An analyst must take a holistic approach to optimize MS quality in an IEX-MS analysis. Considerations need to be made around mobile phases, labware, and reagent quality at all levels. The use of IonHance CX-MS buffers, the substitution of non-volatile salts during sample preparation steps, and the replacement of glass for certified thermoplastics can lead to substantive improvements in spectral quality. With this work, we will demonstrate the benefits of using MS-grade reagents, certified plasticware, and tailored sample preparation techniques.
MS cleanliness is a surprisingly overlooked aspect of protein LC-MS analyses. Leaching from containers, for instance, is an insidious problem that can be too frequently encountered even when sourcing MS-certified reagents and materials. Unwanted metals such as sodium, potassium, and calcium can easily undermine the capabilities of an assay. One reason to be scrupulous about cleanliness is the ability to minimize salt adducts that can cause isotopic overlaps and the broadening of each charge state.1 Beyond this, metal contaminants lead to the loss of resolution, sensitivity, and mass accuracy.2-4 The need for MS cleanliness is even more obvious when performing native mass spectrometry as more adduction occurs with low charge state ions.5
First, reagents such as ammonium acetate need to be of the highest quality, and ideally any MS-grade reagents would be housed, measured, and transferred from and into clean materials. Trace metal certified thermoplastics should thus be used whenever and wherever possible. Glass, unfortunately, has significant levels of metal ions that readily leach into a sample, especially if the glass is subjected to high pH solutions.6 While some borosilicate glass gives minimal leaching, a general avoidance of glass mobile phase bottles, glass autosampler vials, and glass graduated cylinders can pay dividends for eliminating salt adducts in MS data. On the other hand, thermoplastics can leach plasticizers (e.g., phthalates), so they too should be certified for quality purposes.
Understanding that it can be daunting to establish new LC-MS techniques, such as IEX-MS, we have collated best practices to achieve high quality MS data. With this work, we will demonstrate the benefits of using MS-grade reagents, certified plasticware, and tailored sample preparation techniques.
200 μg of NIST mAb was split into two 100 μg aliquots and digested by incubating at 37 °C for 30 min with 100 units of FabRICATOR enzyme (Genovis, A0-FR1-008) in either 105 mM ammonium acetate pH 6.2 or phosphate buffered saline (PBS). The final concentration of digested antibody aliquots was 1.0 mg/mL.
|
LC system: |
ACQUITY UPLC I-Class PLUS |
|
|
Detectors: |
ACQUITY TUV ACQUITY RDa |
|
|
LC column: |
BioResolve SCX mAb, 3 μm, 2.1 × 50 mm |
|
|
Column temp.: |
30 °C |
|
|
Sample vial: |
Polypropylene Screw Neck Vials (p/n: WAT094172) or Glass Screw Neck Total Recovery Vials (p/n: 600000671cv) |
|
|
Mobile phase A: |
IonHance CX-MS pH Buffer A, 10 mM ammonium acetate, pH 5.0 (created from a 50x dilution of CX-MS pH Concentrate A [p/n: 186009280]) |
|
|
Mobile phase B: |
IonHance CX-MS pH Buffer B, 75 mM ammonium acetate, pH 8.5 (created from a 21x dilution of CX-MS pH Concentrate B [p/n: 186009281]) |
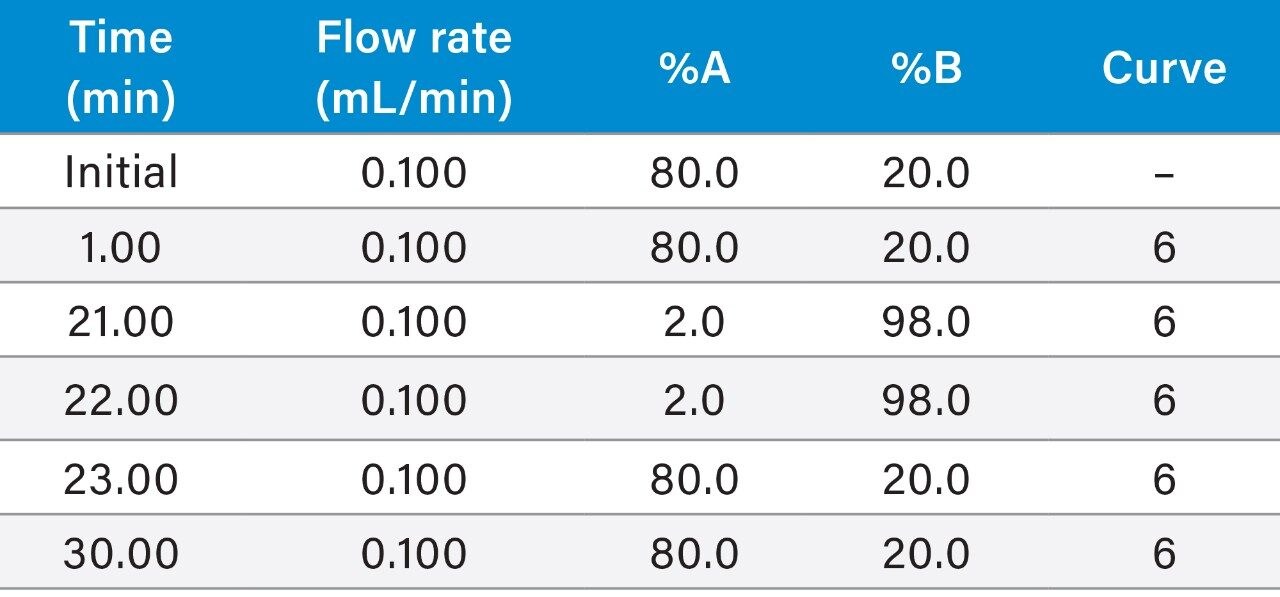
|
Mass range: |
m/z 400–7000 |
|
Mode: |
ESI+ |
|
Cone voltage: |
150 V |
|
Desolvation temp.: |
350 °C |
|
Capillary voltage: |
1.5 kV |
|
Lock mass: |
Leu-enkephalin at 50 fmol/μL in 50/50 water/acetonitrile with 0.1% formic acid |
|
Informatics: |
UNIFI Scientific Information System |
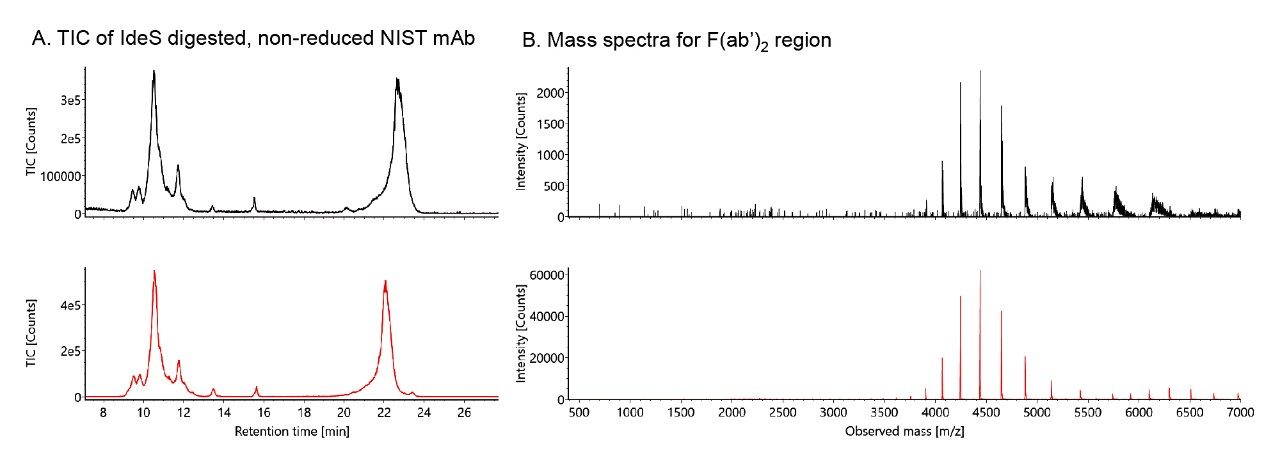
The ability to generate robust IEX separations does not necessarily equate to achieving high quality mass spectra. Figure 1 shows two nearly identical chromatograms of IdeS digested, non-reduced NIST mAb. Despite their comparable chromatographic profiles, these two runs produced mass spectra of considerably different quality. To obtain spectra with high signal-to-noise and low adduct levels, one must make special considerations throughout the entire sample and LC-MS workflow. For simplicity, MS settings were not considered for this application, however, source conditions can have significant effects on MS quality. Optimizing source and desolvation temperatures, as well as cone and desolvation gas flows, are parameters we have found important for sensitive native proteomics.
Various iterations of IEX-MS were performed with a BioResolve SCX mAb Column and IdeS digested, non-reduced NIST mAb, where the objective was to determine the factors of major influence to MS quality and molecular weight calculations. These experiments started with mobile phases prepared from IonHance CX-MS pH Concentrates as they are independently confirmed by ICP-MS to contain low ppb levels of metals and are packaged in trace-metal certified low-density polyethylene (LDPE) bottles with available certificate of analysis documentation.
To validate the use of thermoplastic mobile phase bottle for IEX-MS, we tested identically prepared IonHance CX-MS buffers housed in either glass or Waters MS-Certified LDPE mobile phase bottles. The results indicate that the use of plasticware reduced the intensity of +21 Da sodium adducts (Figure 2). This improvement was observed despite the mobile phase being housed inside the glassware for only a short amount of time (<24 hr); continued incubation would presumably lead to additional leaching and a further increase in adduct levels. Nevertheless, mass spectra obtained with LDPE mobile phase containers were still dissatisfactory. Inspection of the resultant mass spectra continued to show a preponderance of sodium adducts. In the experimental setup leading to this result, mobile phases were carefully prepared, but no additional considerations were made for performing the IdeS digestion or desalting the sample. Indeed, the NIST mAb sample was digested in phosphate buffered saline (PBS) and housed inside glass autosampler vials.
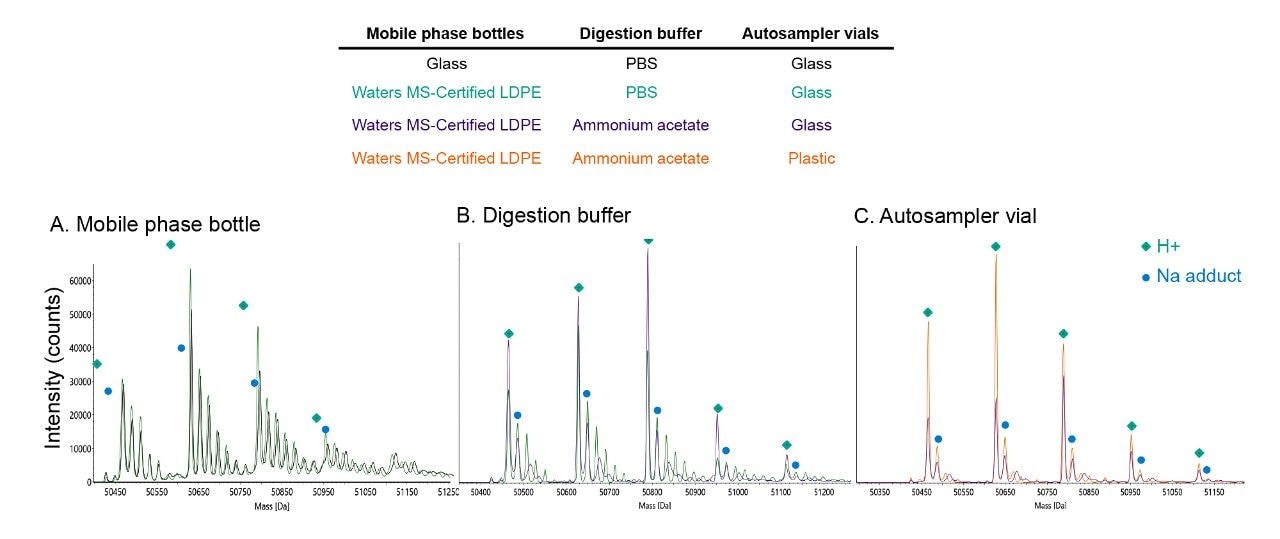
Concerned by these unsatisfactory results, it was considered if sodium from the digestion buffer might be contaminating the MS analysis. It was hypothesized that an IdeS digestion might be sufficiently controlled with a neutral ammonium acetate solution. By digesting the sample in MS-grade ammonium acetate, the column would be subjected to significantly less sodium during sample injection. Figure 2 shows deconvoluted mass spectra obtained from this type of sample preparation, and a marked improvement in spectral quality can be seen. While the formulation buffer was not removed, it was significantly diluted. Presumably, improved results could likewise be achieved by buffer exchanging the sample into the starting mobile phase of the separation. However, the technique presented here provides a pragmatic approach for quick improvement. Overall, it might be important to reduce the amount of sodium injected onto the column. Because sodium has high affinity for cation-exchange ligands, excess loads of the cation might cause lingering contamination and challenges for re-equilibration.
Further improvements to adducts levels were found upon exchanging the sample vial from glass to plastic (Figure 2). Like in previous examples, peaks became sharper and the relative abundance of the protonated ions increased, including that of the (Fc/2)2 subunit. The scale of improvement is likely a function of the high surface area to volume ratio that exists for a sample aliquot in an autosampler vial.
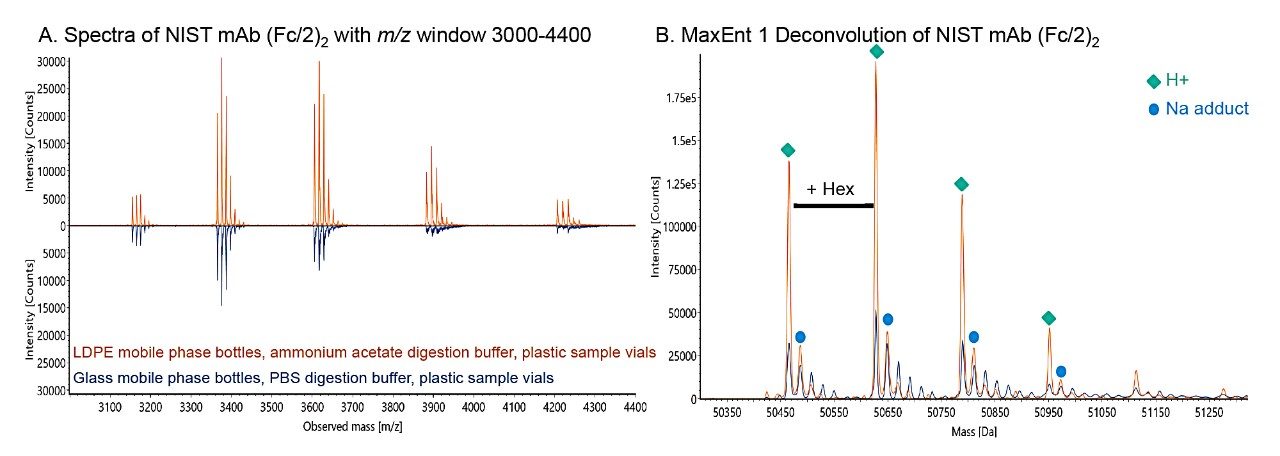
Figure 3 presents a final mass spectral and MaxEnt1 deconvolution comparison between a sample using glassware and digested in PBS compared to a sample using plasticware and digested in MS-grade ammonium acetate. Mass spectra quality were significantly improved with plasticware and digestion buffer substitution to ammonium acetate.
Finally, while optimizing an IEX-MS method, it is worthwhile to consider the use of a divert valve. Upon using such, it is possible to keep sample matrix away from the mass spectrometer and thereby minimizing source contamination.7 Diverting the flow is particularly useful if a formulation buffer contains non-volatile reagents such as sodium or phosphate. These and other sample matrix components can be made to pass through the column and out to waste via the divert valve. In practice, the timing of the flow switching will need to be optimized, and it will remain necessary to use plasticware and to avoid non-volatile salts in order to achieve the best IEX-MS results.
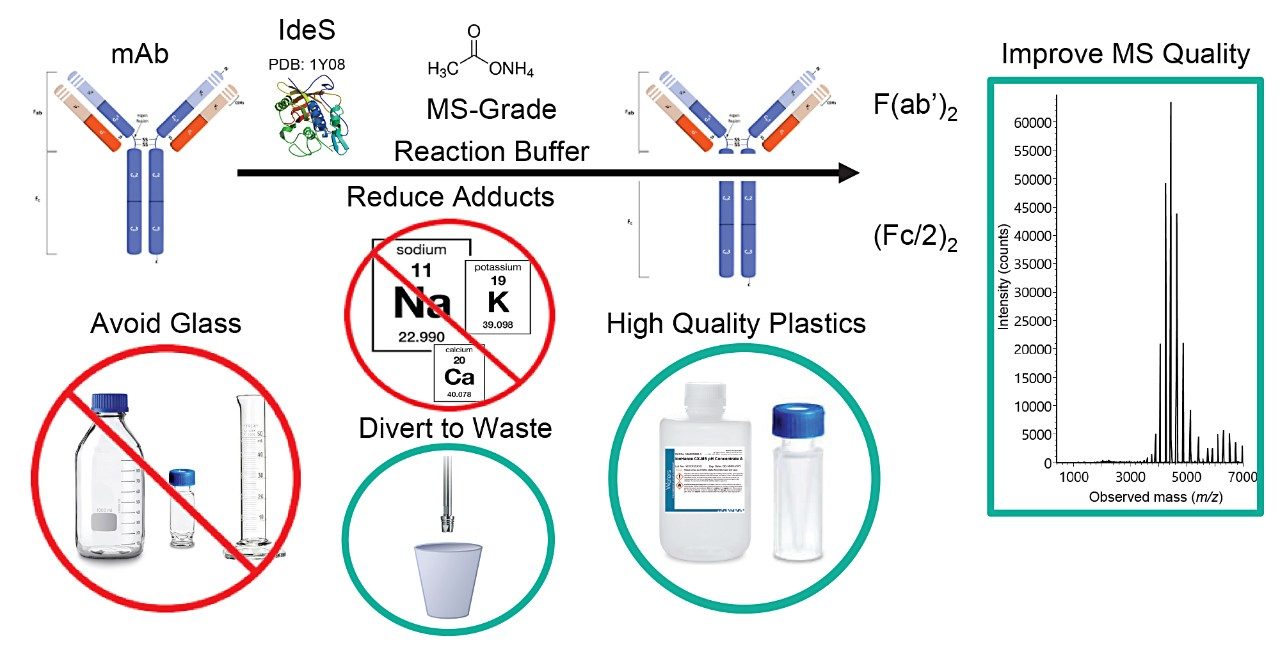
An analyst must take a holistic approach to optimize MS quality in an IEX-MS analysis. Considerations need to be made around mobile phases, labware, and reagent quality at all levels. The use of IonHance CX-MS buffers, the substitution of non-volatile salts during sample preparation steps, and the replacement of glass for certified thermoplastics can lead to substantive improvements in spectral quality. As depicted in Figure 4, these learnings form a blueprint for improved MS-hygiene and some specific practices that are of benefit to IEX-MS.
720006675, January 2020