
In this application note, we demonstrate the use of a glycoprotein BEH amide, 300Å, 1.7 μm column to develop LC-MS and LC-UV techniques that can be used to rapidly profile domain specific information about the N-linked glycosylation of IgG molecules.
Without question, the most successfully exploited protein modality for therapeutic applications has been monoclonal antibodies (mAbs), which currently account for nearly half of the biopharmaceutical market.1 An intriguing characteristic of mAbs, in particular IgG-based mAbs, is that they are formed by the linking of two identical light chains and two identical heavy chains through disulfide bonding and non-covalent interactions. Moreover, the resulting mAb structure exhibits functionally significant subunits, for instance one crystallizable fragment (Fc domain) and two equivalent antigen binding fragments (Fab domains). In what is commonly referred to as a middle-up or middle-down analysis,2-5 native mAbs can be proteolyzed into these and other related subunits enzymatically, as a means to perform cell-based studies and to facilitate characterization. One increasingly popular way to produce subunit digests of mAbs is via the IdeS protease (Immunoglobulin Degrading Enzyme of S. pyogenes).2,6 IdeS cleaves with high fidelity at a conserved sequence motif in the hinge region of humanized mAbs to cleanly produce, upon reduction, three 25 kDa mAb fragments that are amenable to mass spectrometry and useful for localizing different attributes of therapeutic mAbs (Figure 1).3 IdeS digestion combined with reversed-phase (RP) chromatography has, in fact, been proposed as a simple identity test for mAbs and fusion proteins, because IdeS produced subunits from different drug products will exhibit diagnostic RP retention times.3 Additionally, RP techniques have been shown to be useful in assaying and obtaining domain specific information about oxidation, since RP retention can be dramatically affected by the oxidation of protein residues, such as methionine.3
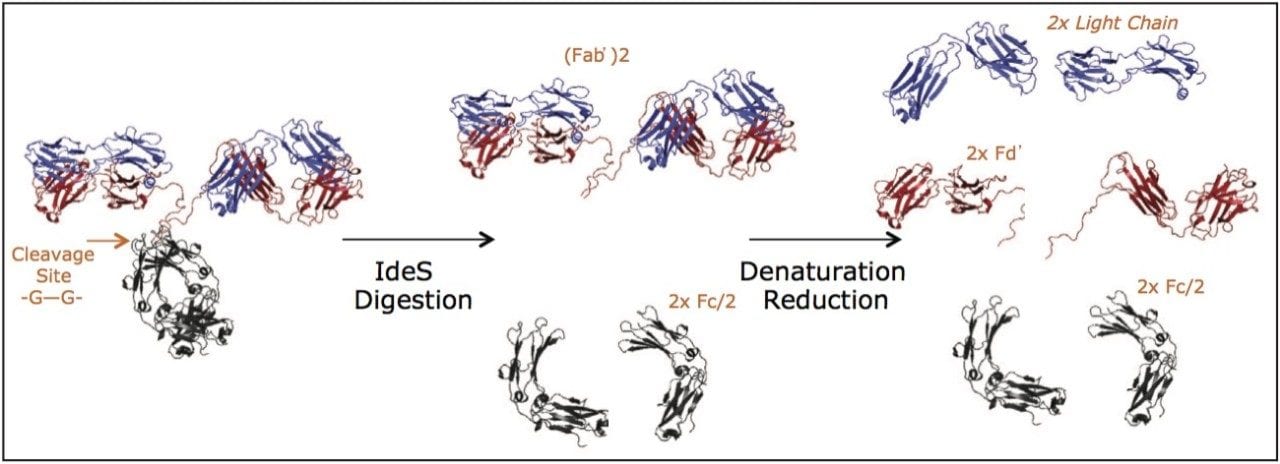
It should, however, be kept in mind that many IgG modifications more strongly elicit changes in the hydrophilicity of a mAb along with its capacity for hydrogen bonding. A very obvious example of this type of modification is glycosylation. Glycans released from a mAb are very often profiled by hydrophilic interaction chromatography (HILIC), in which case an amide bonded stationary phase has historically been used, because it affords high retentivity as a consequence of its hydrophilicity and propensity for hydrogen bonding.7 Here, we propose that HILIC with an amide bonded stationary phase also be considered for IgG subunit separations. For such an application, a stationary phase with a wide average pore diameter is critical, so that large subunit structures will have access to the majority of the porous network and be less prone to restricted diffusion while eluting through a column.8-9 Through the development of a sub-2-μm wide pore amide stationary phase, we have facilitated a novel and complementary workflow to RP based subunit analyses. In this application note, we demonstrate the use of a glycoprotein BEH amide, 300Å, 1.7 μm column to develop LC-MS and LC-UV techniques that can be used to rapidly profile domain specific information about the N-linked glycosylation of IgG molecules.
IdeS digestion and reduction of mAbs:
Formulated trastuzumab was diluted 7 fold into 20 mM phosphate (pH 7.1) and incubated at a concentration of 3 mg/mL with IdeS (Promega, Madison, WI) for 30 minutes at 37 °C at a 50:1 w/w ratio of trastuzumab to IdeS. The resulting IdeS-digested antibody was denatured and reduced by the addition of 1M TCEP (tris(2-carboxyethyl)phosphine) and solid GuHCl (guanidine hydrochloride). The final buffer composition for the denaturation/reduction step was approximately 6 M GuHCl, 80 mM TCEP, and 10 mM phosphate (pH 7.1). IdeS-digested trastuzumab (1.5 mg/mL) was incubated in this buffer at 37 °C for 1 hour. An IdeS digested, reduced sample of an IgG1K mAb obtained from NIST as candidate reference material #8670 (lot #3F1b) was prepared in the same manner.
Prior to digestion with IdeS,10 cetuximab was treated with carboxypeptidase B to complete the partial removal of the lysine- C-terminal residues that is typical of the antibody.4 Formulated cetuximab was mixed with carboxypeptidase B (223 μ/mg, Worthington, Lakewood, NJ) at a ratio of 100:1 (w/w), diluted into 20 mM phosphate (pH 7.1), and incubated at a concentration of 1.8 mg/mL for 2 hours at 37 °C. The carboxypeptidase B treated cetuximab was then added to 100 units of IdeS and incubated for 30 minutes at 37 °C. The resulting IdeS digest was denatured and reduced by the addition of 1 M TCEP and solid GuHCl. The final buffer composition for the denaturation/ reduction step was approximately 6 M GuHCl, 80 mM TCEP, and 10 mM phosphate (pH 7.1). IdeS-digested cetuximab (0.9 mg/mL) was incubated in this buffer at 37 °C for 1 hour.
RapiFluor-MS labeled N-glycans were prepared from cetuximab using a GlycoWorks RapiFluor-MS N-Glycan Kit (p/n 176003606) according to the guidelines provided in its Care and Use Manual (715004793).
Method conditions
(unless otherwise noted)
ACQUITY UPLC Glycoprotein BEH Amide, 300Å, 1.7 μm columns (as well as other amide columns intended for glycoprotein separations) should be conditioned via two sequential injections and separations of 40 μg Glycoprotein Performance Test Standard (p/n 186008010); 10 μL injections of 4 mg/mL in 0.1% trifluoroacetic acid [TFA], 80% acetonitrile [ACN]) or with equivalent loads of a sample for which the column has been acquired. The separation outlined by the following method can be employed for conditioning with the Glycoprotein Performance Test Standard.
|
Column dimensions: |
2.1 x 150 mm |
|
Mobile phase A: |
0.1% (v/v) TFA, water |
|
Mobile phase B: |
0.1% (v/v) TFA, ACN |
|
Time(min) |
%A |
%B |
Curve |
|---|---|---|---|
|
0.0 |
15.0 |
85.0 |
6 |
|
0.5 |
15.0 |
85.0 |
6 |
|
1.0 |
33.0 |
67.0 |
6 |
|
21.0 |
40.0 |
60.0 |
6 |
|
22.0 |
100.0 |
0.0 |
6 |
|
24.0 |
100.0 |
0.0 |
6 |
|
25.0 |
15.0 |
85.0 |
6 |
|
35.0 |
15.0 |
85.0 |
6 |
|
LC system: |
ACQUITY UPLC H-Class Bio System |
|
Sample temp.: |
5 °C |
|
Analytical column temp.: |
45 °C (trastuzumab and NIST IgG1K subunit HILIC separations) 60 °C (cetuximab subunit HILIC separations) 80 °C (trastuzumab reversed phase (RP) subunit separations) |
|
Flow rate: |
0.2 mL/min |
|
Mobile phase A: |
0.1% (v/v) TFA, water |
|
Mobile phase B: |
0.1% (v/v) TFA, ACN |
|
UV detection: |
214 nm, 10 Hz |
|
Injection volume: |
≤1.2 μL (aqueous diluents). Note It might be necessary to avoid high organic diluents for some samples due to the propensity for proteins to precipitate under ambient conditions. A 2.1 mm I.D. column can accommodate up to a 1.2 μL aqueous injection before It might be necessary to avoid high organic diluents for some samples due to the propensity for proteins to precipitate under ambient conditions. A 2.1 mm I.D. column can accommodate up to a 1.2 μL aqueous injection before chromatographic performance is negatively affected. |
|
Waters columns: |
ACQUITY UPLC Glycoprotein BEH Amide, 300Å, 1.7 μm, 2.1 x 150 mm (p/n 176003702, with Glycoprotein Performance Test Standard); ACQUITY UPLC Glycan BEH Amide, 130Å, 1.7 μm, 2.1 x 150 mm (p/n 186004742); ACQUITY UPLC Protein BEH C4, 300Å, 1.7 μm, 2.1 x 150 mm (p/n 186004497) |
|
Other columns: |
Column A: 2.6 μm, 2.1 x 150 mm Column B: 1.8 μm, 2.1 x 150 mm |
|
Vials: |
Polypropylene 12 x 32 mm Screw Neck Vial, 300 μL volume (p/n 186002640) |
|
Time(min) |
%A |
%B |
Curve |
|---|---|---|---|
|
0.0 |
95.0 |
5.0 |
6 |
|
1.0 |
66.7 |
33.3 |
6 |
|
21.0 |
59.7 |
40.3 |
6 |
|
22.0 |
20.0 |
80.0 |
6 |
|
24.0 |
20.0 |
80.0 |
6 |
|
25.0 |
95.0 |
5.0 |
6 |
|
35.0 |
95.0 |
5.0 |
6 |
|
Time(min) |
%A |
%B |
Curve |
|---|---|---|---|
|
0 |
20.0 |
80.0 |
6 |
|
1 |
30.0 |
70.0 |
6 |
|
21 |
37.0 |
63.0 |
6 |
|
22 |
100.0 |
0.0 |
6 |
|
24 |
100.0 |
0.0 |
6 |
|
25 |
20.0 |
80.0 |
6 |
|
35 |
20.0 |
80.0 |
6 |
|
MS system: |
Xevo G2 QTof or SYNAPT G2-S HDMS |
|
Ionization mode: |
ESI+ |
|
Analyzer mode: |
Resolution (~20 K) |
|
Capillary voltage: |
3.0 kV |
|
Cone voltage: |
45 V |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
350 °C |
|
Desolvation gas flow: |
800 L/Hr |
|
Calibration: |
NaI, 2 μg/μL from 500–5000 m/z |
|
Acquisition: |
500–4000 m/z, 0.5 sec scan rate |
|
Data management: |
MassLynx Software (v4.1)/UNIFI V1.7 |
|
LC conditions for RapiFluor-MS Released N-Glycan |
|
|---|---|
|
HILIC separations: |
ACQUITY UPLC H-Class Bio System |
|
Sample temp.: |
10 °C |
|
Analytical column temp.: |
60 °C |
|
Fluorescence detection: |
Ex 265/Em 425 nm (RapiFluor-MS) (5 Hz scan rate [50 mm column], Gain =1) |
|
Injection volume: |
10 μL (DMF/ACN diluted sample) |
|
Mobile phase A: |
50 mM ammonium formate, pH 4.4 (LC-MS grade; from a 100x concentrate, p/n 186007081) |
|
Mobile phase B: |
ACN (LC-MS grade) |
|
Columns: |
ACQUITY UPLC Glycan BEH Amide, 130Å, 1.7 μm, 2.1 x 50 mm (p/n 186004740) |
|
Vials: |
Polypropylene 12 x 32mm, 300 μL, Screw Neck Vial, (p/n 186002640) |
|
Time(min) |
Flow Rate(mL/min) |
%A |
%B |
Curve |
|---|---|---|---|---|
|
0 |
0.4 |
25 |
75 |
6 |
|
11.7 |
0.4 |
46 |
54 |
6 |
|
12.2 |
0.2 |
100 |
0 |
6 |
|
13.2 |
0.2 |
100 |
0 |
6 |
|
14.4 |
0.2 |
25 |
75 |
6 |
|
15.9 |
0.4 |
25 |
75 |
6 |
|
18.3 |
0.4 |
25 |
75 |
6 |
|
MS conditions for RapiFluor-MS N-Glycan HILIC separations |
|
|---|---|
|
MS system: |
SYNAPT G2-S HDMS |
|
Ionization mode: |
ESI+ |
|
Analyzer mode: |
TOF MS, resolution mode (~20 K) |
|
Capillary voltage: |
3.0 kV |
|
Cone voltage: |
80 V |
|
Source temp.: |
120 °C |
|
Desolvation temp.: |
350 °C |
|
Desolvation gas flow: |
800 L/Hr |
|
Calibration: |
NaI, 1 μg/μL from 500–2500 m/z |
|
Lockspray (ASM B-side): |
100 fmol/μL Human Glufibrinopeptide B in 0.1% (v/v) formic acid, 70:30 water every 90 seconds |
|
Acquisition: |
500–2500 m/z, 1 Hz scan rate |
|
Data management: |
MassLynx Software (v4.1) |
To demonstrate a conventional approach to IgG subunit mapping, we first analyzed a reduced/IdeS digest of an IgG1 mAb using a RP chromatographic separation with a wide-pore C4 bonded stationary phase (Protein BEH C4, 300Å, 1.7 μm). The IgG1 mAb selected for this work was trastuzumab, given its prominence as a first generation mAb drug product and a potential target for biosimilar development.11 Figure 2A shows a UPLC chromatogram that is typical for reduced, IdeS-digested trastuzumab, wherein three peaks are near equally spaced with an elution order corresponding to the Fc/2, LC and Fd' subunits, respectively. The conditions to produce this high resolution separation entail the use of TFA for ion-pairing. Interestingly, the same mobile phases have proven to be optimal for protein HILIC, as they reduce the hydrophilicity of protein residues by masking them via a hydrophobic ion pair. This, in turn, leads to improved selectivity for hydrophilic modifications.12 That is, an orthogonal method to the RP separation can be achieved via HILIC by simply reversing a gradient and using a newly developed wide-pore amide bonded stationary phase (glycoprotein BEH Amide, 300Å, 1.7 μm).
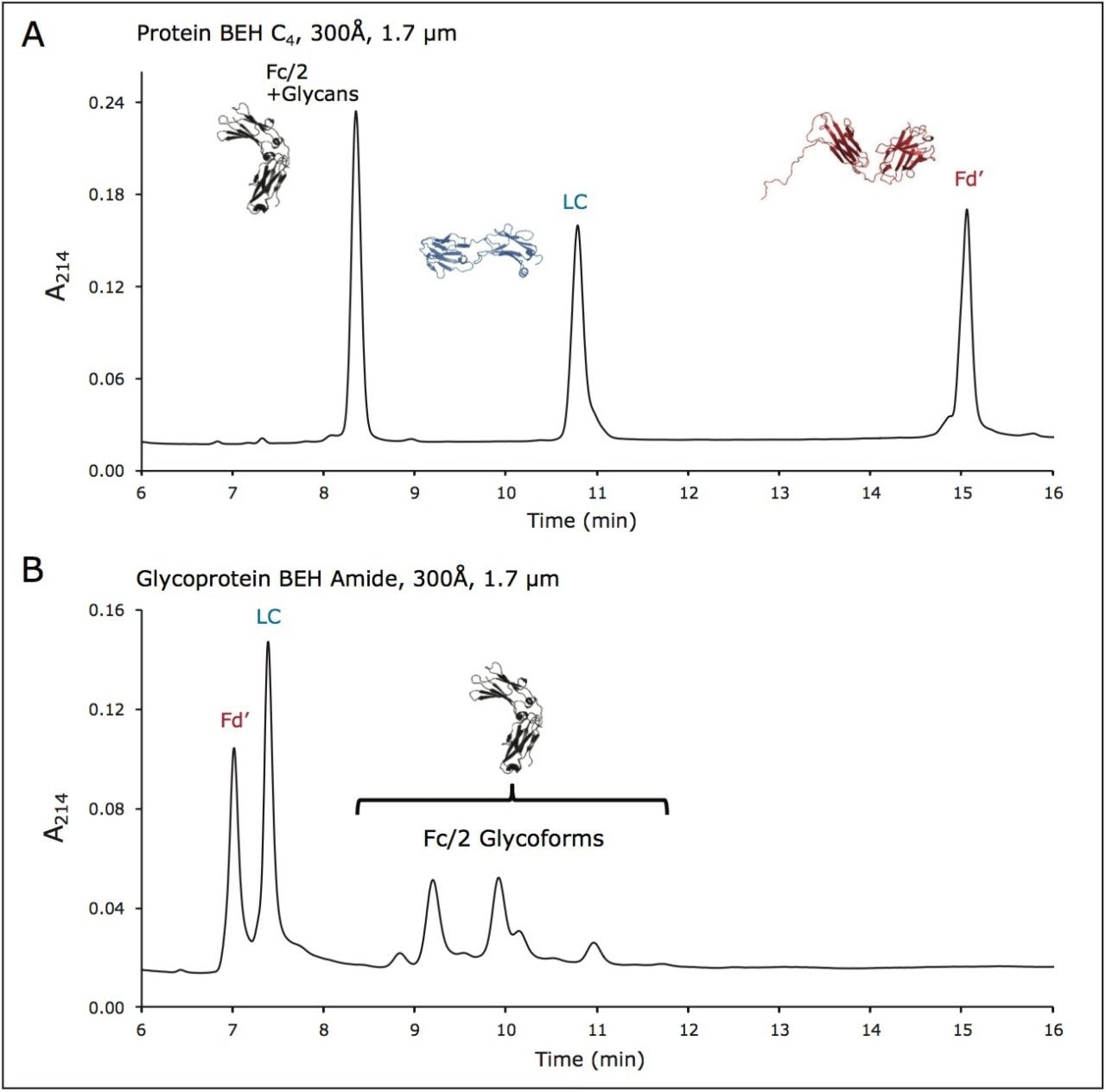
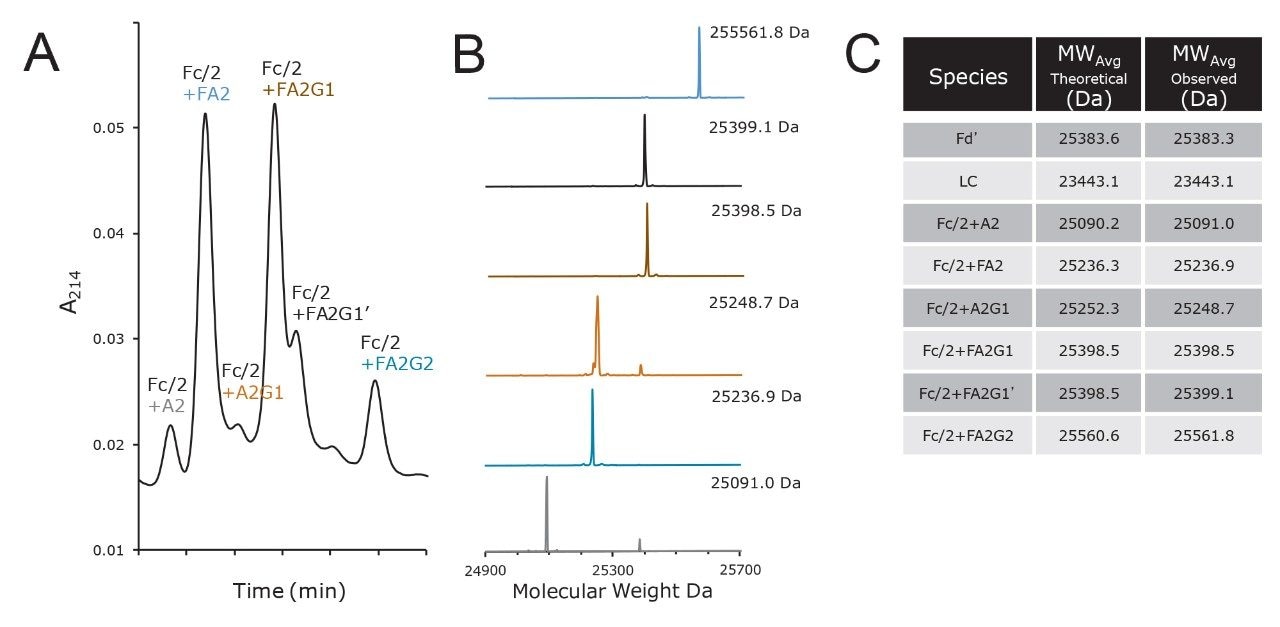
An example of a chromatogram obtained from a column packed with this wide-pore amide material is shown in Figure 2B. Here, the same reduced, IdeS digested trastuzumab is separated into approximately 10 peaks. The first two eluting peaks correspond to the Fd' and LC subunits, while the remaining, more strongly retained peaks correspond to the glycoforms of the Fc/2 subunit. By focusing on the more strongly retained peaks, an analyst can elucidate information about the heterogeneity of glycosylation (Figure 3A). Given that this is a method with volatile mobile phases, the glycoform peaks can be readily interrogated by ESI-MS. Deconvoluted mass spectra and molecular weights corresponding to species in the glycoform profile are presented in Figures 3B and 3C. In Figure 3, chromatographic peaks are labeled with the same color as their corresponding mass spectra. Notice that this HILIC separation facilitates producing deconvoluted mass spectra for individual glycoforms with limited interference between similar molecular weight species, for instance the Fc/2+A2G1 versus the Fc/2+FA2 species (orange versus blue spectrum). In a first pass analysis, all glycan species from trastuzumab that are known to be present at a relative abundance greater than 2% are readily detected.13 It should be noted that lower abundance species, such as Fc/2+M5 (Man5), are also detected and can be observed by extracted ion chromatograms (XICs). This indicates there is a possibility to perform selected reaction monitoring (SRM) MS analyses when and if there is a need to monitor particular low abundance structures. While it is not resolved under these conditions, the M5 Fc/2 glycoform is resolve in a different example separation (see below, Figure 7A).
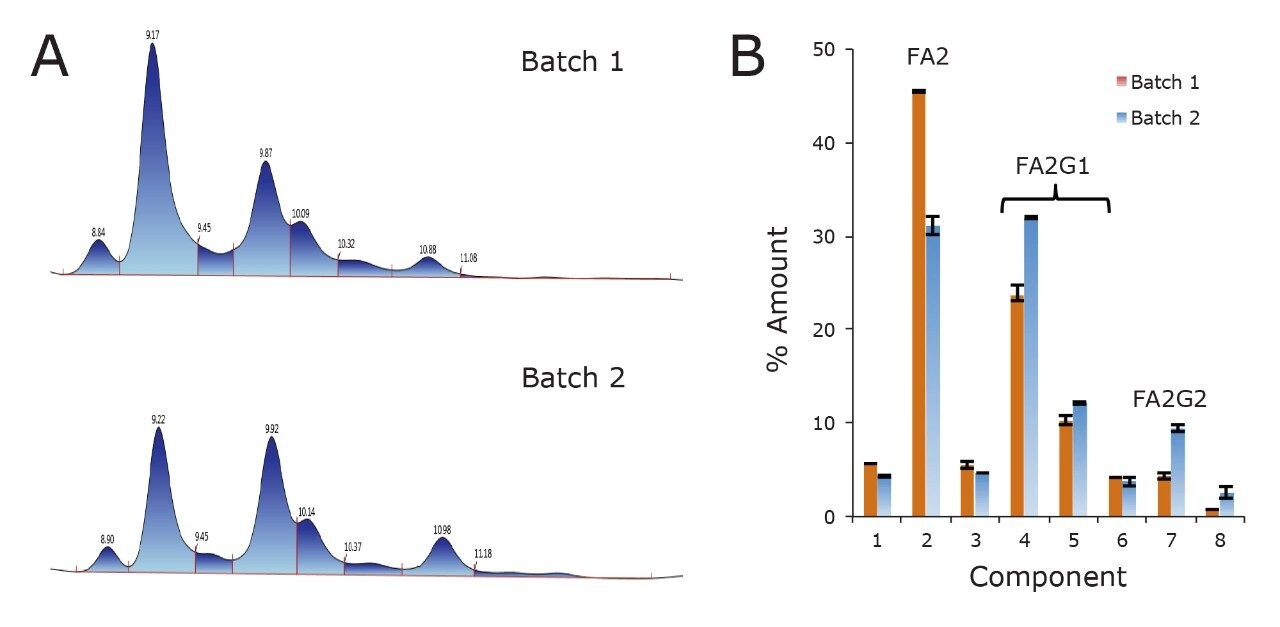
Clearly, data generated by subunit-level HILIC-MS are very information-rich. Optical detection based subunit HILIC separations can be equally informative. To this end, we have applied a HILIC-UV method to perform batch-to-batch analysis of trastuzumab Fc/2 glycosylation, as exemplified in Figure 4. Two example HILIC chromatograms for Fc/2 glycoforms obtained from two different lots of trastuzumab are shown in Figure 4A. Previous testing on these lots has demonstrated differences in glycosylation at the released glycan level.14 Here, by integration of peaks across the profile, we have found that the two lots of trastuzumab indeed differ with respect to their Fc domain glycosylation profiles, in ways consistent with the mentioned released glycan assays. In particular, these lots of trastuzumab differ with respect to their extents of terminal galactosylation, as estimated from the abundances of FA2, FA2G1, and FA2G2 Fc/2 subunits (Figure 4B). This is an informative observation, since the extent of galactosylation can affect complementdependent cytotoxicity (CDC).15
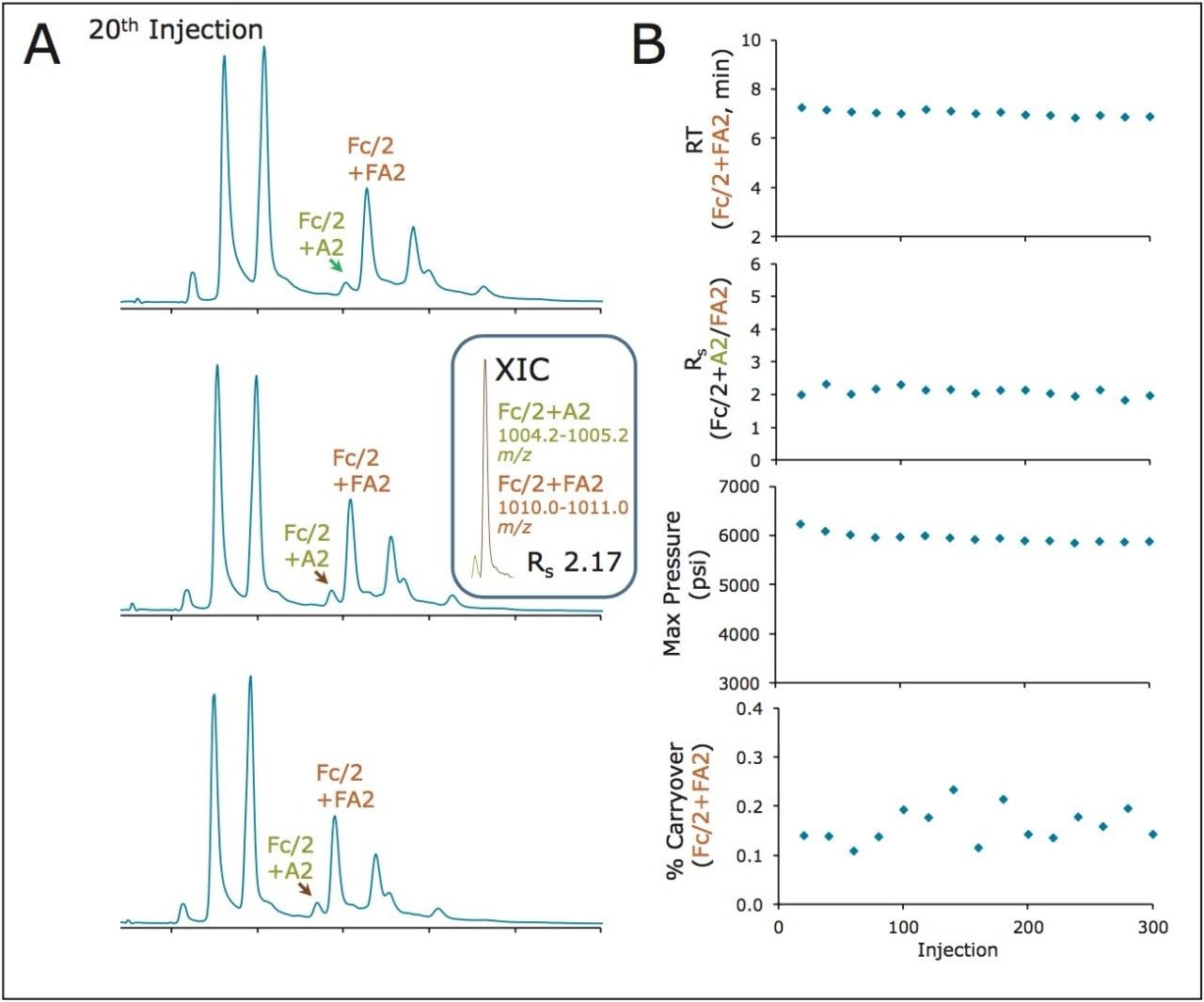
The ability of a BEH amide, 300Å, 1.7 μm column to robustly deliver the above mentioned separations over time was tested by performing a series of experiments involving a single column being subjected to 300 sequential injections of a reduced, IdeS digested trastuzumab sample. This was a potentially challenging use scenario given that the reduced, IdeS digested mAb sample contains both high concentrations of guanidine denaturant and TCEP reducing agent. Total ion chromatograms corresponding to the 20th, 180th, and 300th injections of this experiment are displayed in Figure 5A. In these analyses, particular attention was paid to the half-height resolution of the Fc/2+A2 and Fc/2+FA2 species, which was assessed every 20th separation using extracted ion chromatograms (XICs).
In this testing, several additional chromatographic parameters were also monitored, including the retention time of the Fc/2+FA2 species, the maximum system pressure observed during the chromatographic run, and the percent (%) carryover of the most abundant glycoform, the Fc/2+FA2 species (Figure 5B). Plots of these parameters underscore the consistency of the subunit separation across the lifetime of the column. With noteworthy consistency, the column produced relatively stable retention times, a consistent resolution of the A2 and FA2 glycoforms (Rs≈2), a maximum system pressure consistently at only ~6 Kpsi, and a remarkably low carryover between 0.1 and 0.2%. This latter aspect of the HILIC separations is particularly noteworthy since it indicates that carryover with these methods is almost an order of magnitude lower than analogous C4 based RP methods.
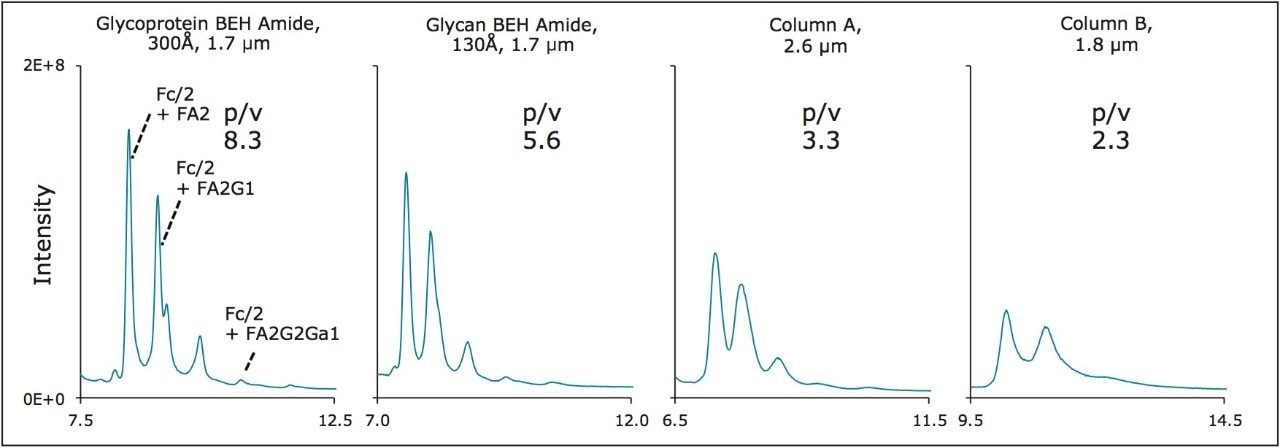
We have benchmarked the performance of this new wide-pore column technology against not only its standard pore diameter analog but also its two most closely related, commercially available alternatives. Figure 6 presents chromatograms obtained for a reduced, IdeS digested sample of an IgG1K mAb acquired from NIST using these different column technologies. In a visual comparison, it is clear that the glycoprotein BEH amide 300Å column significantly outperforms the other three columns. To quantify this assessment, peak-to-valley ratios were calculated for the separation of the FA2 glycoform away from the FA2G1 glycoform. The glycoprotein BEH amide 300Å column was found to demonstrate improvements of 48%, 152%, and 261% over the 130Å glycan BEH amide column and the two alternative, commercially available amide columns, respectively. This mAb sample also has a particularly interesting attribute in that it has a reasonably high relative abundance of an immunogenic alpha-1,3-galactose containing glycan (an FA2G2Ga1 structure).16-17 As shown in Figure 6, this Fc/2+FA2G2Ga1 species can be readily visualized with the wide-pore amide column. This represents a sizeable improvement in the peak capacity of large molecule HILIC separations for this emerging application.
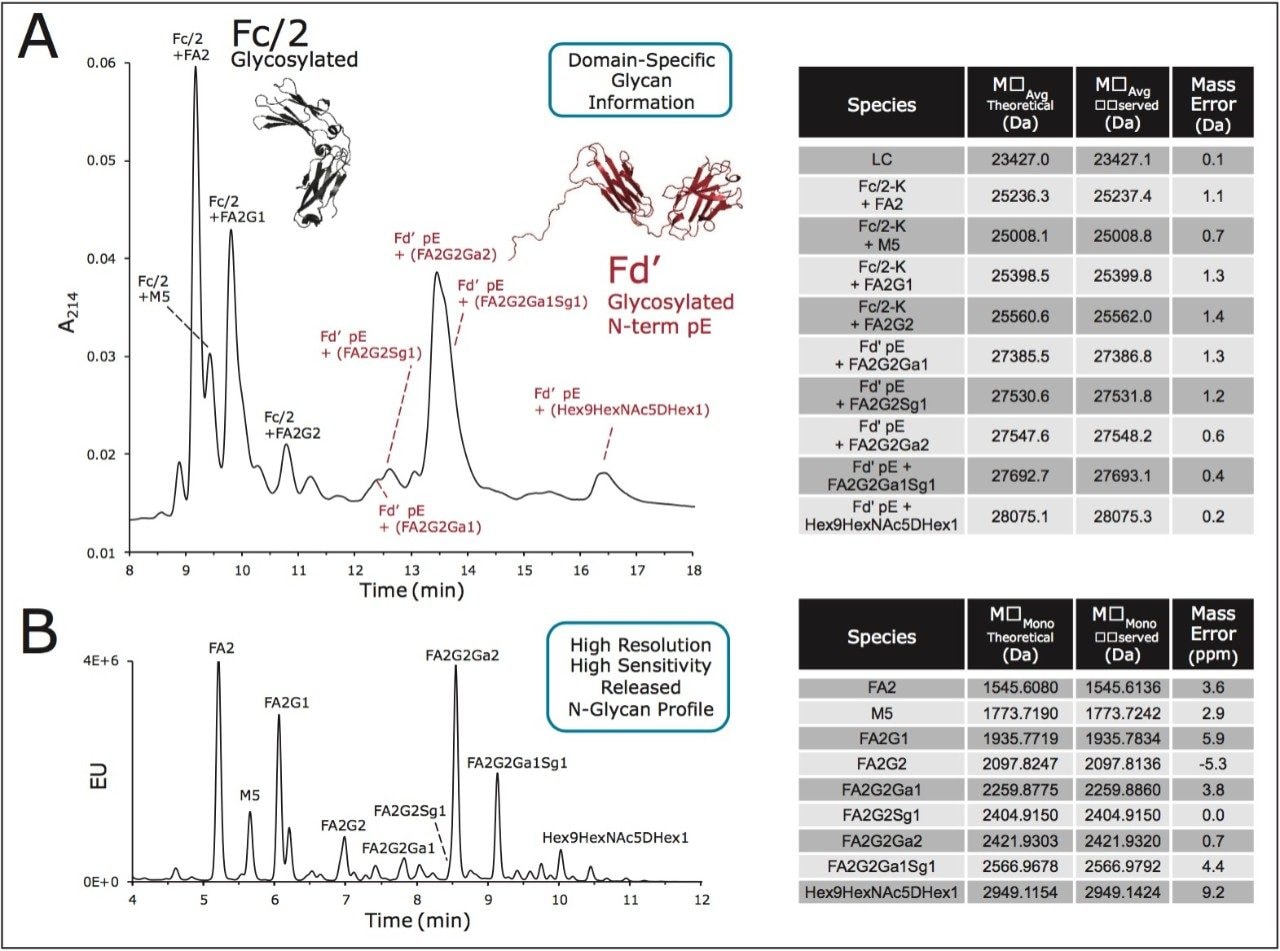
One of the key advantages to profiling IgG subunits by HILIC is being able to elucidate domain specific information about glycosylation. In an IgG structure, there exists one conserved N-glycosylation site at Asn297 of the heavy chain. As a consequence, most IgGs will be modified with two glycans in the CH2 domains (constant heavy chain 2 domains) of the Fc subunit. However, it is estimated that 20% of human IgGs are also modified in their CH1 domains, which reside in the Fab subunits, and more specifically the IdeS generated Fd' subunit.18-19 For example, it is known that cetuximab, a chimeric mAb expressed from a murine cell line, is glycosylated in both its CH1 and CH2 domains.20 Characterization of this mAb has thus proven to be an interesting case study for the application of our newly developed techniques. HILIC separations obtained for a reduced, IdeS digested sample of carboxypeptidase B treated cetuximab showed only one weakly retained subunit species, which could be easily assigned to the LC subunit by online ESI-MS (data not shown). Furthermore, and as shown in Figure 7A, the glycoform retention window for cetuximab was populated with twice as many peaks as had been observed for trastuzumab and its glycosylated Fc/2 subunit. Deconvoluted ESI-MS data from these HILIC-MS separations confirmed that the first grouping of peaks (labeled in gray) corresponded to Fc/2 glycoforms and typical mAb glycan species, such as FA2, FA2G1, M5, and FA2G2. Meanwhile, the second grouping of peaks were found to be distinctively related to glycoforms of the Fd' subunit given their unique masses. Curiously, each of the identified Fd’ glycoforms (labeled in red) are immunogenic in nature, containing either non-human alpha-1,3-galactose or non-human N-glycolylneuraminic acid epitopes.21
The identification of these glycan species has been confirmed through released N-glycan analyses. Using the newly developed GlycoWorks RapiFluor-MS N-Glycan Kit,22 cetuximab N-glycans were rapidly prepared and labeled with the novel fluorescence and MS-active labeling reagent, RapiFluor-MS. The resulting labeled N-glycans were subsequently separated using a glycan BEH amide, 130Å, 1.7 μm column and detected by fluorescence and positive ion mode ESI-MS, as portrayed in Figure 7B. The sensitivity gains afforded by the RapiFluor-MS label facilitated making confident assignments of the released N-glycan structures. The species that have been assigned as a result of both this released glycan analysis as well as the subunit HILIC-UV-MS method are supported by previous reports on cetuximab glycosylation.6,20
With the combination of released glycan and subunit-derived glycan information, cetuximab glycosylation has been characterized with significant detail. With the RapiFluor-MS released glycan analysis, a very high resolution separation has been achieved with an LC-MS compatible method in which glycans can even be subjected to detailed MS/MS analyses. With an equally MS-compatible subunit HILIC separation, domain-specific glycan information has been readily obtained with minimal sample preparation. Each method has therefore provided complementary information on the glycosylation of the mAb. Nevertheless, the widepore amide HILIC method stands out as a useful technique for rapidly screening mAbs for multidomain glycosylation.
Subunit analyses of mAbs represent a useful strategy for rapidly investigating domain-specific modifications. The combination of high fidelity IdeS proteolysis with high resolution LC-UV-MS has presented a new approach to mAb identity testing and assaying oxidation.3 The current subunit mapping strategies have exclusively relied upon reverse phase chromatography. However, since N-linked glycosylation of IgG proteins elicits dramatic changes in hydrophilicity and hydrogen bonding characteristics, a separation by hydrophilic interaction chromatography (HILIC) can be effectively used for this application or as a complementary method to reversed-phase separations since the same mobile phases can be employed. For this reason, we have proposed the use of HILIC with an amide bonded stationary phase that has been optimized for large molecule separations, the wide-pore glycoprotein BEH amide, 300Å, 1.7 μm stationary phase. Along with new developments in released N-glycan analysis afforded by RapiFluor-MS,22 the glycoprotein BEH amide, 300Å, 1.7 μm column enables new possibilities for routine monitoring and detailed characterization of mAb glycosylation, including elucidation of domain-specific glycan information.
720005385, July 2015