For forensic toxicology use only.
This is an Application Brief and does not contain a detailed Experimental section.
This application brief describes the application of a simple sample preparation method to allow for the screening of toxicologically-relevant compounds spiked into human urine or plasma using UPLC separation and Waters ACQUITY QDa Mass Detection.
In the second of a two-part series, we present a simple solid-phase extraction (SPE) protocol for the preparation of compounds in biological matrices and its use with the Waters STA screening method and the ACQUITY QDa Mass Detector.
The success of any LC-MS screening method is dependent on the use of a sample preparation protocol, which is efficient and robust, yet will extract as many analytes as possible from the matrix. Waters Oasis PRiME HLB has been designed to be simple and quick, eliminating the need for sorbent preconditioning and equilibration, while allowing for a more rapid workflow and cleaner extracts than traditional sample preparation solutions.
In this application brief, we describe the use of Oasis PRiME HLB in conjunction with UPLC separation and the ACQUITY QDa Mass Detector using the Waters STA screening application.1-3 Since its introduction over a decade ago, the associated library has been expanded to contain 1200 toxicologically-relevant substances. The purpose of the current work was to evaluate the feasibility of using the STA methodology with the ACQUITY QDa Mass Detector (Figure 1) to provide a simple, sensitive, low-cost qualitative screening tool for use in forensic toxicology laboratories for the determination of toxicologically-relevant substances in biological matrices.

Reference material for 100 analytes (Table 1) was obtained from Sigma-Aldrich (Poole, UK); typically, the individual drugs were supplied at a concentration of 1 mg/mL. Six different sources of urine were obtained from volunteers (Wilmslow, UK) and six different sources of plasma (collected using sodium fluoride/potassium oxalate tubes) were purchased from BIOIVT (West Sussex, UK).
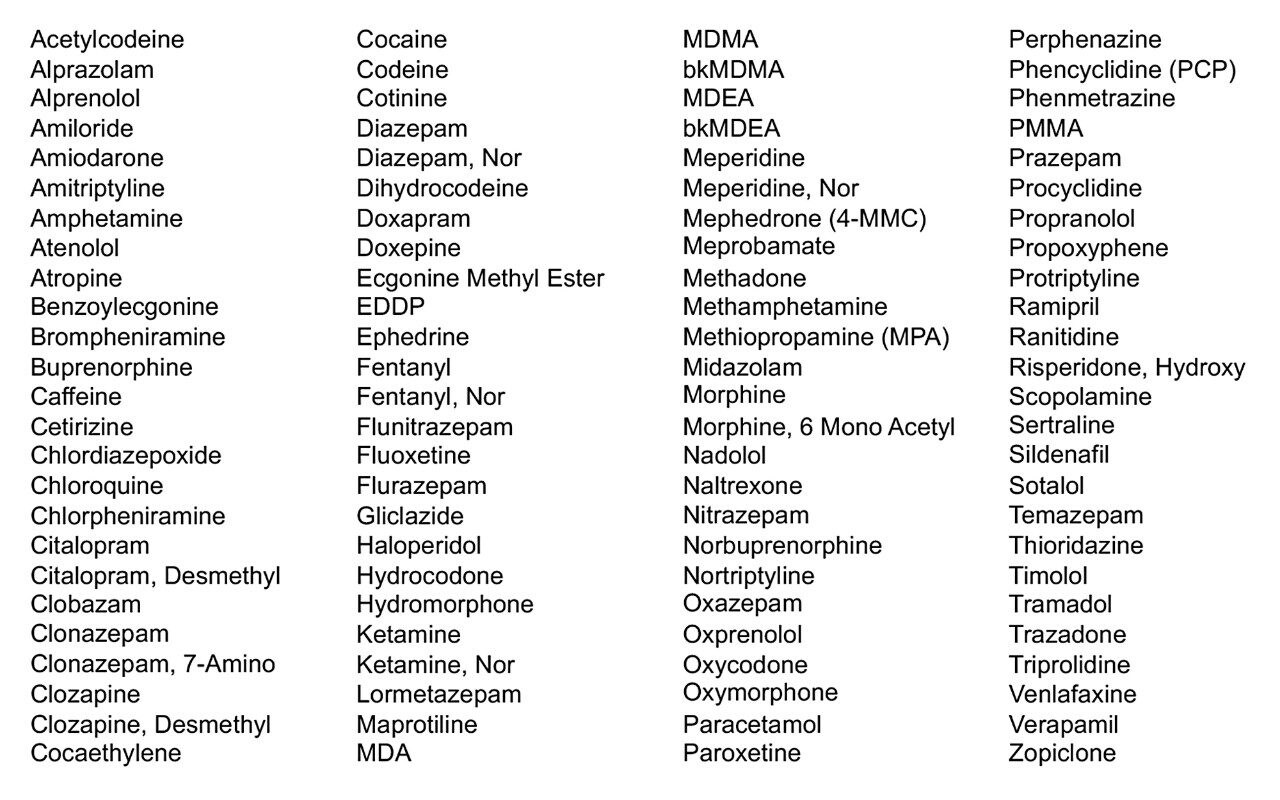
Analytes were combined into several mixtures (each mix containing a maximum of 10 analytes) to yield mixed spiking solutions at a concentration of 25 μg/mL in acetonitrile.
Each mixture was spiked individually into the blank matrices to give final concentrations of 200 and 500 ng/mL.
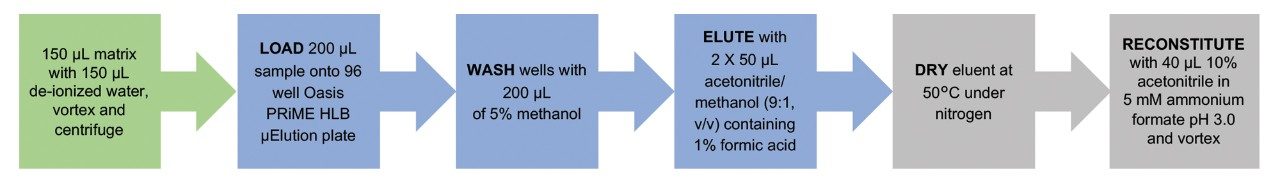
Spiked matrix (150 μL) was added to 150 μL de-ionized water and the sample was vortex-mixed for 60 sec and centrifuged. Two-hundred microliters of the spiked matrix/ water mix was loaded onto the Oasis PRiME HLB μElution plate (p/n: 186008052). After loading, the wells were washed with 200 μL of 5% methanol. The analytes were eluted with 2 × 50 μL of acetonitrile/methanol (90/10, v/v) containing 1% formic acid into a 800-μL roundwell collection plate (p/n: 186002481).
The samples were evaporated to dryness under a stream of nitrogen at 50 °C using a Porvair sample concentrator and reconstituted in 40 μL of 5 mM ammonium formate pH 3.0 containing 10% acetonitrile. The collection plate was covered with a Waters silicone/PTFE-treated cap mat and placed on a multi-tube vortex mixer for 3 min. Figure 2 shows a schematic that illustrates the extraction protocol used for sample preparation.
Samples were analyzed using the previouslydeveloped Waters STA screening method, which had been modified to be used in combination with the Waters ACQUITY UPLC H-Class PLUS System and ACQUITY QDa Mass Detector.²
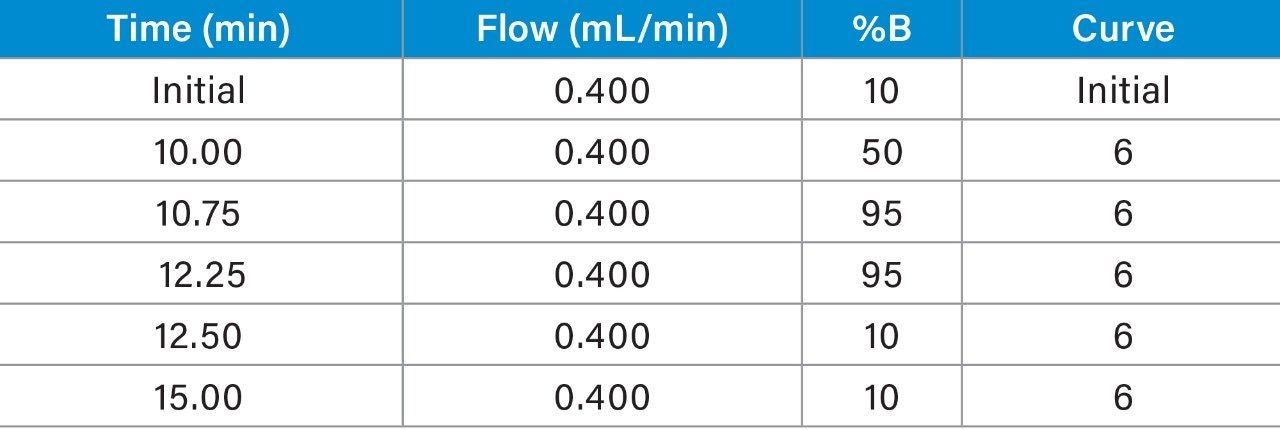
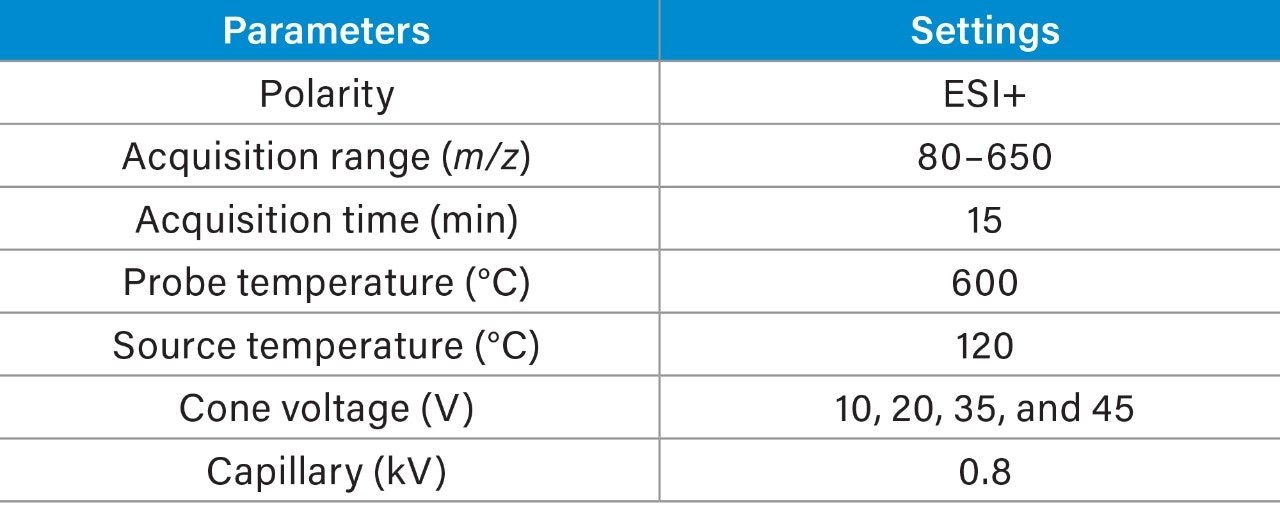
Chromatographic separation was achieved using a 15-min gradient elution. Data were acquired in full scan mode at multiple cone voltages, to generate spectral data in positive mode using in-source collision-induced dissociation (CID). Tables 2 and 3 list the LC conditions and the ACQUITY QDa settings respectively.
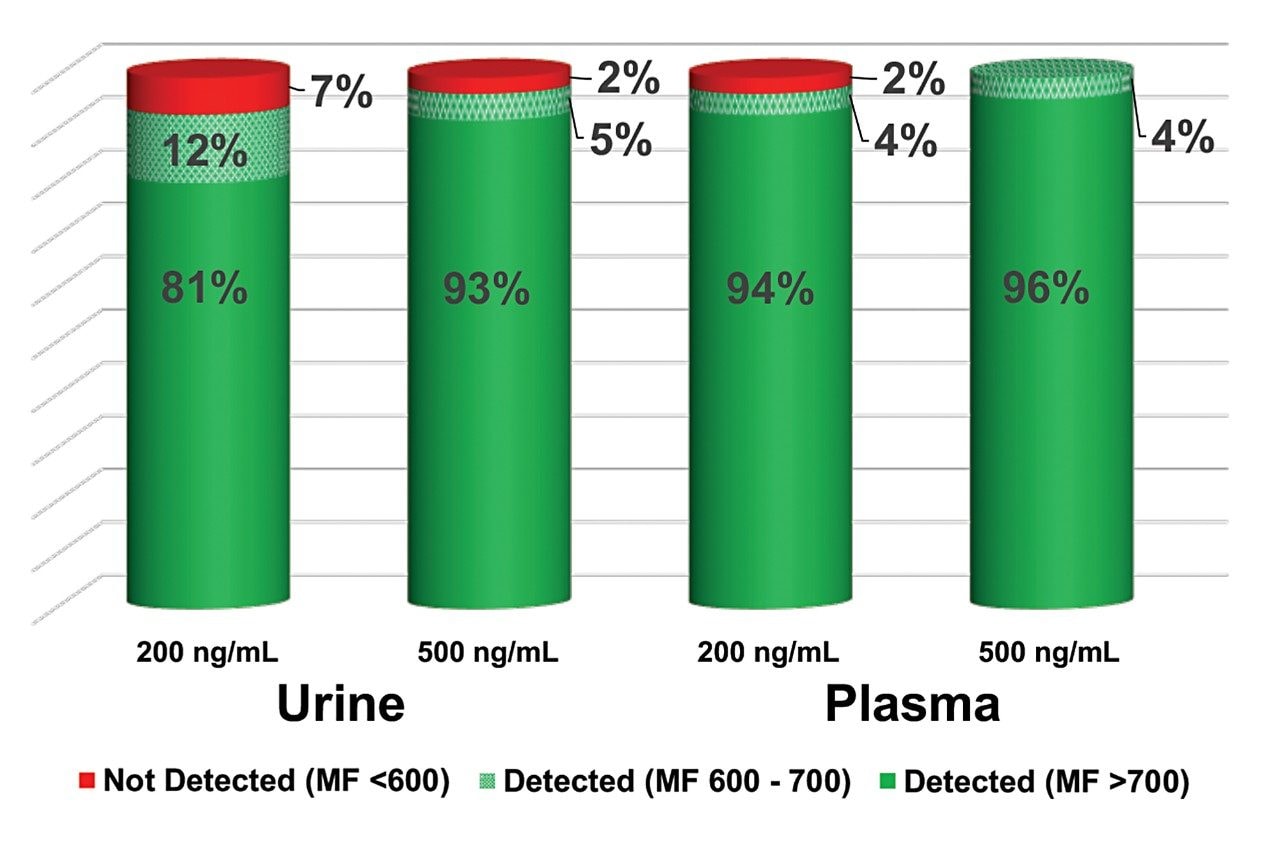
The spiked samples prepared by the described Oasis PRiME HLB μElution method were analyzed using the Waters STA screening method on an ACQUITY QDa Mass Detector. Acquired data was automatically processed using the ChromaLynx Application Manager (with MassLynx Software) and compared with a prepared library, which provides an identification through spectral library matching. The confidence with which a substance is identified is presented as an average match factor, which has a maximum value of 1000. The average match factor is determined by comparing the acquired and library spectra for each of the four cone voltages. In this study, putative positive identifications had an average match factor greater than 700, while compounds with an average match factor of between 600 and 700 were classed as tentative identifications. In addition, retention time needed to be within 0.35 min of the reference, as specified in the Waters STA library.
In this study, compounds were categorized as detected if they were tentatively or positively identified. At 200 ng/mL, 93% of the investigated analytes could be detected in urine and 98% in plasma. Figure 3 summarizes the total number of detections at 200 and 500 ng/mL in both matrices.
In this technology brief, a simple sample preparation method using Oasis PRiME HLB μElution in conjunction with UPLC separation and ACQUITY QDa Mass Detection has been shown to provide a quick, simple, and effective way to screen for toxicologically-relevant compounds in human urine and plasma.
The use of Oasis PRiME HLB μElution in a 96-well plate format will allow for the sample preparation process to be automated and thus increase sample throughput.
The ACQUITY QDa is a low-cost, sensitive, and versatile instrument that can be used to successively detect toxicologically-relevant compounds in biological matrices in combination with UPLC chromatographic separation.
720006726, January 2020