
This application note focuses on the selection of appropriate column chemistries to separate eighteen polymer additives routinely monitored in extractables and leachables (E&L) workflows. Often an analyst will select columns for new work based on past experience or a literature search.
For novice chromatographers, method development in high performance liquid chromatography (HPLC) can be a daunting task. With the large number of method development approaches and columns to choose from, knowing where to start and how to achieve the best results can be difficult. This difficulty is magnified when working with complex samples containing analytes with a wide range of chemical properties. Regardless of the inherent complexity, method development can be simplified to basic steps which may be adapted for different types of assays. For instance, the steps needed to develop a separation method for the analysis of pharmaceutical degradation products are similar to those for the analysis of extracted polymer samples. While these two examples may have different end goals, the steps of method development are similar, and require only minor changes to achieve the desired results.
This application note focuses on the selection of appropriate column chemistries to separate eighteen polymer additives routinely monitored in extractables and leachables (E&L) workflows. Often an analyst will select columns for new work based on past experience or a literature search. However, another approach is to screen a group of columns that covers a wide range of selectivities, then select the column that best meets the separation goals. Understanding the differences in the column chemistries available and selecting the most appropriate columns to screen for a given assay can not only speed up method development but can also lead to better separations.
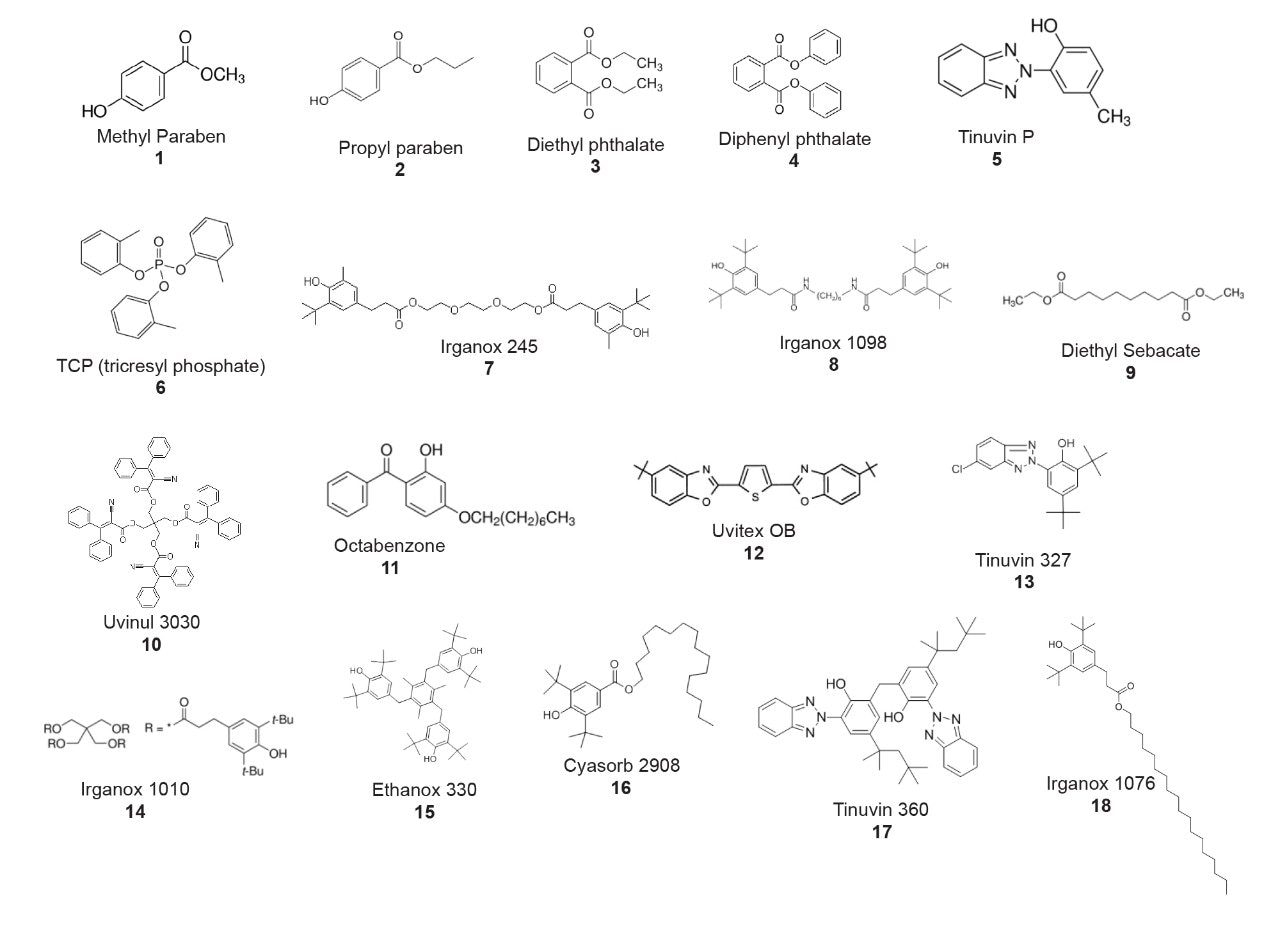
Standards of each analyte were purchased, and a mixture was created as outlined below. These compounds are present in the Waters Extractables and Leachables Standard (p/n: 186008063).1 However, the Extractables and Leachables Standard is intended for mass spectrometry (MS) detection and is not applicable for this work, which utilized ultra-violet (UV) detection. Therefore, a more concentrated sample was created to ensure good UV detection. The following analytes were dissolved in methanol at the indicated concentrations: Irganox 1010 (0.1 mg/mL), Irganox 245 (0.075 mg/mL), Irganox 1076 (0.5 mg/mL), Irganox 1098 (0.075 mg/mL), Cyasorb 2908 (0.15 mg/mL), Uvinul 3030 (0.05 mg/mL), propyl paraben (0.075 mg/mL), Ethanox 330 (0.05 mg/mL), Tinuvin 327 (0.025 mg/mL), diethyl phthalate (0.05 mg/mL), diphenyl phthalate (0.05 mg/mL), TCP (0.075 mg/mL), octabenzone (0.05 mg/mL), Tinuvin P (0.025 mg/mL), Uvitex OB (0.075 mg/mL), dibutyl sebecate (2.0 mg/mL), methyl paraben (0.075 mg/mL), and Tinuvin 360 (0.075 mg/mL). The chemical structures of these analytes are shown in Figure 1.
|
Instruments: |
ACQUITY UPLC H-Class with 6 column positions (Column Manager and 2 Column Manager Auxiliaries) and PDA Detector |
|
Data management: |
MassLynx Software |
|
Columns: |
All columns were 2.1 × 50 mm 1.x μm. See Figure 3 for column selection. |
|
Mobile phase A: |
Water with 0.1% formic acid |
|
Mobile phase B: |
Acetonitrile with 0.1% formic acid |
|
Flow rate: |
0.5 mL/min |
|
Gradient: |
Initial conditions of 10% B, linear ramp to 100% B in 6.85 minutes, hold for 4.92 minutes. Return to starting conditions and hold at 10% B for 2.66 minutes. Total run time: 14.45 minutes. |
|
PDA detection: |
220 nm |
Many method development strategies recommend screening 3–5 columns with varying ligand and particle types in order to provide the best starting point for new methods.2-5 The United States Pharmacopeia (USP) has different designations for HPLC columns, called L#, which are assigned based on the chemical and physical properties of the stationary phase. For instance, an L1 designation is given to columns with C18 ligands bonded to porous or non-porous silica or ceramic particles 1.5 to 10 μm in diameter. The L1 designation includes columns such as ACQUITY BEH C18, ACQUITY HSS C18, and Symmetry C18. There are many different L designations, most with a variety of different brands to choose from, making the selection of the right columns to screen difficult. The column choice can be narrowed down by selecting those appropriate for the analytes being tested. For instance, L3 columns (porous silica with no bonded ligand) are typically used for normal phase separations of polar analytes, and may not be suitable for the analysis of plastic additives, which are moderately to highly hydrophobic. For these analytes, reversed-phase chromatography using an L1 column would be a better choice. Other suitable choices include L7 (C8 ligand bonded to a silica particle) and L11 (phenyl ligand bonded to a silica particle) columns. The lower hydrophobic retention of the C8 and Phenyl Columns may elute the more hydrophobic plastic additives faster, reducing the overall analysis time. The Phenyl Column also offers different selectivity, particularly for analytes containing multiple pi bonds, which interact in unique ways with the stationary phase when certain mobile phases are used.
In the example described here, a method was developed for separating eighteen plastic additives. Five columns were selected and screened to cover a range of selectivities while still achieving a reasonable time for method development (Figure 2). Each column was chosen specifically for the mixture of analytes being tested as each has a distinct advantage compared to the other columns. When used together, this panel of columns offers a wide range of selectivities which provide a good starting point for method development. The first column chosen, an L1-designated BEH (ethylene-bridged hybrid) C18 is a good general-purpose column which has been used in many analyses. This stationary phase contains a C18 ligand attached to a fully porous hybrid silica particle, which is stable across a wide pH and temperature range, allowing considerable flexibility in method development. The next column, CSH (charged surface hybrid) C18 is also designated as an L1 column. However, the CSH Column is based on a positively charged hybrid silica particle which provides better peak shapes for basic analytes at low pH.6,7 The next column selected was the L7-designated CORTECS C8, which uses a solid-core or superficially porous silica particle. The solid-core particle allows CORTECS Columns to achieve higher efficiencies compared to fully porous particles.8 The fourth column selected was an L11-designated CORTECS Phenyl, which uses the same superficially porous particle as the CORTECS C8 but has a phenyl-hexyl ligand attached. As mentioned previously, for moderately to highly hydrophobic analytes, the decreased retentivity of the C8 and phenyl-hexyl columns may prove to be advantageous. Lastly, to maximize the overall selectivity of the panel we selected the L43- designated HSS (high strength silica) PFP column, containing a pentafluorophenyl ligand attached to a fully porous silica particle. The PFP ligand allows several secondary interactions to take place in addition to the hydrophobic interactions. By screening all the columns shown as part of a method development process different separations can be achieved.
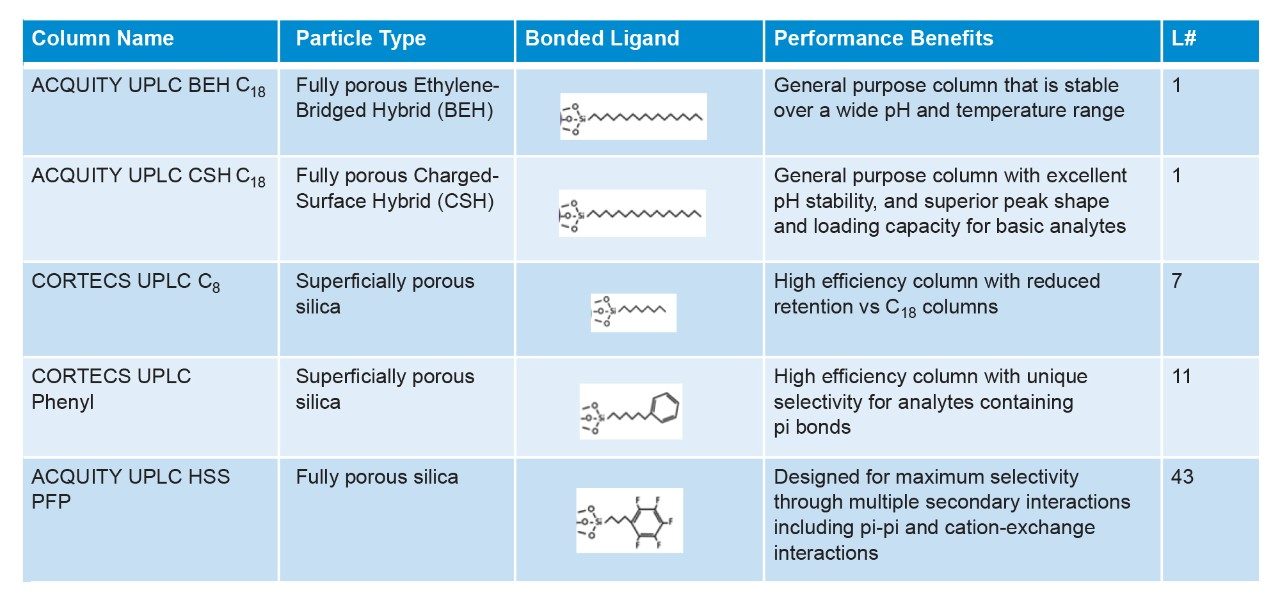
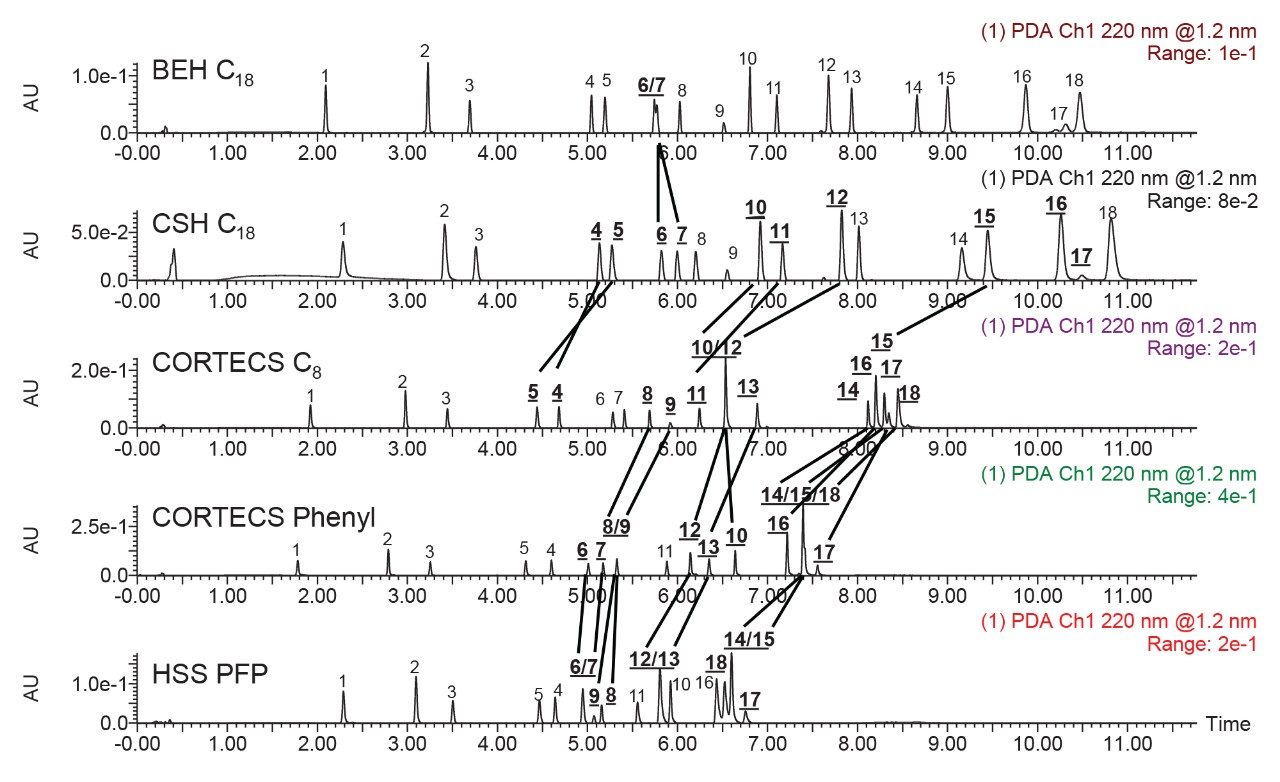
Figure 3 shows the chromatograms obtained using the five columns and an acetonitrile gradient. The use of five stationary phases has led to different chromatographic results. For instance, comparing the separations obtained using the two C18 columns, components 6 and 7 (TCP and Irganox 245) were not well separated on the BEH C18 column, but were baseline resolved on the CSH C18 Column. This demonstrates the selectivity of the base particle separate from the bonded ligand. In looking at the separations achieved using the CORTECS C8 and CORTECS Phenyl Columns, there are also significant differences. The cluster of analytes 14, 15, and 18 co-eluted when the CORTECS Phenyl Column was used but were separated on the CORTECS C8 Column. However, the C8 column did not separate components 10 and 12. These examples speak to the need for screening multiple types of bonded ligand, even if they are on the same base particle. For this mixture, the best separation was achieved using an ACQUITY CSH C18 Column. Column screening is only the first step in method development. The screening of different mobile phase conditions including different strong solvents and different additives provide additional options for optimizing separations.2-4
Screening multiple columns is highly recommended during HPLC method development. Appropriate column chemistries should be selected based on the analytes of interest. Using columns packed with stationary phases made from different base particles and bonded ligands can provide a range of selectivities for the separation, leading to a better method. This application note focused on the importance of column screening for developing a method to separate a mixture of plastic additives. Five column chemistrieswere evaluated to separate a mixture of eighteen analytes. In most cases, the mixture was well separated except for a few co-eluting analytes. For this application, a CSH C18 Column gave the best separation, with baseline resolution of all analytes within 11 minutes. Selecting appropriate column chemistries to screen allowed the identification of the best separation conditions with just five analytical runs. Knowing the types of analytes being tested, and choosing the columns based on that information provides a good starting point for all method development activities.
720006777, March 2020