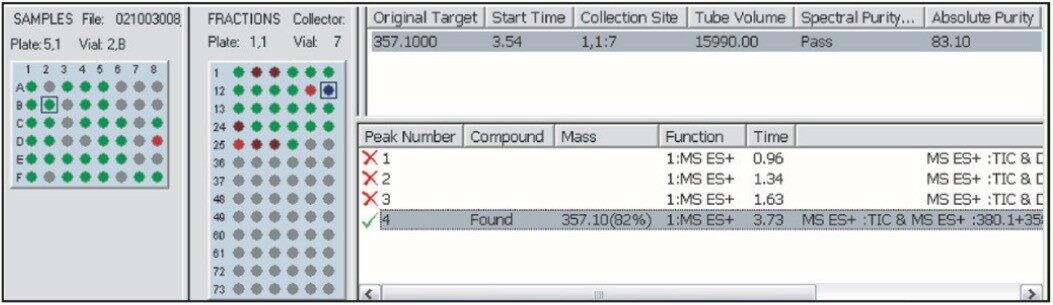
The sample in this case eluted at 4.04 min (Figure 7), so the narrow gradient chosen for the purification was “Narrow Gradient C,” the one that targeted the solvent change that occurred between 4 and 5 minutes. This gradient is denoted by the green line, which changes from 24% to 37% organic over 6.5 min, and is defined graphically as below.
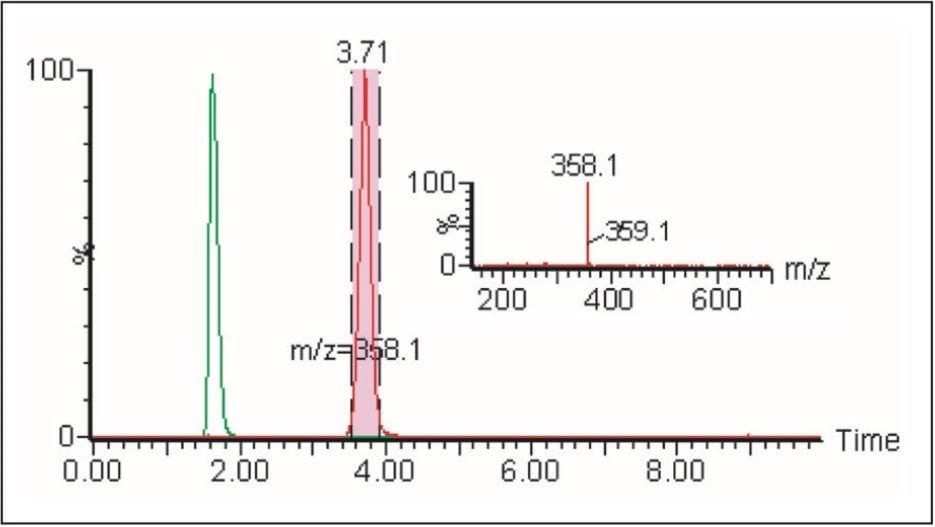
The improved separation is more clearly displayed when the chromatograms of the two co-eluting compounds, as seen in Figure 4, are extracted and their chromatograms reviewed. Figure 8 shows the two chromatograms of masses 255 and 358, overlaid, and the improved separation achieved.
With the first two steps of the process complete, the user can also decide to analyze the fractions (Figure 9). AutoPurify creates a sample list containing the fractions required for analysis and automatically runs them.
To ensure that the portion of the sample taken for analytical analysis is representative of the entire collected fraction, it may be necessary to pre-mix fractions prior to injection (done with the injector/collector). Once homogenized, analysis can be performed on an analytical scale.
Automation of the three-step purification process is accomplished through AutoPurify.
A FractionLynx browser is created after each of the three stages to display results of the analysis and to report the recommended strategy for the next stage in the process. The software can automatically create and run the list of samples that are to continue to the next step. The user has a choice whether to allow the three stages to run unattended, or to manually review the results of each stage and edit the software’s decision.
The determined strategy can be adjusted as necessary by the user through the interactive browsers that are produced. By automating the process, decisions can be made after regular work hours, allowing the work to continue unattended, saving time and resources.
The root name of the data, the sample ID, sample list, and the FractionLynx browser, A123, as shown in Figure 10, are edited by the software and carried through the purification process to make sample and results tracking easier.
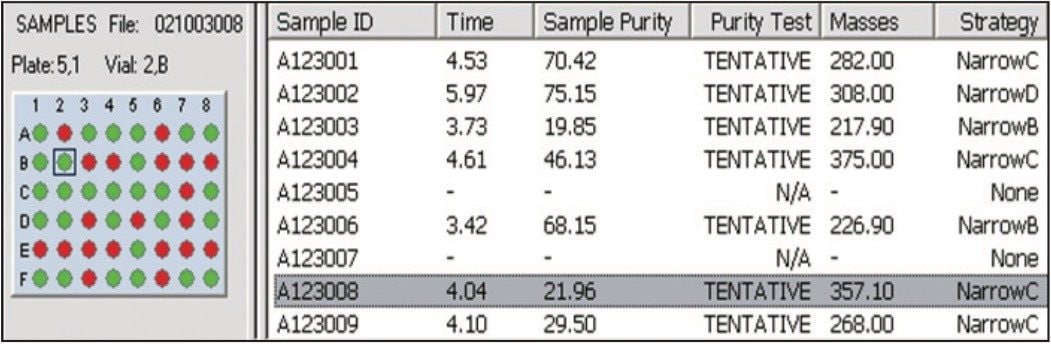
FractionLynx browsers also include chromatograms and spectral information that are not shown in this application brief. The portion of the browser file in Figure 10 shows sample purity and the prep strategy decision that was determined after the samples were analyzed on an analytical scale.
The preparative sample list is automatically created and run after the analytical analysis. Once the purifications are complete, the results are processed and a new FractionLynx browser report is generated (Figure 11).
Upon completion of the processing of the purification results, a sample list is generated and automatic analysis of the fractions generated is performed (Figure 12).
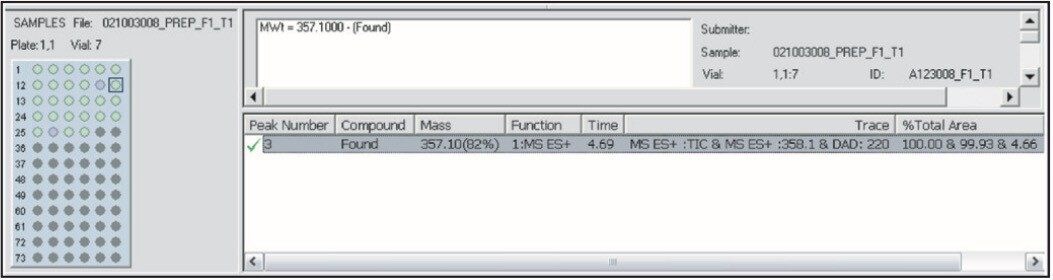
The final report shows the locations of the fractions, chromatograms, and spectra. The information in the reports can then be easily exported in different file formats such as .xml, .csv, and .tab, to easily interface with sample handling software packages such as liquid handlers or weighing devices.
This application brief shows how a library of compounds can easily and efficiently be purified using the AutoPurify capabilities within the FractionLynx Application Manager. The software is capable of automating the entire purification process, from the original analytical purity assessment, to purification, and finally to the analysis of the fractions.
AutoPurify allows the process to be performed intelligently. Analytical results are used to determine if the target is present and its purity. Based on these criteria, only samples that truly require purification continue on through the process. Samples that do not contain the target compound, not enough of the target, or are already pure enough can simply be excluded from purification.
The benefits of using AutoPurify can be measured in time savings, reduced solvent consumption, and overall productivity gains. This is noticeable in several main areas:
720001466, June 2007