mAbs Testing in Inflammation Using Mass Spectrometry – Combination of mAbXmise™ Kits and XEVO™ TQ Absolute XR Mass Spectrometer
Davor Fielitza, Dorothée Lebertb
a Waters Corporation, Eschborn, Germany
b Promise Proteomics, Grenoble, France
Published on February 18, 2026
Disclaimer:
The data presented in this application note combines the use of two kits dedicated to the preparation of samples and the use of liquid chromatography and mass spectrometry instrumentation to perform quantitative analyses. The mAbXmise kits described have not been market-authorized by any regulatory entity for diagnostic purposes outside of Europe. Furthermore, the Waters Xevo TQ Absolute XR Mass Spectrometer has not been market-authorized by any regulatory entity. Per the IFUs for the ITDM2 and IADA1 kits, analytical performance depends on the instrument characteristics and its settings. The analytical method shall be validated according to your internal practices. The end user is responsible for completion of the method development and validation. Promise Proteomics mAbXmise kits are not available for sale in all countries. For information on availability, please contact your local sales representative.
Abstract
Providing a mass spec workflow for monitoring inflammation monoclonal antibodies (mAbs) and anti-drug antibodies levels, combining sample preparation, analyses, and data processing that can be rapidly implemented in clinical labs willing to use mass spec for mAbs testing.
Introduction
For 25 years, mAbs have revolutionized the management of inflammatory bowel diseases. The original anti-TNF drugs, such as infliximab and adalimumab, and more recently additional biologics, including ustekinumab and vedolizumab, have demonstrated their efficacy for the induction and maintenance of remission in Crohn’s disease and ulcerative colitis.1,2
Therapeutic drug monitoring (TDM) has also been incorporated into daily clinical practice, especially for patients losing response to therapeutics (reactive monitoring). Hence, measuring serum drug levels and anti-drug antibodies is useful for clinicians to explain drug failure and adapting the medication regimen for patients.
For the 4 mAbs cited above, minimal therapeutic target trough levels in maintenance have been published by international groups of experts (ECCO, ACG, AGA) and are: infliximab >5 µg/mL, adalimumab >7,5 µg/mL, vedolizumab >15 µg/mL, and ustekinumab >1 µg/mL.3-5
The difficulties for a laboratory willing to offer TDM of mAbs reside in the limited offer of assays for the concerned mAbs and the variability and lack of robustness of these assays. The challenge is to offer a more robust approach with optimal turnaround time and cost-effectiveness.
Liquid chromatography - mass spectrometry (LC-MS/MS) is a method of choice for this application, now described and used by multiple clinical teams.6-8 LC-MS/MS enables direct, peptide-level detection and quantification and thus provides optimal specificity, with reduced susceptibility to matrix interference, enabling unambiguous identification of drug-derived peptides and immune responses even in patients receiving multiple biologics. By doing so, false-negative and false-positive rates are largely reduced, enabling users to drastically reduce errors and samples reanalysis.9-11 Due to its dynamic range, LC-MS/MS offers an extended measuring range and enables multiplex quantification, which is useful to simultaneously monitor two biologics after a switch or in the case of combination therapies. Finally, a key benefit is the addition of internal standards, which correct the variability (sample variability, variability during technical operations or analyses) and improve the standardization across laboratories.
This application note presents a streamlined workflow dedicated to inflammation mAbs testing using mass spectrometry. This workflow combines ready-to-use kits that largely facilitate the implementation of the assay in the clinical laboratory and use for routine clinical care, and the analytical sensitivity, precision, and accuracy of Waters Xevo TQ Absolute XR Tandem Quadrupole Mass Spectrometer. The analytical methods have been set up and optimized for 4 inflammation mAbs, commonly used to treat inflammatory bowel disease (IBD) patients – infliximab, adalimumab, ustekinumab, and vedolizumab – as well as two anti-drug antibodies – anti-IFX and anti-ADL. This global solution can be implemented rapidly in clinical labs that are willing to perform mAbs testing and looking for an alternative to immunoassays.
Experimental
Materials and Methods
Materials
- Sample Preparation Kits
- ITDM2 Monoclonal Antibodies Quantification Kit – Multiplex infliximab, adalimumab, ustekinumab, vedolizumab, obinutuzumab, secukinumab, and tocilizumab (PROMISE Proteomics, France).
- IADA1 Anti-Drug Antibodies Quantification Kit – multiplex ANTI-Infliximab and ANTI-Adalimumab (PROMISE Proteomics, France).
- Equipment
Equipment recommended: Multichannel pipettes multi-volumes, orbital shaker, vacuum manifold and centrifuge for 96-well plates or positive pressure manifold, nitrogen evaporator or speed-vacuum, pre-slit seal mat for 96 well plates.
- Mass Spec Analysis
- ACQUITY™ Premier Peptide CSH™ C18, 130 Å, 1.7 µm Column (p/n : 186009488)
- Sample analyses were performed on a Xevo TQ Absolute XR Mass Spectrometer
- Data processing was performed using MassLynx™ Software version 4.2 SCN1050
Methods
Sample Preparation
CALs, QCs, and patient serum samples were prepared for LC-MS analysis using the mAbXmise kits according to the kit instructions. For drug-mAbs quantification, 10 µL of serum samples were used with an ITDM2 kit and 100 µl of serum samples were used for antidrug mAbs quantification using an IADA1 kit. After sample preparation with the kits, samples were digested with CutXmise protease, provided in the kits and incubated at 37 °C overnight.
LC Conditions for ITDM2 and IADA1
Chromatographic separation was achieved using an ACQUITY Premier System equipped with a flow-through needle injector (FTN), a binary pump (BSM), and a column manager compartment (CM-A). The separation was performed within 8 min on an ACQUITY Premier Peptide CSH C18 Column (100 x 2.1 mm, 130 Å, 1.7 µm) with 0.1% formic acid in water and in ACN as mobile phases A and B, respectively. The gradient is shown in Table 1.
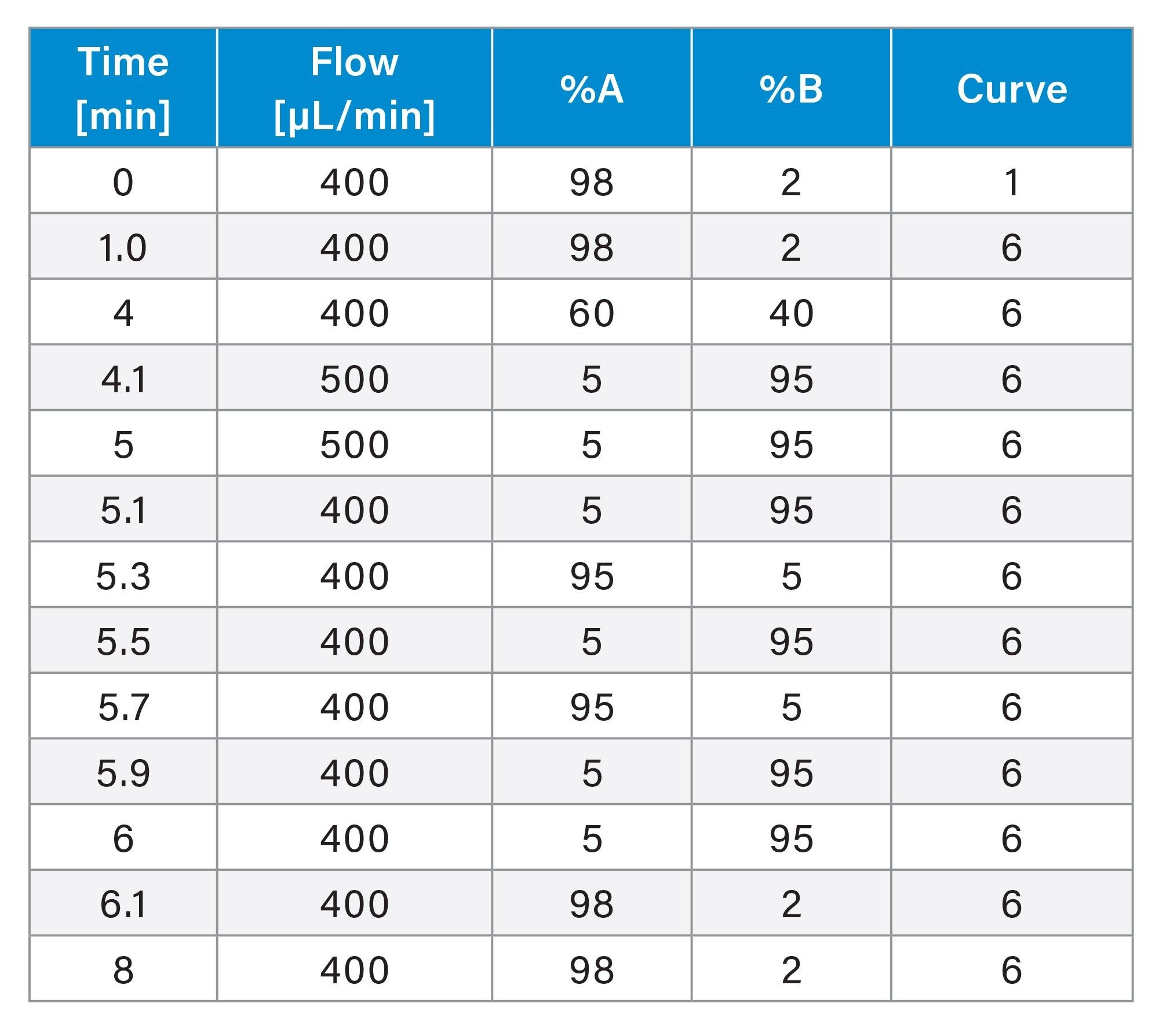
MS Conditions for ITDM2 and IADA1
The mass detection of the signature peptides was performed in MRM mode on a Xevo TQ Absolute XR Tandem Quadrupole Mass Spectrometer, equipped with an ESI source. The detection was achieved in positive polarity with 1.0 kV capillary voltage, 650 °C desolvation temperature, and 1100 L/h desolvation gas flow. The MRM transitions for detecting the peptides and their corresponding internal standard are listed in Tables 2a and 2b.
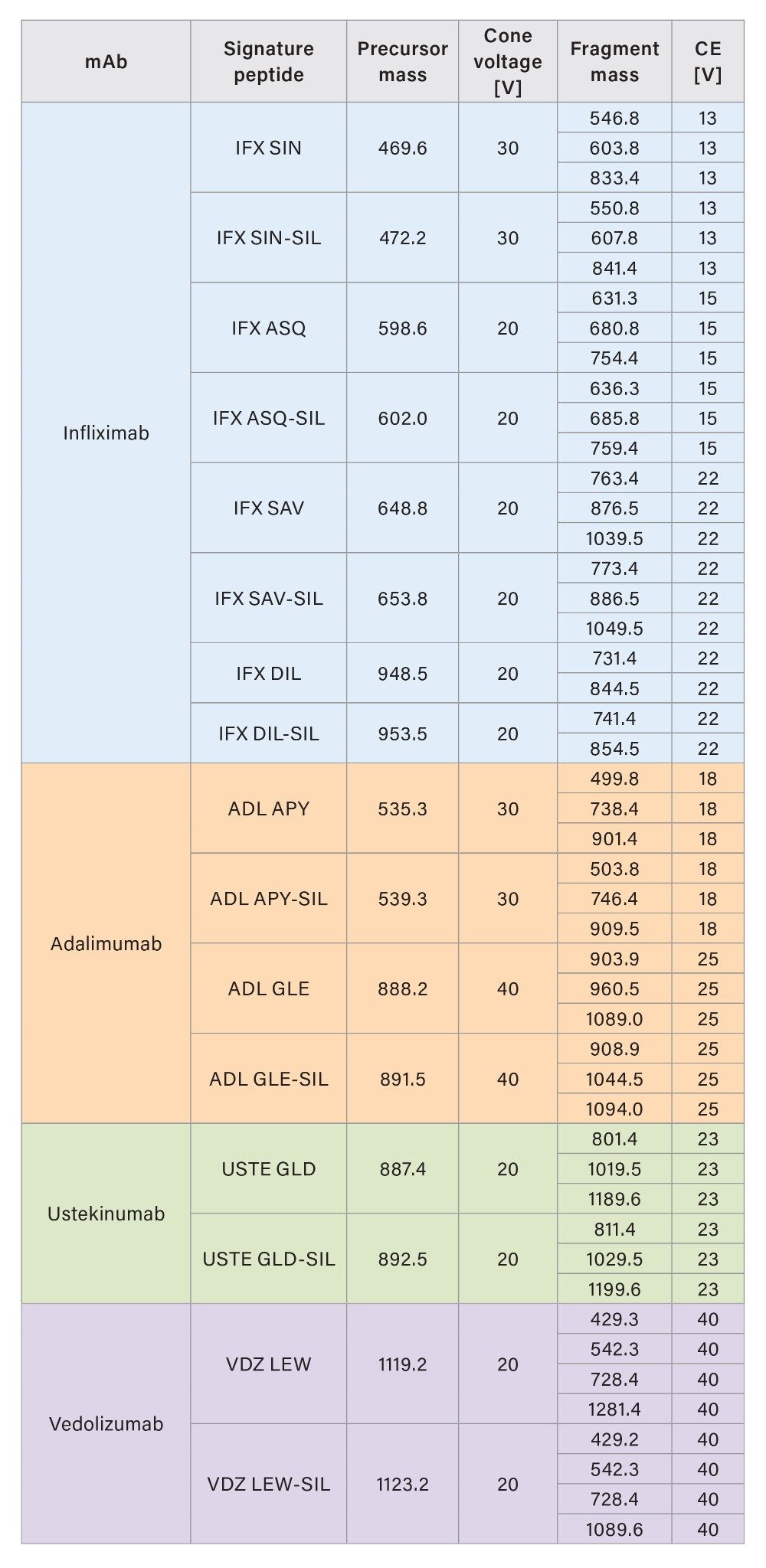
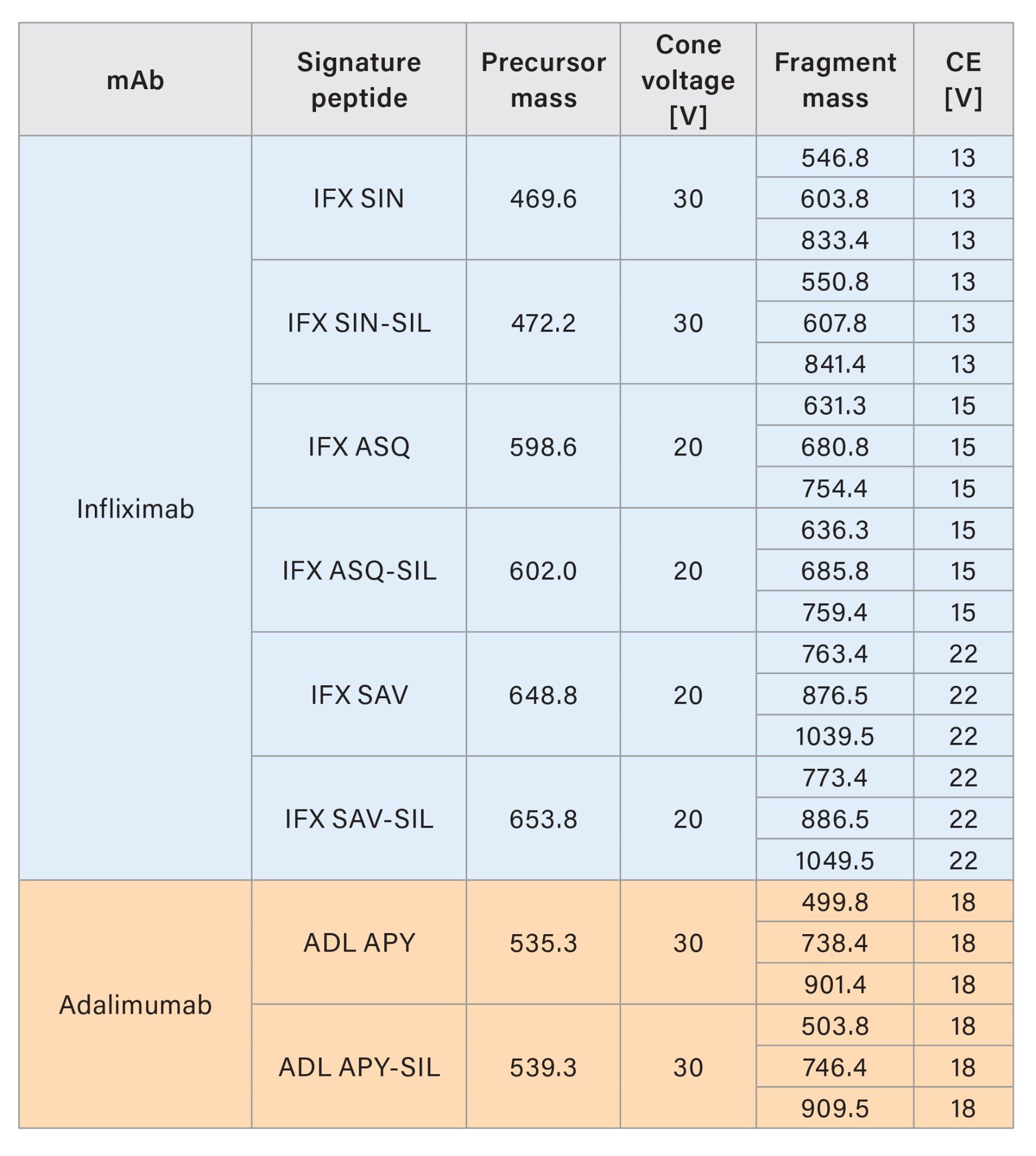
Results and Discussion
ITDM2 Kit for Infliximab, Adalimumab, Ustekinumab, and Vedolizumab Drug Levels
Analytical Sensitivity and Calibration Linearity - ITDM2
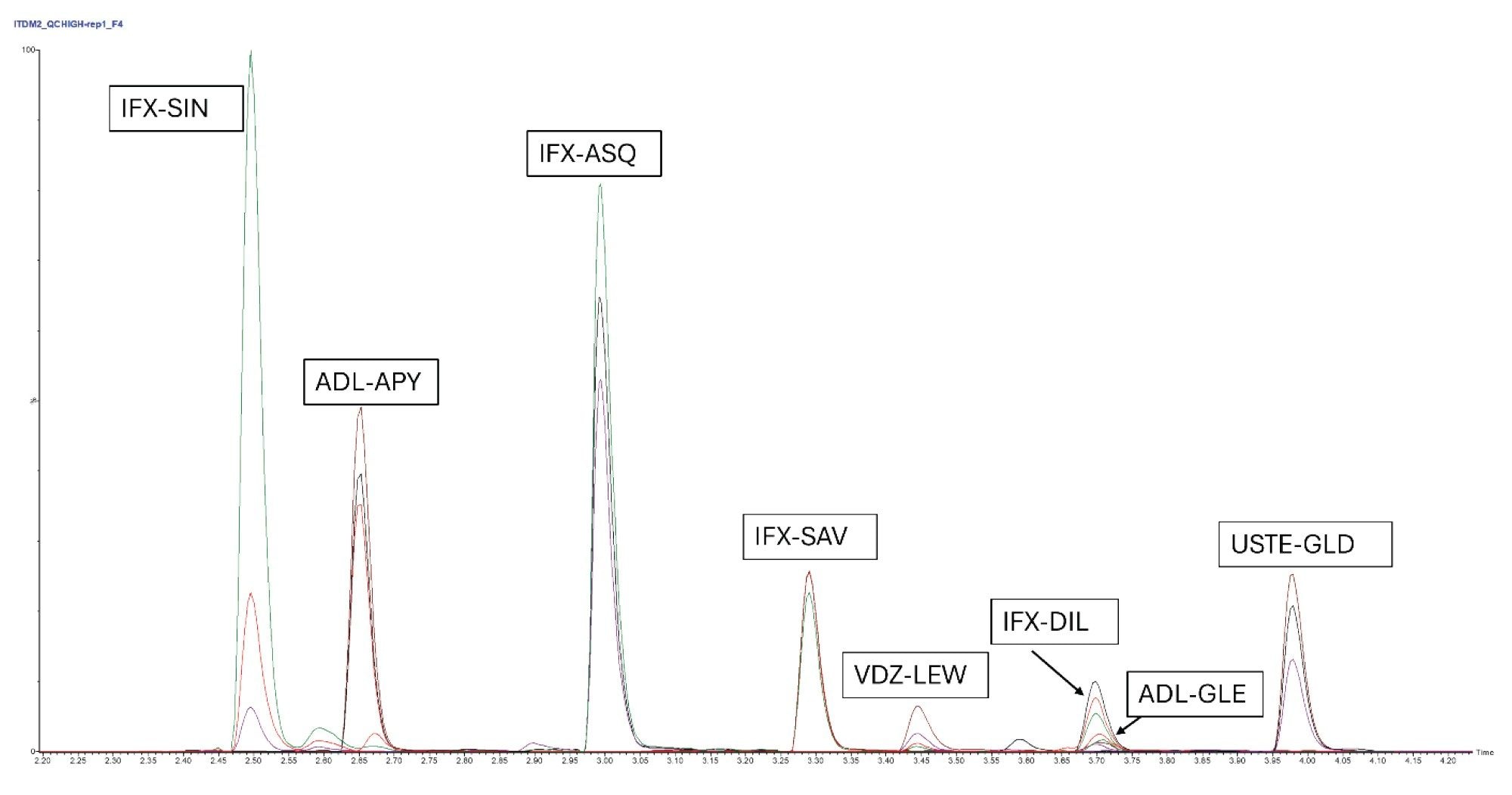
For all peptides, calibration samples were prepared in triplicates and analyzed in a concentration range between 0.5 µg/mL and 100 µg/mL. The MRM chromatogram for QC high at 50 µg/mL is presented in Figure 1.
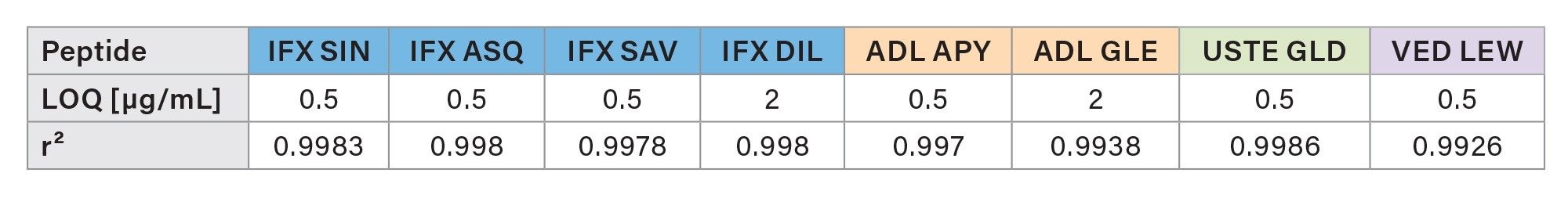
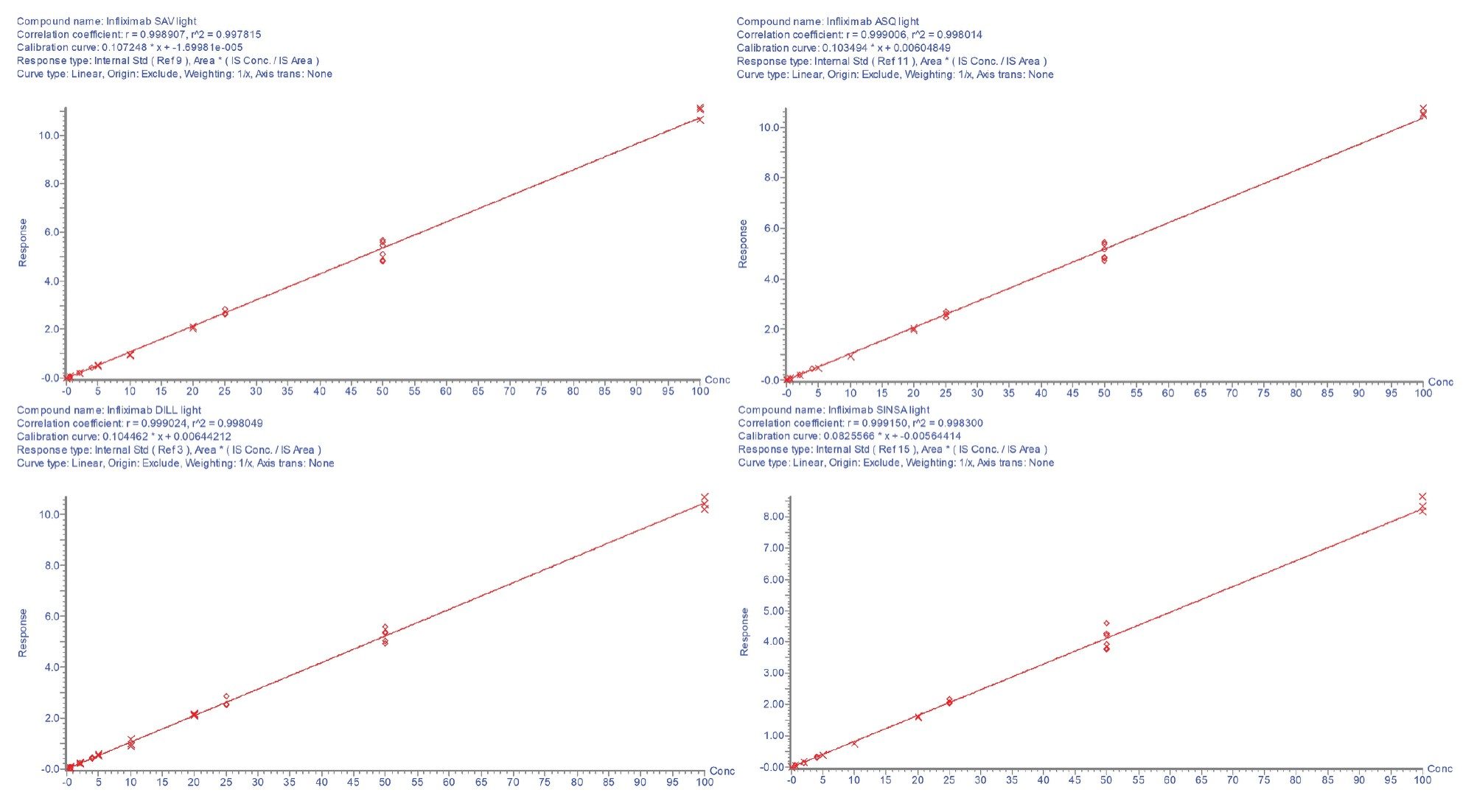
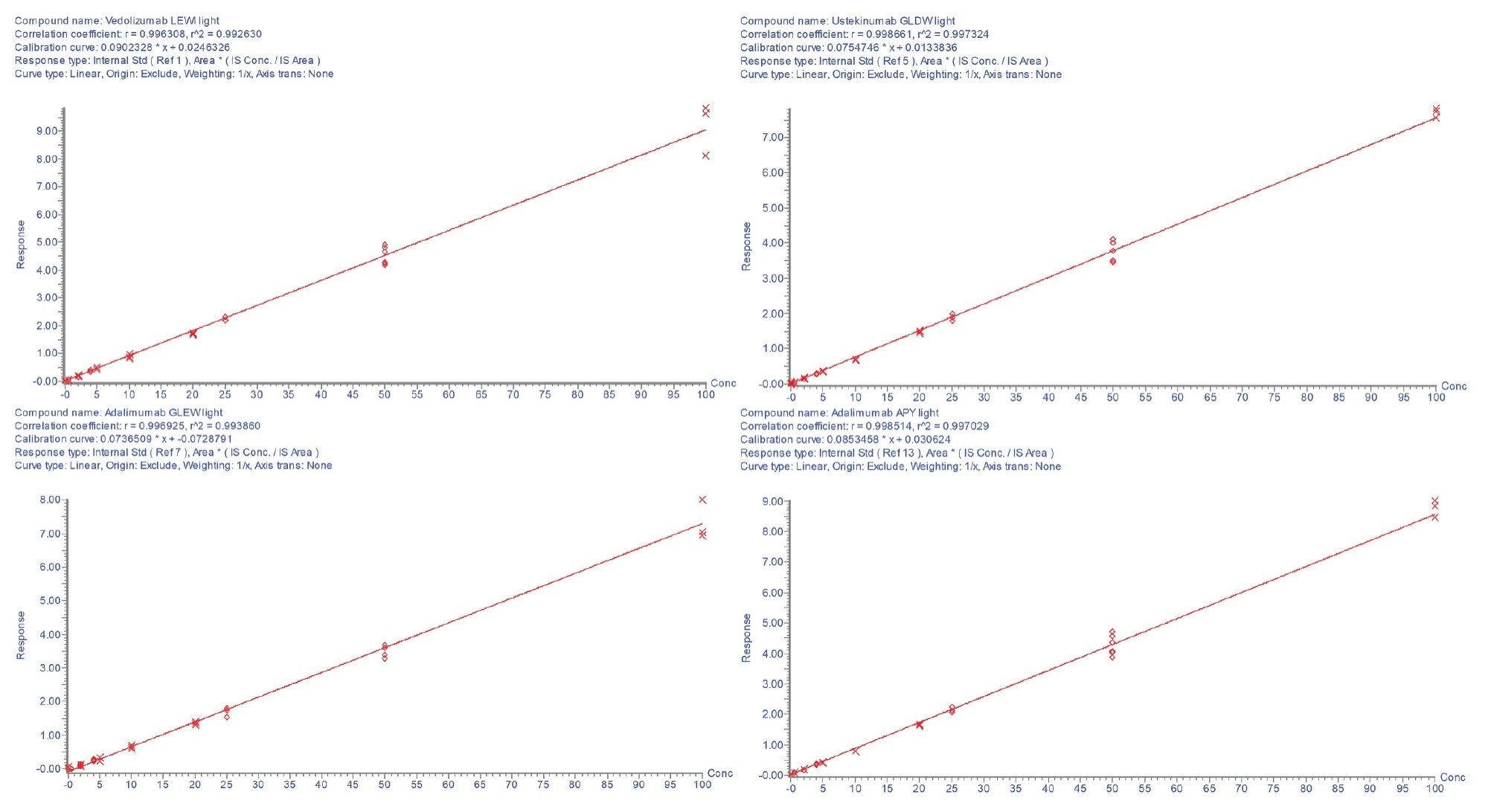
The calibration lines were found to be linear with r²>0.99 for each of the peptides. The calibration curves are shown in Figures 2a and 2b. All individual calibration point deviations were within the acceptance criteria (≤±15%). LOQ and r² values for each peptide are shown in Table 3.
Precision and Accuracy - ITDM2
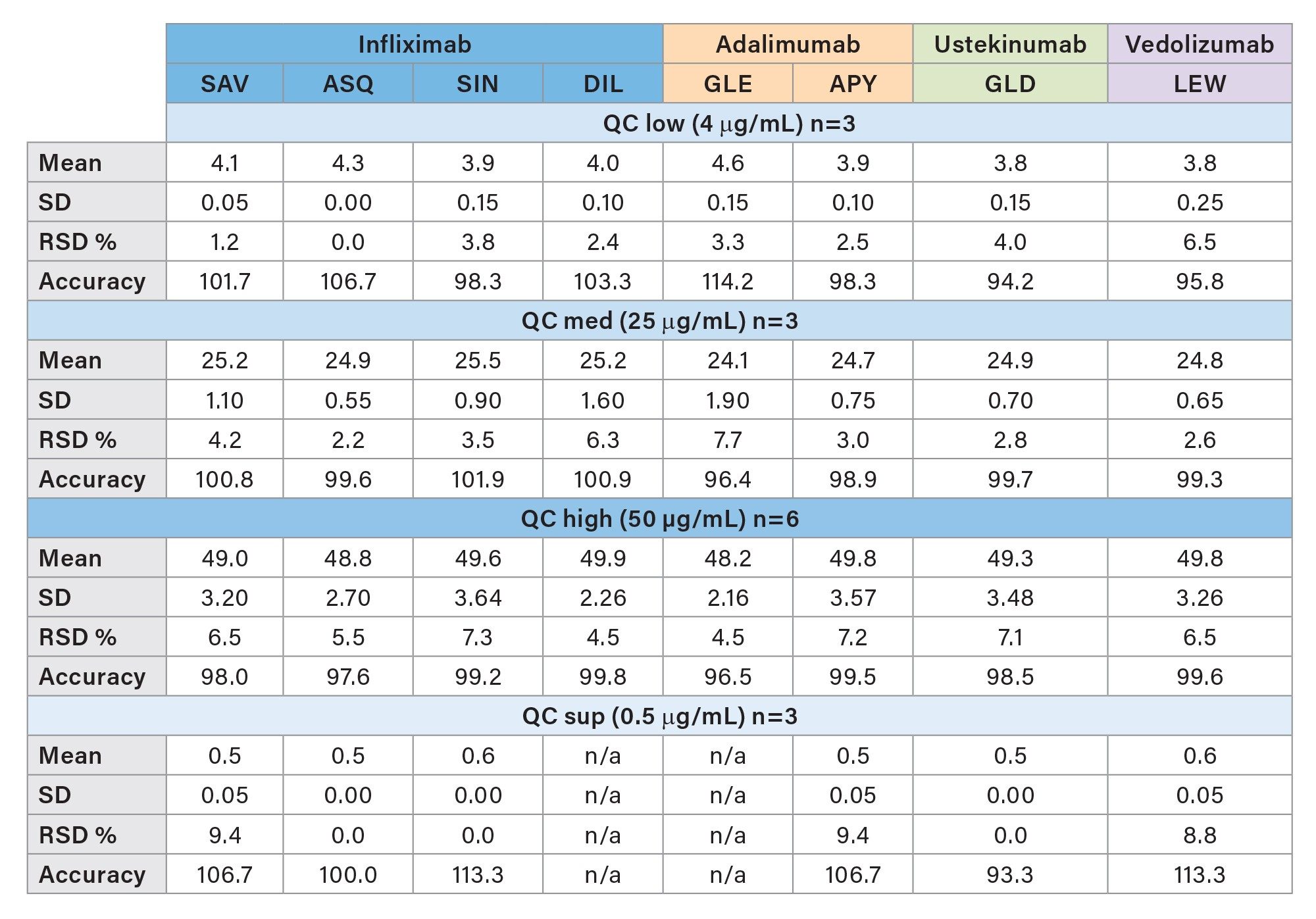
Method precision and quantitative accuracy were estimated based on various injections of QC samples at different concentrations. The relative standard deviation and accuracy of QC sup (0.5 µg/mL; n=3), QC low (4 µg/mL; n=3), QC med (25 µg/mL; n=3) and QC high (50 µg/mL; n=6) were calculated for each peptide and sample group individually. All peptides show an RSD <10% and are accurate within a range between 94.2% and 114.2%. Results are shown in Table 4.
IADA1 Kit for Anti-Infliximab and Anti-Adalimumab Levels
IADA1 Kit Results
Analytical Sensitivity and Calibration Linearity – IADA1
For all peptides, calibration samples were prepared in duplicates and analyzed in a concentration range between 10 ng/mL and 400 ng/mL. The MRM chromatogram for QC at 75 ng/mL is presented in Figure 3.
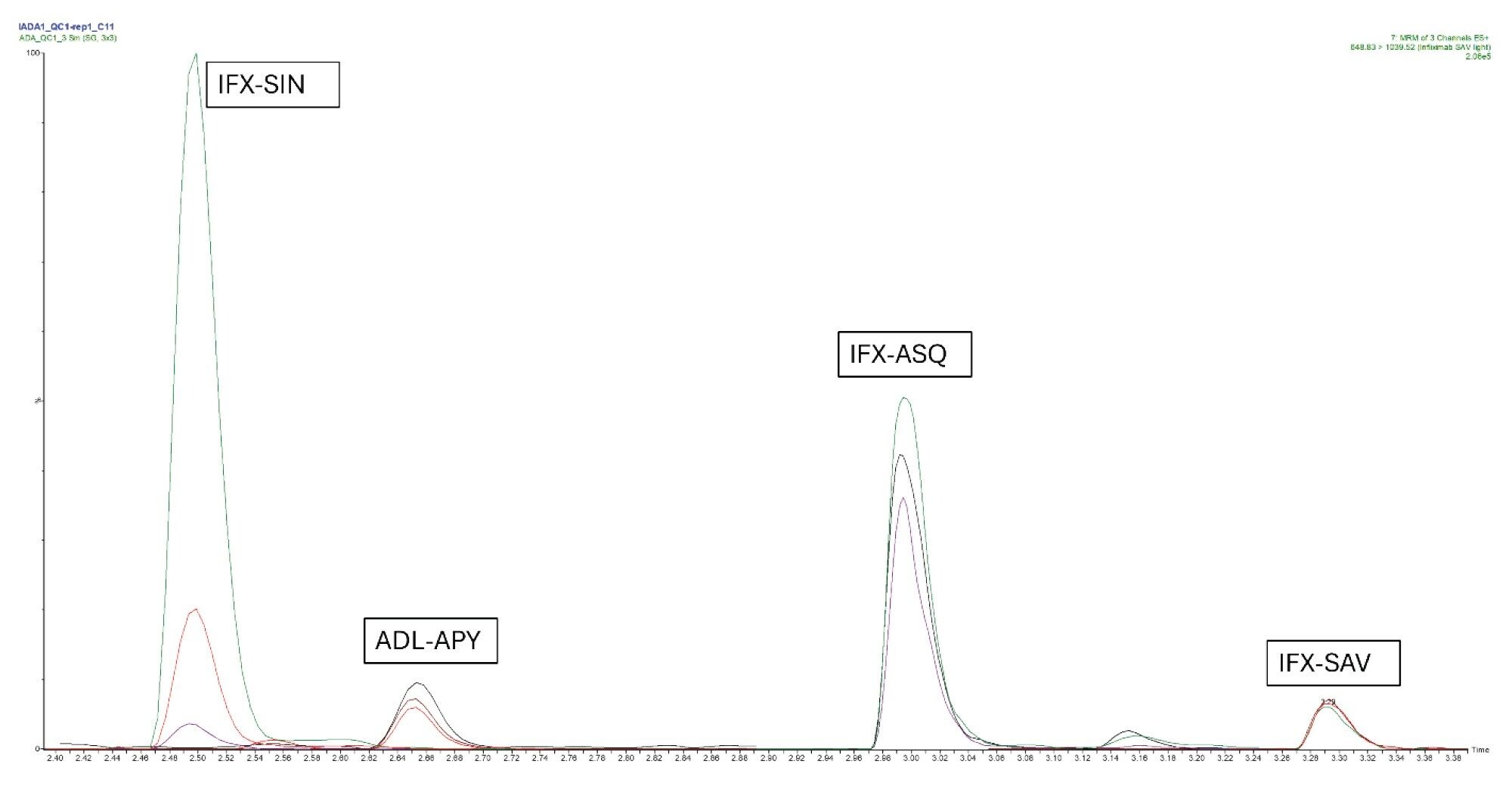
Data evaluation was performed using Arigo Biolaboratories software with 4PL regression.
The linearity plot is calculated based on the following parameters:
x = concentration [µg/mL]
y = (MS response labelled)/(MS response unlabelled ) x100
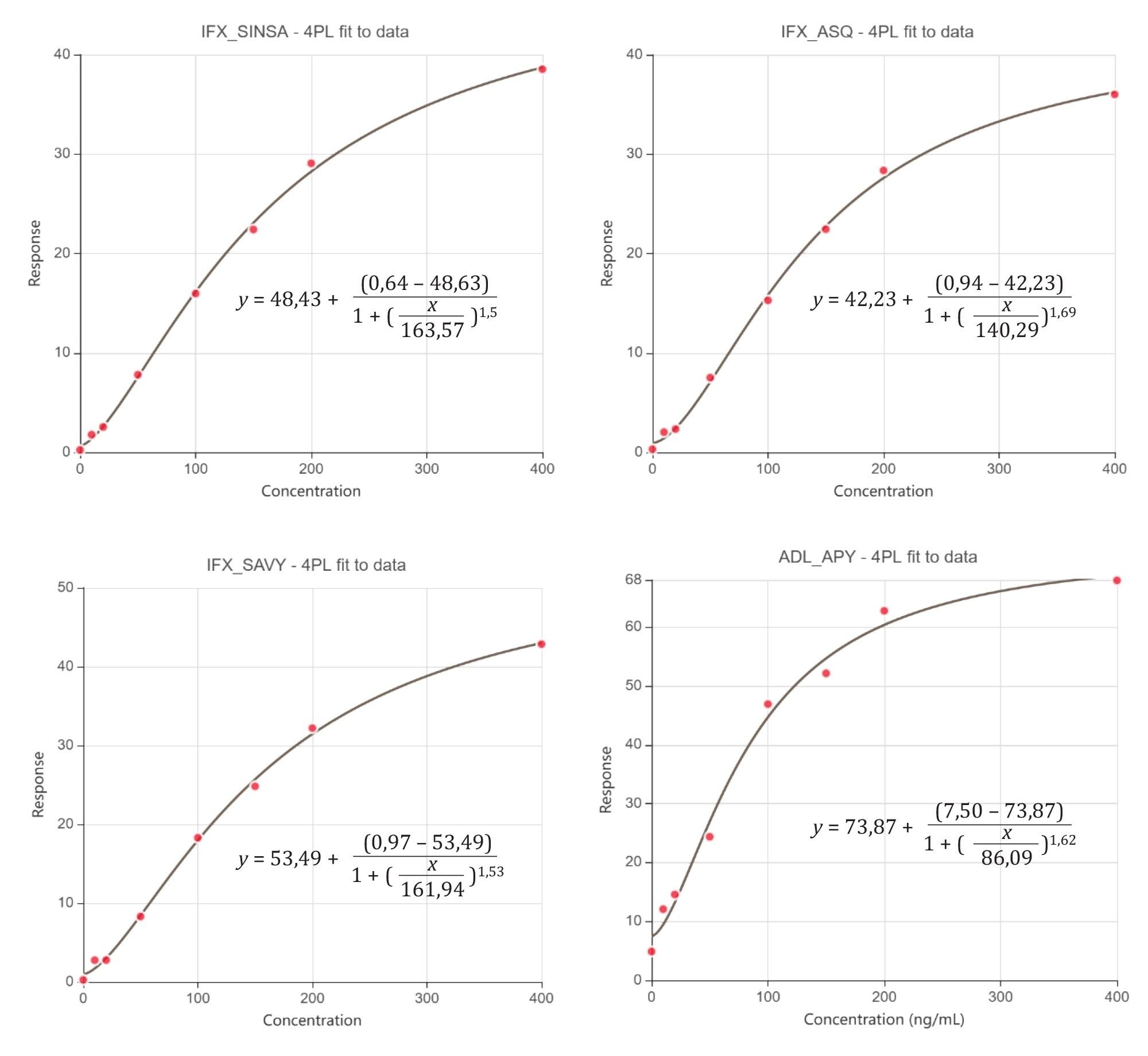
The calibration curves were found quadratic as expected, where y=d+ (a-d)/(1+(x/c)b , with a= Theoretical response at zero concentration, b= Slope factor, c=Inflection point (EC50/IC50), and d=Theoretical response at infinite concentration.
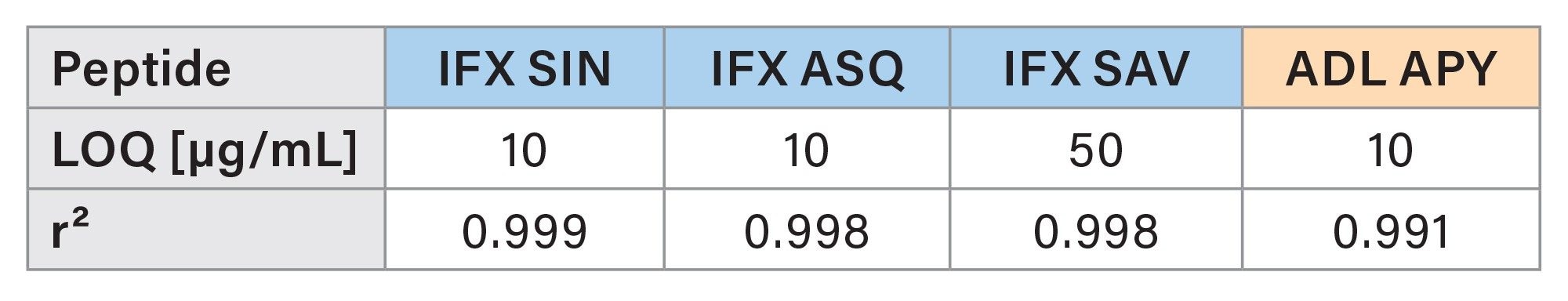
For each peptide, LOQ and r² values are presented in Table 5.
The calibration curves for adalimumab and infliximab peptides are shown in Figure 4.
LOQ and r² values for each peptide are shown in Table 5.
Precision and Accuracy IADA1
Method precision and quantitative accuracy were estimated based on 4 injections of individually prepared QC samples at 75 ng/mL. The relative standard deviation and accuracy of the QC samples (n=4) were calculated for each peptide individually. All peptides show an RSD <10% and are accurate within a range between 90.5% and 105.6%. Results are shown in Table 6.
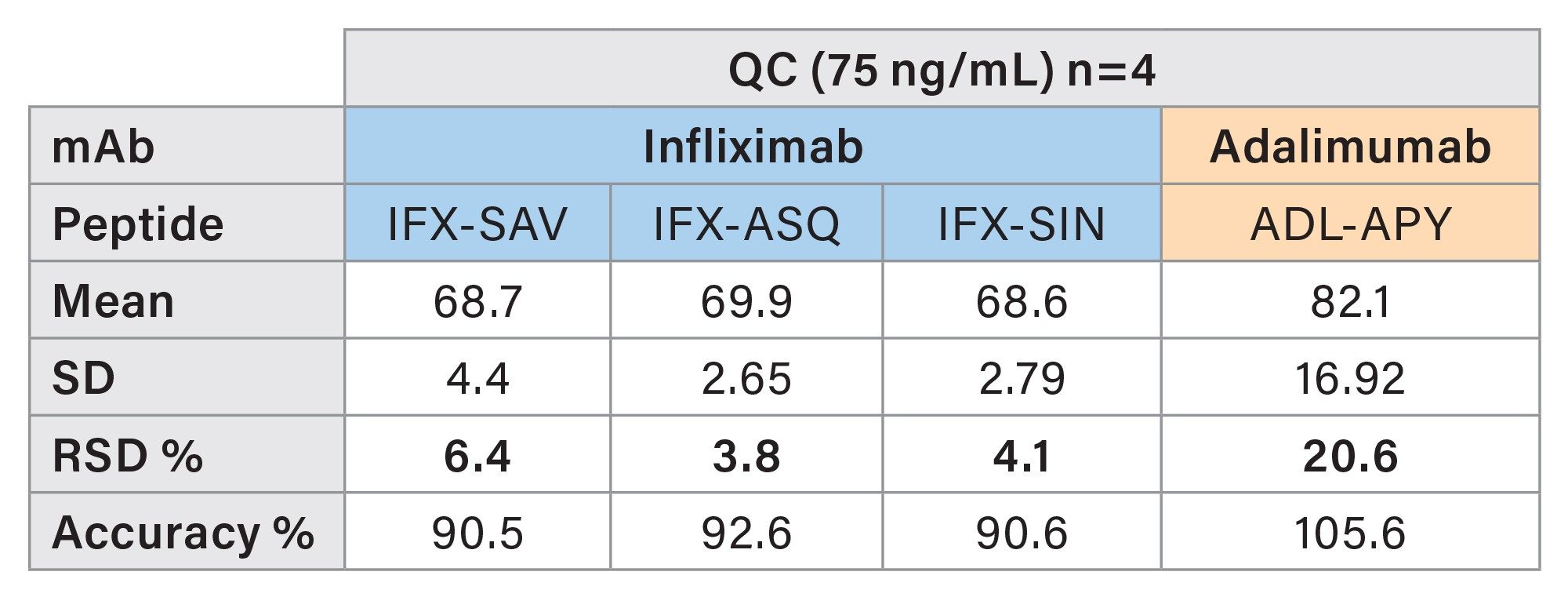
Quantitation of Patient Samples
Seven patient samples were analyzed using the Promise Proteomics ITDM2 kit and cross-compared to values assigned by immunoassay. Patients were under treatment of adalimumab or infliximab. No ustekinumab or vedolizumab were present in these samples. The results are in good accordance with the reported values achieved by immunoassays and shown in Table 7. As expected, in some cases the absolute values differ in direct comparison of both detection techniques. These findings have been already reported in literature.12, 13 However, good correlation between mAbXmise kits and immunoassay has already been reported in the literature.8
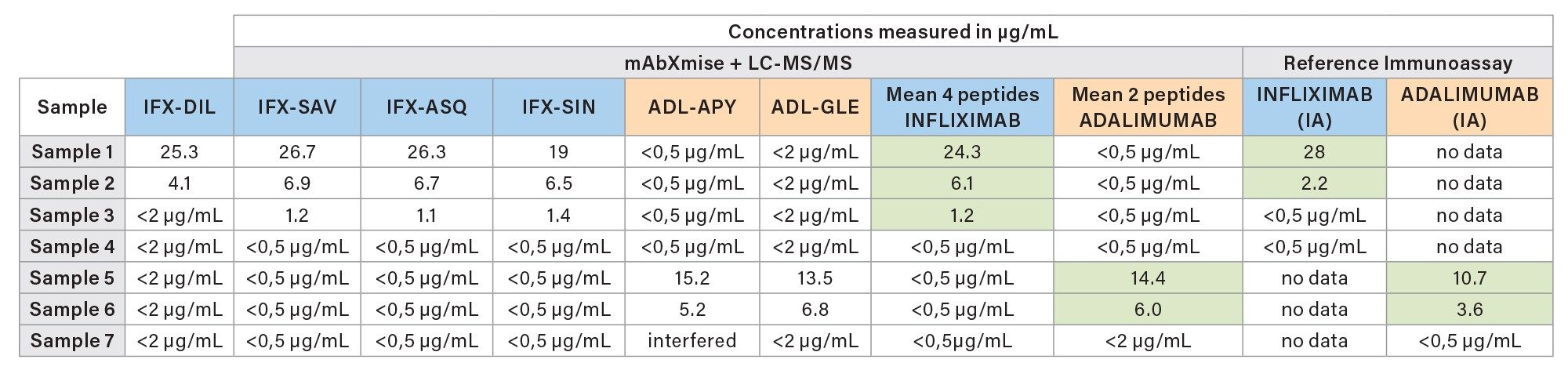
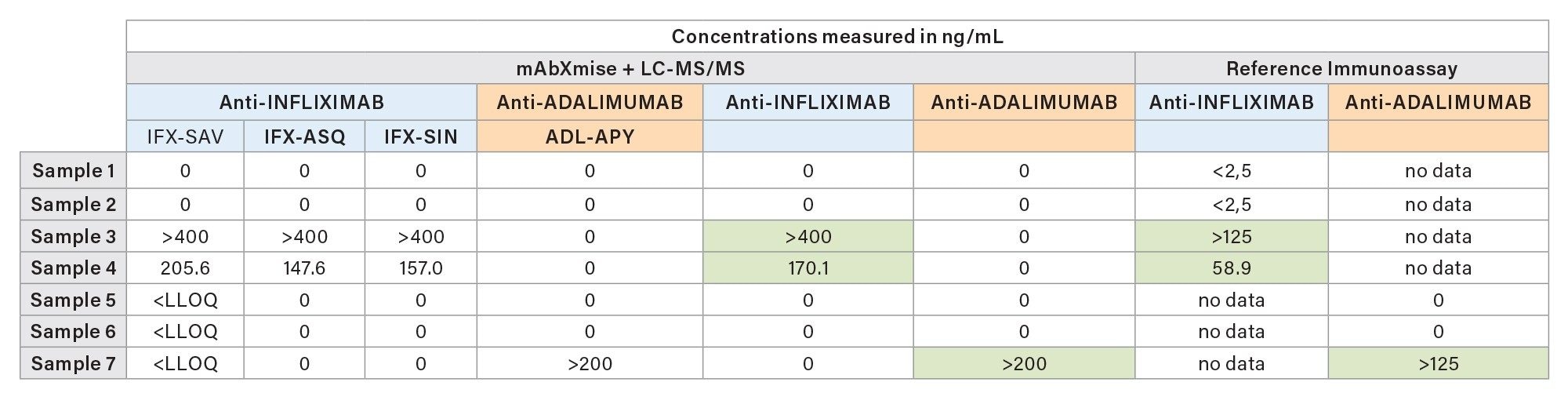
In parallel to ITDM2 analysis, the seven patient samples were analyzed with the IADA1 kit. The presence of antidrug antibodies were observed for two patients treated with infliximab and for one patient treated with adalimumab, in good accordance with the results of the ELISA antidrug antibodies. Both methods show the presence of anti-drug antibodies. Some values were above their specific method ULOQ.
Conclusion
This application note highlights a comprehensive suite dedicated to the quantification of monoclonal antibodies and anti-drug antibodies. The solution combines a range of kits for sample preparation (mAbXmise, Promise Proteomics, France) and associated analytical methods, optimized for the Waters Xevo TQ Absolute XR Mass Spectrometer.
The results demonstrate excellent analytical performance (accuracy, precision). The key advantage of the described solution lies in its rapid implementation in clinical laboratories already familiar with mass spectrometry. The transition from small-molecule analysis to antibodies is made easier and faster, thanks both to the kits that simplify sample preparation for this application and to the analytical methods that are ready to use on the instrument.
Acknowledgements
The authors would like to thank Markus Mallek (MVZ Dortmund, Germany) who kindly provided anonymized samples for comparison.
Specific mentions
The analytical data presented in this material are intended to demonstrate the robustness of a clinical research method. These data in no way substitute for independent method validation required by any applicable legal or laboratory standards.
In Europe, mAbXmise kits are in vitro diagnostic medical devices for professional laboratory use. mAbXmise kits determine the plasma concentration of monoclonal antibodies. Analytical performance depends on the instrument’s characteristics and its settings. A validation of the analytical method shall be conducted according to internal practices. Consult the specific instructions for more information. Promise Proteomics products are distributed globally, so uses, applications, and availability of product in each country depend on local regulatory registration status. Products are not registered outside of Europe. Manufacturer PROMISE PROTEOMICS SAS.
Waters, Xevo, ACQUITY, CSH, and MassLynx are trademarks of Waters Technologies Corporation.
References
- Juillerat P, Grueber MM, Ruetsch R, Santi G, Vuillèmoz M, Michetti P. Positioning biologics in the treatment of IBD: A practical guide - Which mechanism of action for whom?. Curr Res Pharmacol Drug Discov. 2022 Apr 28;3:100104. doi: 10.1016/j.crphar.2022.100104. PMID: 35570855; PMCID: PMC9092374.
- Irving PM, Gecse KB. Optimizing Therapies Using Therapeutic Drug Monitoring: Current Strategies and Future Perspectives. Gastroenterology. 2022 Apr;162(5):1512–1524. doi: 10.1053/j.gastro.2022.02.014. Epub 2022 Feb 12. PMID: 35167865.
- Gordon H, Minozzi S, Kopylov U, Verstockt B, Chaparro M, Buskens C, Warusavitarne J, Agrawal M, Allocca M, Atreya R, Battat R, Bettenworth D, Bislenghi G, Brown SR, Burisch J, Casanova MJ, Czuber-Dochan W, de Groof J, El-Hussuna A, Ellul P, Fidalgo C, Fiorino G, Gisbert JP, Sabino JG, Hanzel J, Holubar S, Iacucci M, Iqbal N, Kapizioni C, Karmiris K, Kobayashi T, Kotze PG, Luglio G, Maaser C, Moran G, Noor N, Papamichael K, Peros G, Reenaers C, Sica G, Sigall-Boneh R, Vavricka SR, Yanai H, Myrelid P, Adamina M, Raine T. ECCO Guidelines on Therapeutics in Crohn's Disease: Medical Treatment. J Crohns Colitis. 2024 Oct 15;18(10):1531–1555. doi: 10.1093/ecco-jcc/jjae091. PMID: 38877997.
- Rubin DT, Ananthakrishnan AN, Siegel CA, Barnes EL, Long MD. ACG Clinical Guideline Update: Ulcerative Colitis in Adults. Am J Gastroenterol. 2025 Jun 3;120(6):1187–1224. doi: 10.14309/ajg.0000000000003463. PMID: 40701556.
- Lichtenstein GR, Loftus EV, Afzali A, Long MD, Barnes EL, Isaacs KL, Ha CY. ACG Clinical Guideline: Management of Crohn's Disease in Adults. Am J Gastroenterol. 2025 Jun 3;120(6):1225–1264. doi: 10.14309/ajg.0000000000003465. PMID: 40701562.
- Ladwig PM, Rivard AL, Barbeln A, Maus A, Murray DL, Snyder MR, Willrich MAV. Infliximab Therapeutic monitoring by tryptic peptide LC-MS/MS method improvements lead to improved accuracy with decreased imprecision and turnaround time. J Mass Spectrom Adv Clin Lab. 2024 Feb 3;32:24-30. doi: 10.1016/j.jmsacl.2024.01.007. PMID: 38405411; PMCID: PMC10884749.
- Marin C, Khoudour N, Millet A, Lebert D, Bros P, Thomas F, Ternant D, Lacarelle B, Guitton J, Ciccolini J, Blanchet B. Cross-Validation of a Multiplex LC-MS/MS Method for Assaying mAbs Plasma Levels in Patients with Cancer: A GPCO-UNICANCER Study. Pharmaceuticals (Basel). 2021 Aug 12;14(8):796. doi: 10.3390/ph14080796. PMID: 34451893; PMCID: PMC8401780.
- Tron C, Lemaitre F, Bros P, Goulvestre C, Franck B, Mouton N, Bagnos S, Coriat R, Khoudour N, Lebert D, Blanchet B. Quantification of infliximab and adalimumab in human plasma by a liquid chromatography tandem mass spectrometry kit and comparison with two ELISA methods. Bioanalysis. 2022 Jun;14(11):831-844. doi: 10.4155/bio-2022–0057. Epub 2022 Jun 23. PMID: 35735172.
- Hoofnagle AN, Wener MH. The fundamental flaws of immunoassays and potential solutions using tandem mass spectrometry. J Immunol Methods. 2009 Aug 15;347(1–2):3–11. doi: 10.1016/j.jim.2009.06.003. Epub 2009 Jun 16. PMID: 19538965; PMCID: PMC2720067.
- Jourdil JF, Némoz B, Gautier-Veyret E, Romero C, Stanke-Labesque F. Simultaneous Quantification of Adalimumab and Infliximab in Human Plasma by Liquid Chromatography-Tandem Mass Spectrometry. Ther Drug Monit. 2018 Aug;40(4):417–424. doi: 10.1097/FTD.0000000000000514. PMID: 29608486.
- Ladwig PM, Barnidge DR, Willrich MAV. Mass Spectrometry Approaches for Identification and Quantitation of Therapeutic Monoclonal Antibodies in the Clinical Laboratory. Clin Vaccine Immunol. 2017 May 5;24(5):e00545-16. doi: 10.1128/CVI.00545-16. PMID: 28274937; PMCID: PMC5424237.
- Clarke WT, Papamichael K, Vande Casteele N, Germansky KA, Feuerstein JD, Melmed GY, Siegel CA, Irving PM, Cheifetz AS. Infliximab and Adalimumab Concentrations May Vary Between the Enzyme-Linked Immunosorbent Assay and the Homogeneous Mobility Shift Assay in Patients With Inflammatory Bowel Disease: A Prospective Cross-Sectional Observational Study. Inflamm Bowel Dis. 2019 Oct 18;25(11):e143–e145. doi: 10.1093/ibd/izz202. PMID: 31559417.
- Willrich MAV, Lazar-Molnar E, Snyder MR, Delgado JC. Comparison of Clinical Laboratory Assays for Measuring Serum Infliximab and Antibodies to Infliximab. J Appl Lab Med. 2018 May 1;2(6):893–903. doi: 10.1373/jalm.2017.024869. PMID: 33636834.
720009223, February 2026