This work provides a single, simple method for the simultaneous, direct quantification of intact human insulin and multiple insulin analogs in human plasma.
Insulin is perhaps one of the best known and earliest peptide therapeutics. Multiple long and fast-acting analogs have also been developed, and a patient may often be prescribed one of each for diabetes control. Quantification of biologics, such as insulins, has historically been performed using ligand binding assays (LBAs) such as ELISAS. LC-MS/MS, however, has certain advantages over LBAs, such as shorter development times, higher accuracy and precision, the ability to multiplex, no cross-reactivity, and the ability to readily distinguish between closely related insulins. Intact insulins are particularly difficult to analyze by LC-MS/MS, as MS sensitivity is low due to poor transfer into the gas phase, and poor fragmentation patterns exist due to the presence of multiple stabilizing disulfide bonds. In addition, insulin and its analogs suffer from non-specific binding and poor solubility, making LC and sample preparation method development difficult. A few LC-MS/MS methods do exist; however, most of those methods involve time-consuming and laborious immunoaffinity purification and/or nano-flow LC. Distinguishing between human insulin and insulin lispro (Humalog) is a very specific challenge for quantifying insulins, as they differ by a simple reversal in the position of two amino acids. Only a single low-molecular weight fragment differentiates the two, making selective sample preparation and chromatography critical.
This study takes advantage of mixed-mode solid-phase extraction (SPE) and a high-efficiency, solid-core particle column that contains a low-level positive surface charge to eliminate interferences while facilitating high sensitivity quantification. Furthermore, selectivity studies show that the presence of high levels of human insulin, such as one might encounter in type II diabetic patients, does not interfere with quantification of lispro or any of the other analogs. This work provides a single, simple method for the simultaneous, direct quantification of intact human insulin and multiple insulin analogs in human plasma (Figure 1), achieving LODs of 50 to 200 pg/mL for each. Average accuracy for standard curve points was 99% to 100%. Average inter- and intra-day accuracies for QC samples were 98% and 94%, respectively. Average inter- and intra-day precisions for QC samples were 7.5% and 5.3%, respectively. Matrix factors for all analogs were calculated in six sources of human plasma and CVs of matrix factors were <15% in all cases, further supporting the selectivity of the method.
25 µL of bovine insulin (internal standard, final concentration 10 ng/mL) was added to 250 µL human plasma and mixed.
Samples were precipitated with 250 µL 1:1 methanol/acetonitrile containing 1% acetic acid, and centrifuged for 10 minutes at 13,000 rcf.
The supernatant was transferred to a 2-mL 96-well plate containing 900 µL of 5% concentrated NH4OH in water (v:v) and mixed.
|
Condition: |
200 μL methanol |
|
Equilibrate: |
200 μL water |
|
Load sample: |
Entire diluted PPT supernatant was loaded onto the extraction plate in two steps of approximately 700 μL each |
|
Wash: |
200 μL 5% NH4OH in water |
|
Wash: |
200 μL 5% methanol + 1% acetic acid |
|
Elute: |
2 × 25 μL 60:30:10 methanol/water/ acetic acid |
|
Dilute: |
50 μL water |
|
Inject: |
30 μL |
|
System: |
ACQUITY UPLC I-Class with 2D Technology, configured for at-column dilution with trap and back elution |
|
Analytical column: |
CORTECS UPLC C18+ 1.6 μm, 2.1 × 50 mm (p/n 186007114)* |
|
Trap column: |
XBridge C18 IS 3.5 μm, 2.1 × 20 mm (p/n 186003019) |
|
Elution mobile phase A: |
0.1% HCOOH in water |
|
Elution mobile phase B: |
0.1% HCOOH in acetonitrile |
|
Loading solvent: |
85:15 mobile-phase A:B; 0.1 mL/min for first two minutes, reverse valve, then clean trap column with ramp from 40% to 90% B |
|
Dilution solvent: |
100% mobile-phase A, 0.3 mL/min for first two minutes |
|
Valve position: |
Initial position one (forward loading of trap), switch to position two at two minutes (back elute of trap onto analytical column), back to position one at 6.5 minutes |
|
Gradient: |
Load for two minutes; switch valve and back elute from trap column onto analytical column with a linear gradient from 15% to 40% B over four minutes |
|
Elution flow rate: |
0.25 mL/min |
|
Column temp.: |
60 °C |
|
Sample temp.: |
15 °C |
|
Injection volume: |
30 μL |
|
Run time: |
8.5 minutes |
|
Collection plates: |
Waters 1-mL ACQUITY collection plates |
|
Mass spectrometer: |
Xevo TQ-S |
|
Ionization mode: |
ESI positive |
|
Capillary voltage: |
3.0 kV |
|
Desolvation temp.: |
600 °C |
|
Cone gas flow: |
150 L/h |
|
Desolvation gas flow: |
1000 L/h |
|
Collision cell pressure: |
3.8 × 10 (-3) mbar |
|
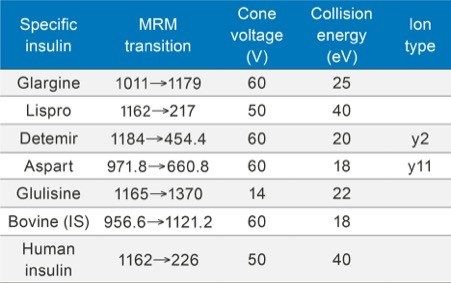
Collision energy: |
Optimized by component, see Table 1 |
|
Cone voltage: |
Optimized by component, see Table 1 |
|
Chromatography software: |
MassLynx |
|
Quantification software: |
TargetLynx |
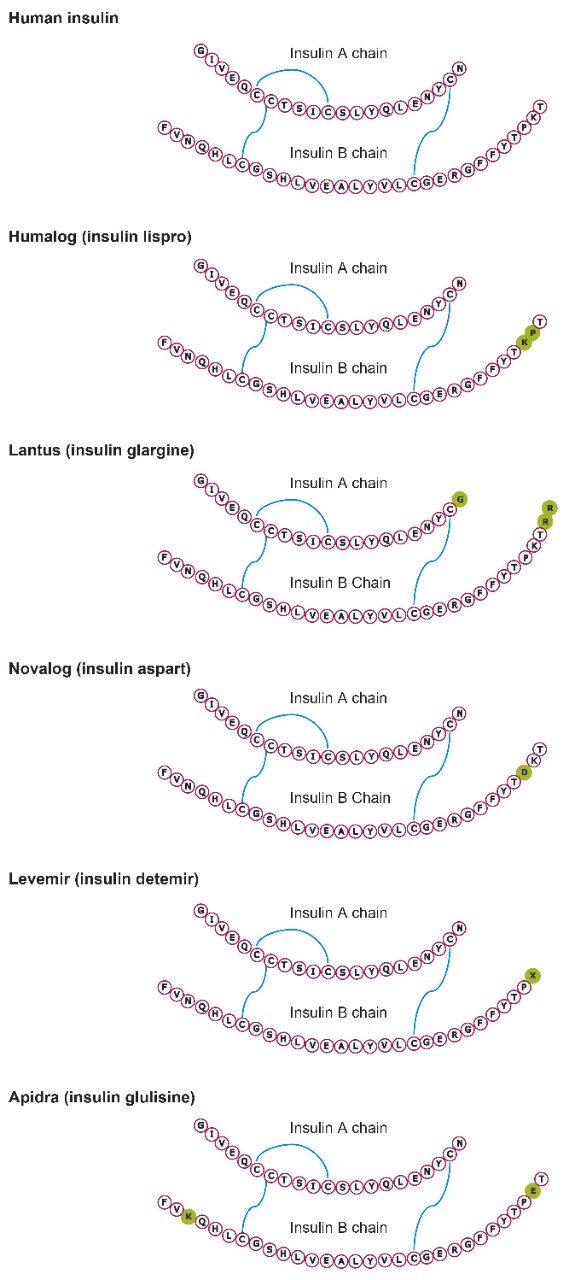
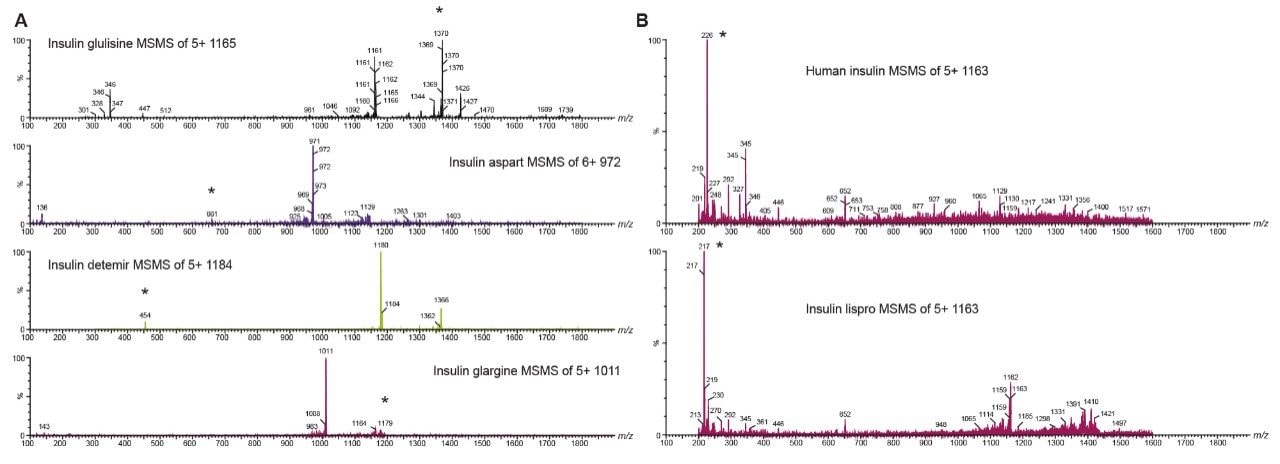
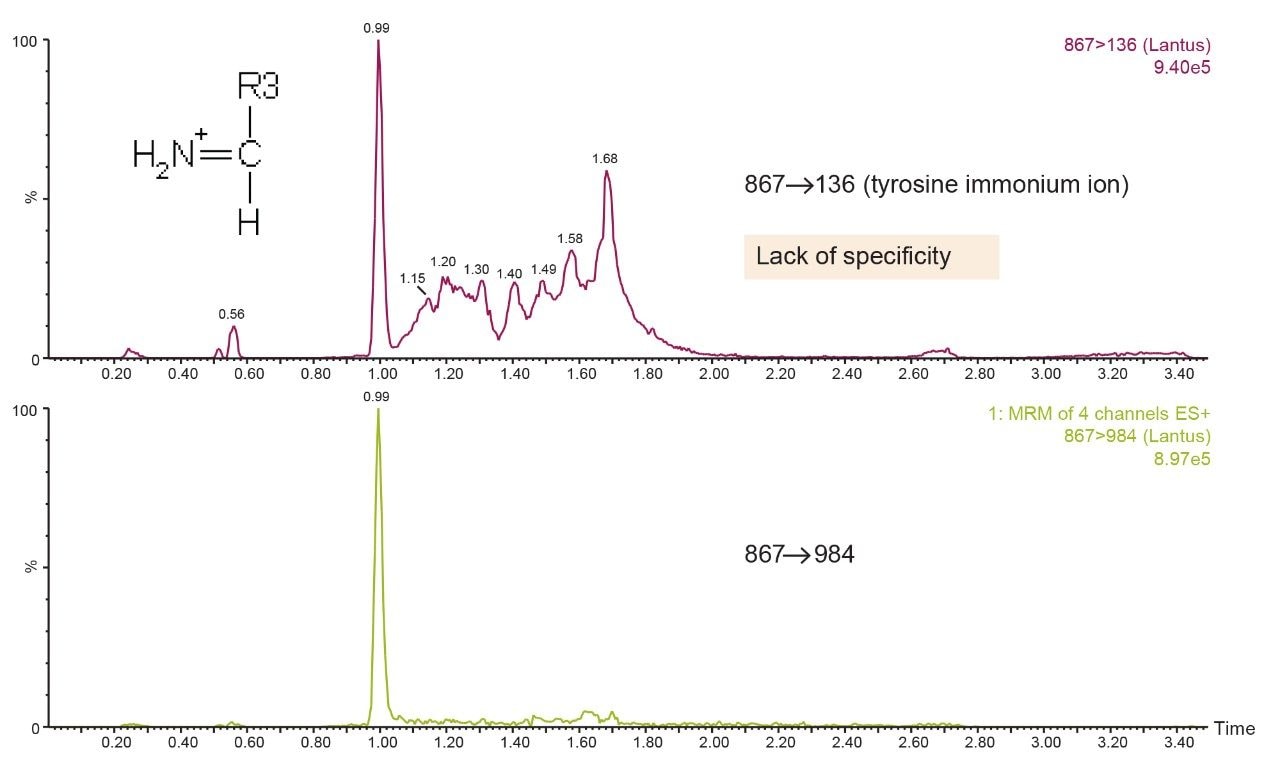
Several multiple charged precursors were observed for each of the analogs, typically the two most abundant were selected for CID, and either one or two diagnostic fragments monitored during method development. Representative MS/MS spectra at the optimal collision energies for the primary transitions chosen for quantification are shown in Figures 2A and 2B. In several cases, higher intensity MRM transitions existed than the one that was ultimately chosen. Specifically, most insulins will produce very intense immonium ion fragments. Glargine, for example, produces a tyrosine immonium ion fragment at m/z 136. However, tests in extracted plasma demonstrated that the use of higher m/z precursor and fragment pairs (Table 1) yielded significantly improved specificity in matrix, outweighing any apparent sensitivity difference (Figure 3). This facilitates the use of analytical scale LC and traditional SPE methodologies versus nano-scale chromatography and affinity-based purification schemes previously reported. Human insulin and lispro, however, share almost complete overlap in their fragmentation patterns due to a simple reversal in the positions of amino acids 28 and 29 in the B chain (Figure 1). A single low-molecular weight fragment differentiates the two, with human insulin yielding a fragment at m/z 226 and lispro producing a fragment at m/z 217 (Figure 2B), both arising from the last two amino acids in the B chain. Without baseline chromatographic separation, the MS must rely on these non-specific fragments for quantification, making both selective sample preparation and high efficiency chromatography crucial.
This section details the methodology for column conditioning and optimization which uses a simple 1D gradient method. For best insulin peak widths and area counts, the CORTECS C18+ Column must be conditioned prior to use. Injections of the supernatant from precipitated rat plasma have been shown to effectively condition the CORTECS C18+ Column for the analysis of insulin and its analogues under reverse-phase conditions.
Injections are made using a 2 minutes linear gradient from 20%B to 65%B with a ramp to 98%B in 0.1 minutes and a hold time of 0.5 minutes. Re-equilibrate back to 20%B in 0.2 minutes and hold for 1.8 minutes before the next injection. Mobile-phase A is 0.1% formic acid in water. Mobile-phase B is 0.1% formic acid in acetonitrile. The flow rate is maintained at 0.25 mL/min using a column temperature of 60 °C. The conditioning injections are best made without the mass spectrometer in-line. The effectiveness of the conditioning should be confirmed as shown in Figure 4. The mass spectrometer settings and conditions are the same as those stated on page 3.
1D Linear gradient used for conditioning and obtaining chromatograms in Table 2.
The following describes the supernatant preparation method:
Combine equal volumes of rat plasma and a solution containing 1:1 acetonitrile/methanol with 1% acetic acid. Mix to precipitate, and centrifuge for 10 minutes at 13,000 rcf. Transfer supernatant to a fresh tube. Vortex/mix the tube containing the supernatant, and centrifuge a second time to further clarify the supernatant. Transfer this supernatant to a sample vial and use to condition the column.
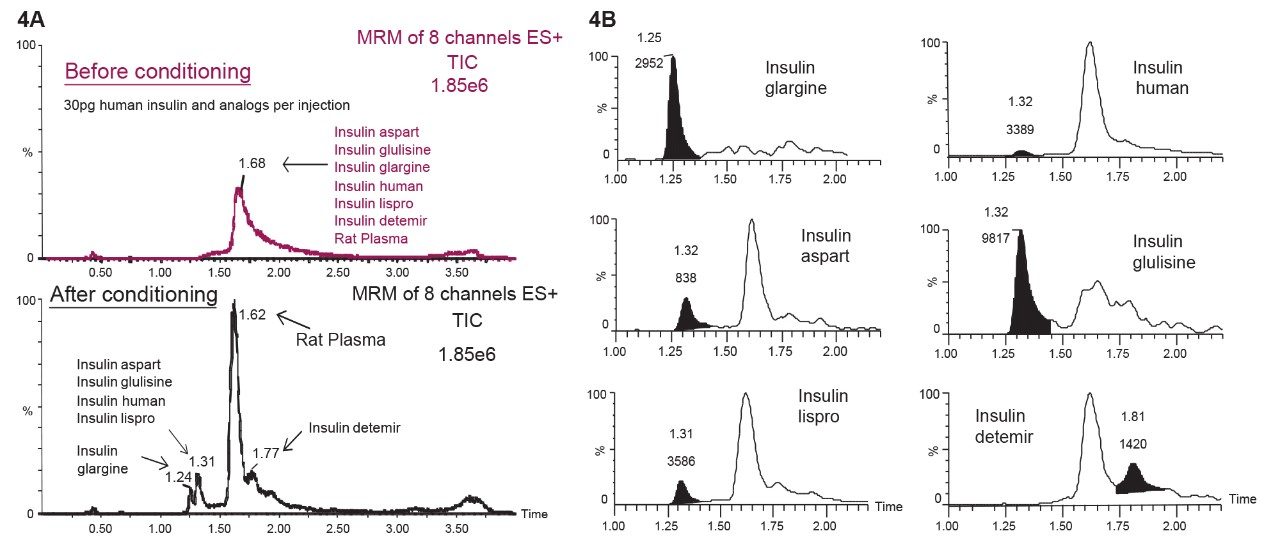
Perform multiple injections totaling 250 µL of the prepared rat plasma supernatant to fully condition the column. Figure 4A shows typical UPLC-MS/MS of total ion chromatograms (TIC) for human insulin and five analogs both before and after conditioning a CORTECS C18+ Column under current 1D methods and conditions.
The insulin mixture used in the chromatograms of figure 4A and 4B was prepared from formulations of insulin and analogs present at 100 IU/mL for Lantus, Apidra, Novolog, Humalog, and Levemir which are equivalent to 3.64, 3.49, 3.5, 3.5, and 14.2 mg/mL of insulin glargine, insulin glulisine, insulin aspart, insulin lispro, and insulin detemir, respectively (values as stated in product inserts). Lyophilized human insulin was dissolved in 0.01 M HCl to generate a concentrated stock at 1 mg/mL.1 The sample diluent was 65/30/5 Milli-Q water/methanol/acetic acid (v/v/v) containing 0.05% rat plasma, added to prevent non-specific binding of the insulin and its analogs.
A 2 µL injection of human insulin and the five analogs at 15 ng/mL (total in diluent) was made on the unconditioned and conditioned column shown in Figure 4A. Upon conditioning, the human insulin and its five analogs could be quantitatively identified exhibiting peak widths of <4 seconds with increased area counts as shown in Figure 4B.
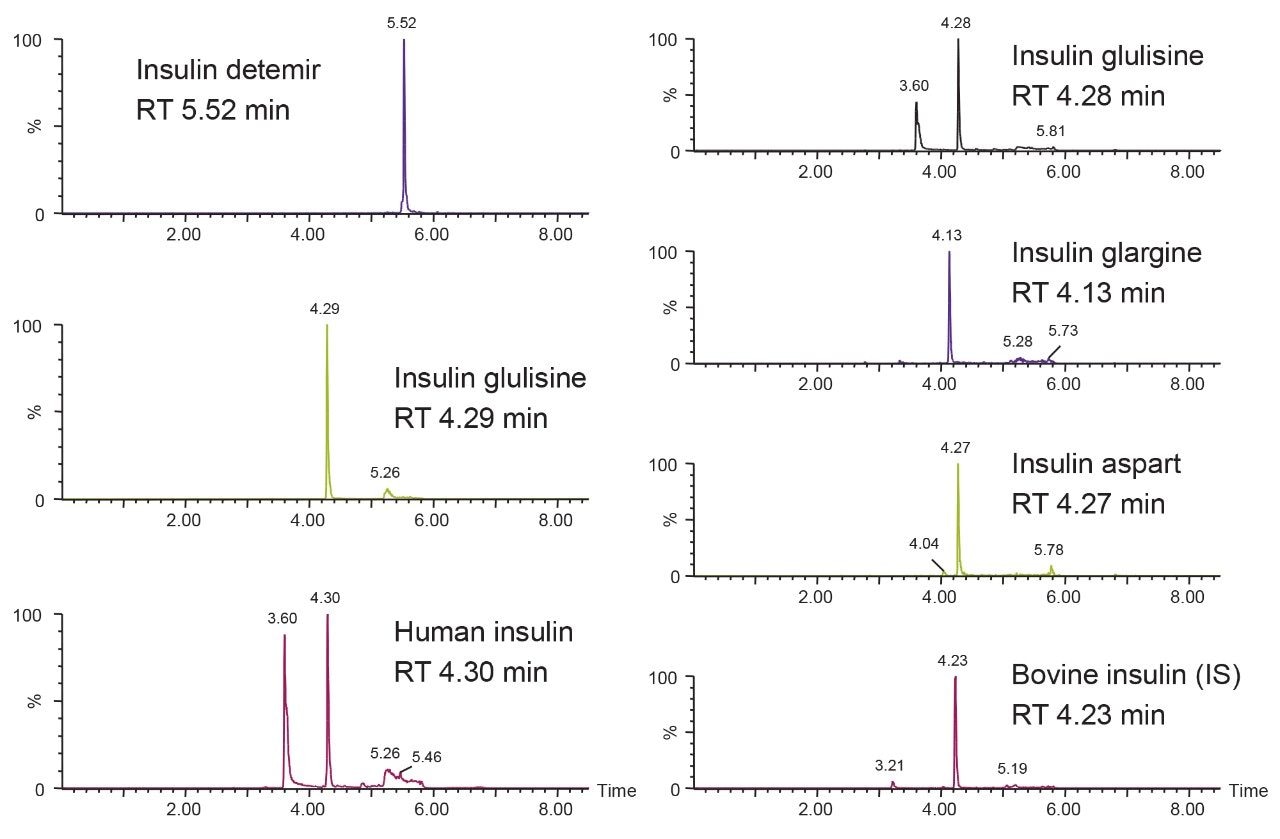
Unlike small molecules, larger peptides and small proteins, i.e., insulins, suffer from poor mass transfer in and out of fully-porous particles. Thus, using a column packed with solid-core particles allows for sharper peak shapes at the higher flow rates typically needed for bioanalytical studies.1,2 Specifically for insulins, it is documented that using a column packed with particles containing a low-level positive surface charge gives superior peak shape and resolution to other columns.3 CORTECS C18+ Columns combine the benefits of solid-core particle technology and a low-level positive surface charge to obtain a new level of separation performance for this application. With CORTECS C18+, insulin peaks are typically 4.0 to 4.5 seconds wide with 2X higher area counts than state-of-the-art, fully-porous particle columns. Representative chromatograms are shown in Figure 5.
The use of multidimensional chromatography facilitated higher injection volumes, through the use of at-column dilution (ACD), while providing additional sample cleanup by employing a trap and back-elute strategy. The combination of these chromatographic system elements allowed us to inject up to 40 µL without experiencing insulin breakthrough. Chromatographic breakthrough was evident on a 1D system with injection volumes >10 µL (data not shown).
A proof-of-concept study published earlier in 20133 used reversed-phase only SPE to extract four insulin analogs. Although the method clearly demonstrated feasibility, endogenous background was high in the transitions for human insulin and lispro, with average LLOQs of 200 to 500 pg/mL reported for the four insulins, making accurate low-level quantification and distinction between the lispro and human insulin impossible. This application presents a significantly more selective two-step extraction utilizing a protein precipitation (PPT) pre-treatment step and a strong anion-exchange mixed-mode SPE device to effectively reduce endogenous background, as well as improve specificity and detection limits.
During optimization of protein precipitation conditions for insulin, various ratios of organic solvent to plasma, as well as different precipitation solvents were tested. Protein precipitation (PPT) with a 1:1 ratio of various solvents (ACN or ACN modified with one of the following: 5% NH4OH, 1% acetic acid, or 2% formic acid) resulted in >90% recovery of the six insulins without precipitating the peptides themselves. Protein precipitation with higher ratios of organic resulted in peptide loss due to undesired precipitation of the insulins. The PPT pre-treatment effectively eliminated protein binding and reduced endogenous interferences from large proteins such as albumin.
The PPT supernatants were then diluted with 5% concentrated NH4OH to bring the pH to ~11. At this pH, insulin and its analogs will carry a net negative charge, putting them in the proper charge state to bind to Oasis MAX by ion exchange. A 2007 study4 determined that mixed-mode SPE (especially in its ion-exchange elution) was significantly more selective than reversed-phase only SPE. For this assay specifically, where the use of low m/z fragments for human insulin and lispro was necessary, it was imperative to have the most selective sample preparation possible.
The diluted supernatant was then applied to conditioned SPE plates, and analytes were well retained on the SPE sorbent during the basic pH load step. Optimization of the elution solution was critical to fully elute the insulins, maintain solubility, and minimize interferences from the plasma matrix. The optimum elution solution was 60% organic with 10% acetic acid. The final eluate was diluted with an equal volume of water.
To generate standard curves, human plasma was fortified with a mixture of the insulins at the following final concentrations: 50, 100, 200, 500, 1000, 2000, 5000, and 10,000 pg/mL. Quality control (QC) samples were prepared in human plasma at the following concentrations: 150, 750, 2500, and 7500 pg/mL.
Bovine insulin (final concentration 10 ng/mL) was used as the internal standard (IS) for all insulins. Peak area ratios (PARs) of the analyte peak area to the IS peak were calculated. Calibration curves, prepared in human plasma, were constructed using PARs of the calibration samples by applying a one/concentration weighted linear regression model. All QC sample concentrations were then calculated from their PARs against their respective calibration lines. All curves were linear using the 1/x regression. A summary of standard curve performance for all insulins is shown in Table 3.
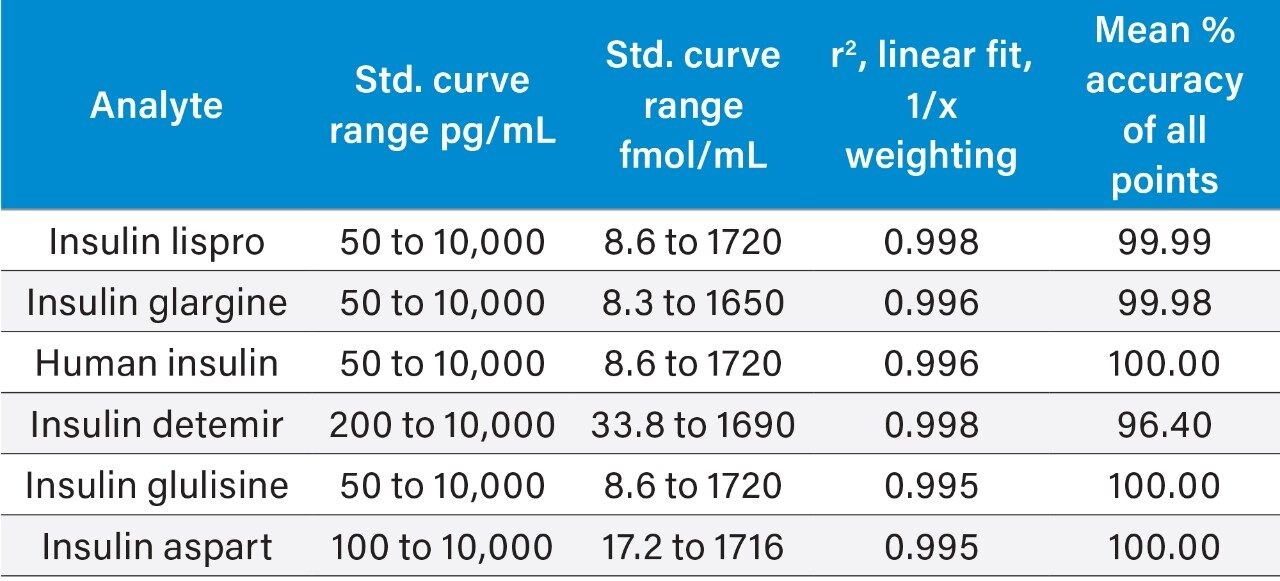
For human insulin, the basal concentration in pooled or individual control plasma was determined by calculating the x-intercept. The basal level of human insulin (average = 1937 pg/mL) was then added to the spiked concentration for all standard curve and QC samples to enable accurate quantification. A representative standard curve for insulin lispro is shown in Figure 6.

Inter- and intra-day accuracy and precision were calculated for all QC samples. Representative summary statistics for insulin glargine, lispro, and human insulin can be found in Tables 4, 5, and 6. All other analogs exhibited similar performance.
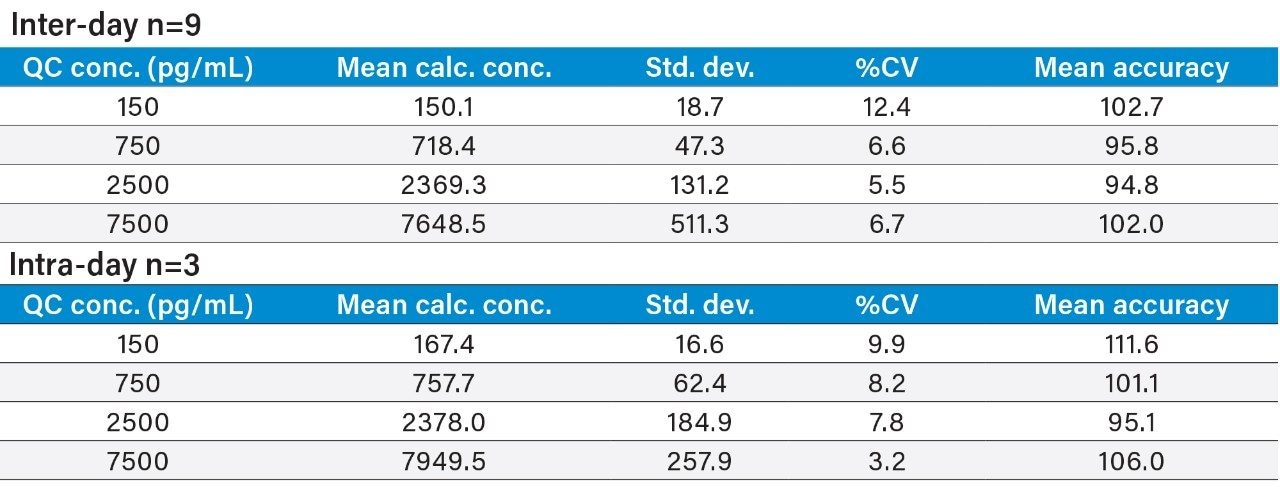
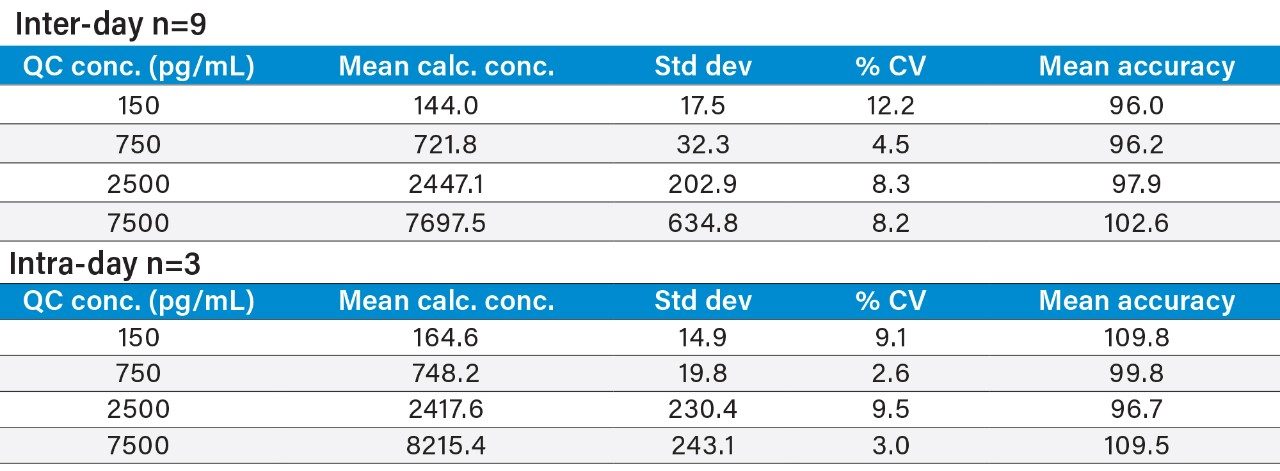
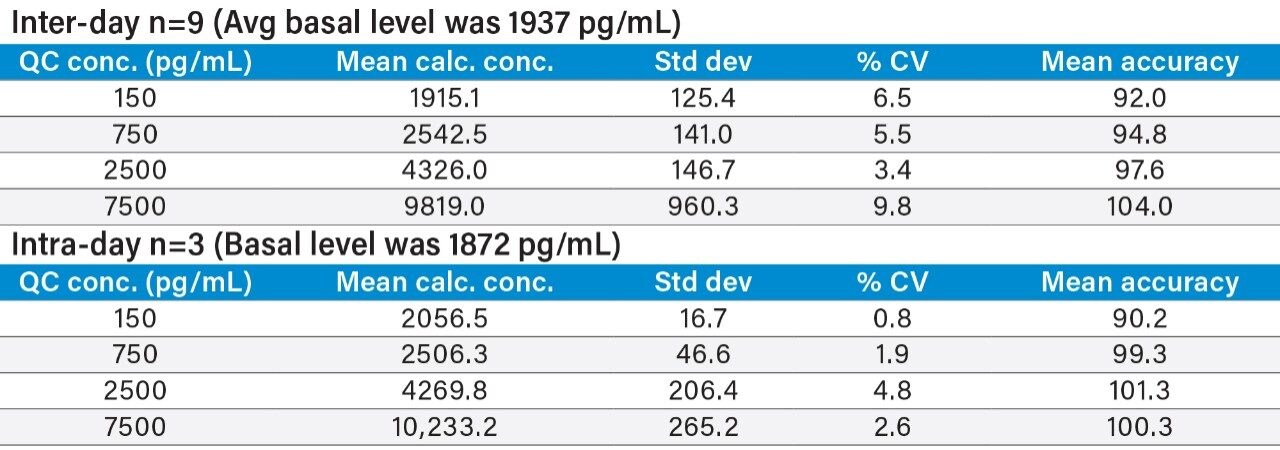
Matrix factors and CVs of matrix factors for all analogs were calculated in six individual sources of human plasma, as outlined in the 2007 AAPS white paper.5 The % CV of the matrix factors were 12.3, 11.6, 11.8, 9.0, and 7.7 for insulin detemir, glargine, aspart, glulisine, and lispro, respectively. All easily met the recommended criteria of <15% CV.
In addition, a study was performed to assess the impact of high levels of human insulin, expected to be found in type II diabetic patients, on the assay specificity. This is particularly important for two reasons. First, not all analytes are chromatographically resolved from one and another. Second, due to the use of a nominal mass instrument (triple quadrupole) and the high degree of sequence homology between the analogs themselves and with human insulin, the possibility of MS overlap in some of the transitions exists. For this test, plasma samples were fortified to a final concentration of 5 ng/mL with all of the analogs. A subset of these samples was also spiked with human insulin at 200X higher concentration, to a final concentration of 1 µg/mL human insulin. All samples were then pre-treated, extracted, and quantified. There was no significant change in area counts for any of the analogs when human insulin was present in high concentration.
Using analytical scale chromatography and simple 96-well SPE sample preparation, this method reaches detection limits for intact insulins, comparable to those previously achieved using immuno-precipitation and nano-scale chromatography.6-11
Development of this assay was challenging due to a high degree of non-specific binding (NSB), protein binding, and difficulty maintaining peptide solubility throughout the SPE extraction and elution process. Sample pre-treatment prior to SPE proved to be critical in improving recovery and specificity. Protein precipitation with 1% acetic acid 1:1 acetonitrile/methanol resulted in 80% to 100% recovery without precipitating the peptides themselves. Mixed-mode strong anion SPE provided an additional layer of selectivity, and facilitated the use of the low m/z MS fragments that were necessary to distinguish human insulin from lispro.
The use of the CORTECS UPLC C18+ Column provided significantly improved sensitivity and peak shape for insulin and its analogs versus charged-surface fully-porous columns and traditional C18 solid-core columns. This led to an improvement in detection limits of approximately 2X, when combined with proper MS fragment choice, and selective SPE cleanup enabled quantification limits of 50 to 200 pg/mL for the six insulins.
All FDA criteria12 for accuracy and precision of the method were easily met. Average accuracies for standard curve points and QC samples were >92%, with most close to 99%. Inter- and intra-day precision for all QC samples was greater than 7.5%. CVs of matrix factors across six lots of human plasma were <15%, further supporting the selectivity of the method.
720004727, June 2019