
For forensic toxicology use only.
This is an Application Brief and does not contain a detailed Experimental section.
This Application brief describes to develop an efficient extraction method for the UPLC-MS/MS analysis of synthetic fentanyl analogues from whole blood while removing residual phospholipids for forensic toxicology.
Synthetic fentanyl compounds were rapidly and reproducibly isolated from whole blood while minimizing residual phospholipids.
The need for the analysis of synthetic fentanyl analogues from blood for forensic toxicology has increased significantly in the last few years as the abuse of these compounds, as well as associated overdoses and fatalities, have increased substantially.1 One of the main analytical challenges is the low concentrations of these compounds typically seen in blood samples. Because of this, robust and reproducible sample preparation methods can be crucial to achieving the required sensitivity. One of the components of biological samples that can impact sensitivity is the presence of phospholipids. Traditional mixed-mode cation exchange methods tend to have substantial amounts of residual phospholipids compared to other ion-exchange sorbents. This is of particular relevance when working with whole blood, which comprises up to 45% red blood cells, and contains a larger amount of phospholipids than plasma.
This work details the extraction of fentanyl analogs from whole blood using Oasis PRiME MCX μElution plates. Extraction efficiency, matrix effects, and phospholipid removal are all highlighted. In addition, sample pretreatment was optimized to efficiently process whole blood samples prior to solidphase extraction.
Synthetic fentanyl compounds were spiked into whole blood. Blood samples (100 μL) were treated with a solution of zinc sulfate and ammonium acetate (ZnSO4/NH4OAc) to lyse the cells, followed by precipitation with 50:50 ACN:MeOH. After centrifugation, the supernatant was diluted with 4% phosphoric acid (H3PO4) and extracted using Oasis PRiME MCX μElution plates. Samples were eluted with 50:50 ACN:MeOH containing 5% ammonia solution. Extracts were analyzed using an ACQUITY™ UPLC I-Class/Xevo TQ-S micro System.
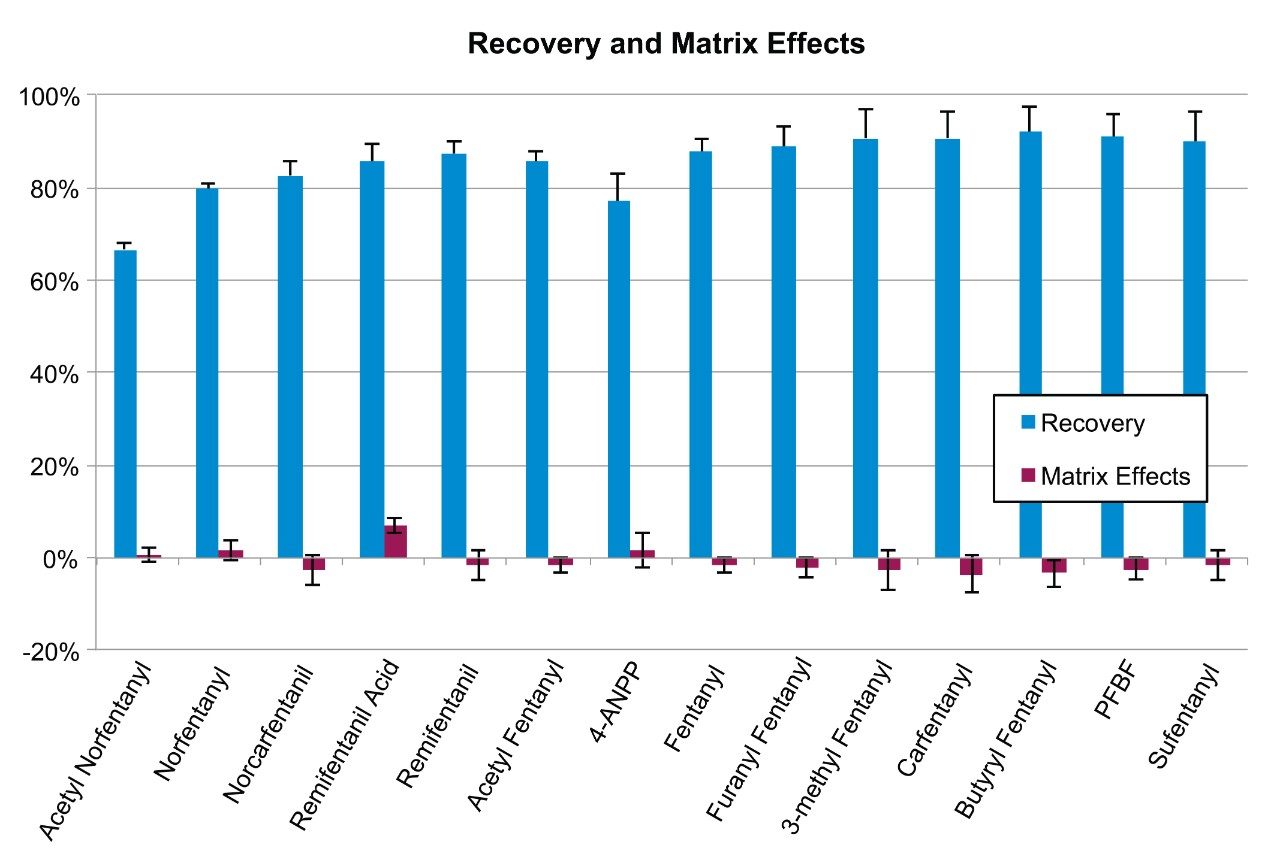
Figure 1 shows the recovery of the synthetic fentanyl analogs from whole blood. Recoveries ranged from 67 to 91%, with an average of 86.4%. The values represent the mean obtained for three different concentrations (250, 1000, and 10,000 pg/mL). Recoveries were consistent, with all %RSDs under 10%. Matrix effects were calculated and are also shown in Figure 1. All matrix effects were less than 10% with RSDs less than 5% demonstrating that there was no ion suppression associated with this method. No concentration dependent bias was seen for recovery or matrix effects.
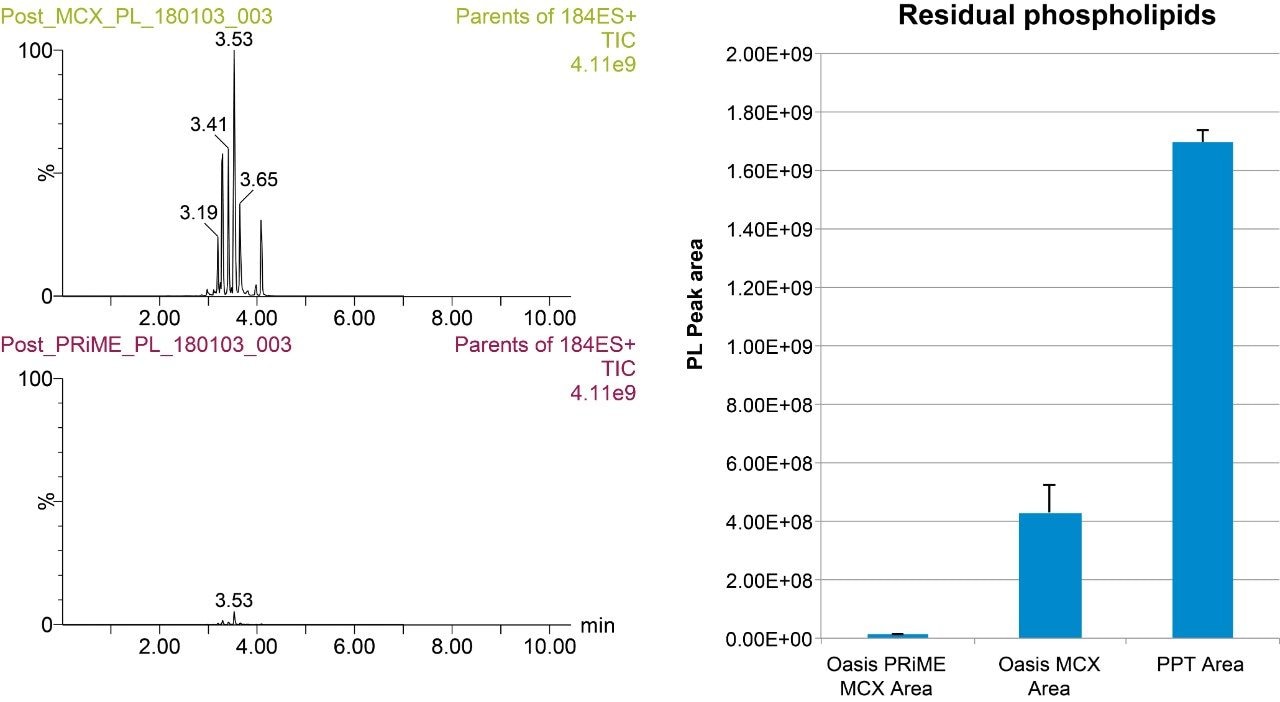
One of the novel properties of Oasis PRiME MCX is a superior removal of phospholipids. Figure 2 compares the phospholipids remaining from whole blood samples after protein precipitation, extraction with Oasis MCX, and extraction with Oasis PRiME MCX. The chromatogram and associated graph show that Oasis PRiME MCX removes greater than 99% of phospholipids compared to protein precipitation and 97.4% of phospholipids compared to Oasis MCX, a substantial improvement of an already clean extract.
Synthetic fentanyl compounds were extracted from whole blood using Oasis PRiME MCX μElution plates prior to UPLC-MS/MS analysis for forensic toxicology. In addition to providing high and consistent recoveries for all analytes with negligible matrix effects, residual phospholipids were nearly eliminated, with greater than 99% removed compared to protein precipitation and greater than 97% removed compared to Oasis MCX. This translates to cleaner extracts that can improve column lifetime, minimize MS maintenance, and is likely to contribute to the lack of matrix effects seen with this assay. This group of fentanyl analogues spans a wide range of polarities and this method should be applicable to additional fentanyl analogues as necessary.
720006330, July 2018