
For forensic toxicology use only.
This application brief describes an improved chromatographic method that enables the analytically sensitive measurement of PEth 16:0/18:1 in whole blood, using Waters ACQUITY UPLC I-Class/Xevo TQD and compare a new sample preparation method to a widely used liquid:liquid extraction.
The ACQUITY UPLC I-Class/Xevo TQD System allows analytically sensitive detection of PEth in whole blood for forensic toxicology analysis.
As the legal consequences of alcohol abuse can have significant and far-reaching implications, specific and reliable biomarkers are needed. PEth comprises a group of abnormal phospholipids that are enzymatically formed in cell membranes only in the presence of ethanol. The determination of PEth in blood is attractive owing to high specificity, as formation is reported to correlate with ingested ethanol dose.¹ PEth can also be used to distinguish drinking patterns and behaviors, e.g., to identify moderate or excessive drinkers, or to identify episodes of binge drinking.²
A number of LC-MS/MS methods for PEth have been described, but some aspects of the procedure can be challenging, e.g., for the determination of PEth in blood, sample preparation procedures using liquid:liquid extraction (LLE) have been reported but recoveries can vary. Furthermore, the lipophilicity of the molecule can result in poor chromatographic peak shape and/or high background responses. All of these issues can affect overall sensitivity of the assay and robustness.
PEth 16:0/18:1 was obtained from Sigma-Aldrich (UK). The deuterated analogue d5-PEth 16:0/18:1, was used as an internal standard (IStd) and was from RedHot Diagnostics AB (Sweden). A working solution of IStd was prepared, at a concentration of 1 µg/mL, in acetonitrile.
Thirty microlitres of IStd were added to a vial with 30 µL whole blood (calibrator or sample) and 600 µL of acetonitrile/1% formic acid, and extracted for 20 minutes at room temperature. The extract was applied to an Ostro Protein Precipitation & Phospholipid Removal Plate (P/N 186005518), washed with acetonitrile/1% formic acid, and eluted twice with dichloromethane/methanol (1/1, v/v). The pooled eluate was dried under a stream of nitrogen. Following reconstitution in 300 µL mobile phase A, samples were transferred to UPLC Total Recovery Vials (P/N 186000384C) before analysis of 2 µL using an ACQUITY UPLC I-Class (FTN)/Xevo TQD System.
Separation was achieved using a CORTECS UPLC C8 Column (P/N 186008399) maintained at 35 °C and eluted with a gradient comprising ammonium acetate buffer (mobile phase A) and acetonitrile (mobile phase B). An extended washing step was included at the end of the run to provide robustness and to avoid use of strong additives, such as tetrahydrofuran, which have been used to prevent carryover in methods with shorter run times. The following MRM transitions were monitored in electrospray negative (ES-): PEth 16:0/18:1, m/z 701.5 > 255.3 (quantifier) and 281.3 (qualifier), d5-PEth 16:0/18:1, m/z 706.5 > 255.3.
Figure 1 shows a comparison of the responses for a calibrator prepared using the described Ostro Plate extraction protocol and a calibrator prepared by LLE (hexane). The lower limit of quantification (LLOQ) for PEth (based on a minimum signal:noise ratio of 5:1 for the qualifier transition) was estimated at 10 ng/mL for the Ostro Plate procedure and 50 ng/mL for LLE.
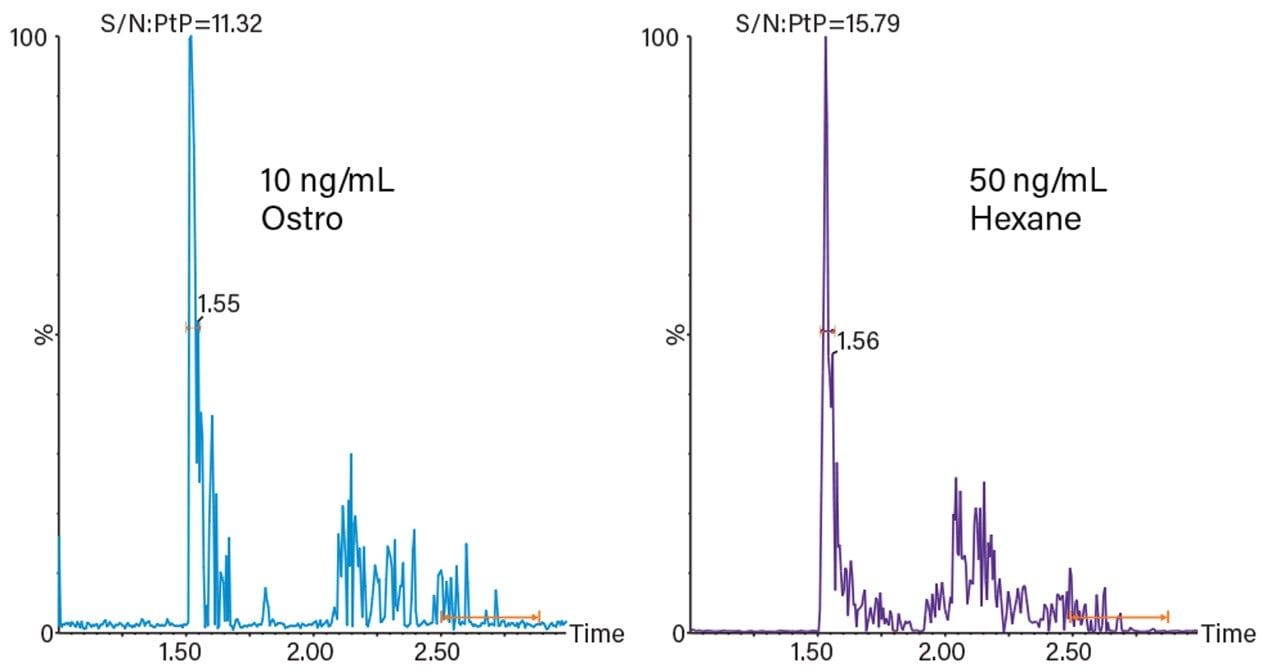
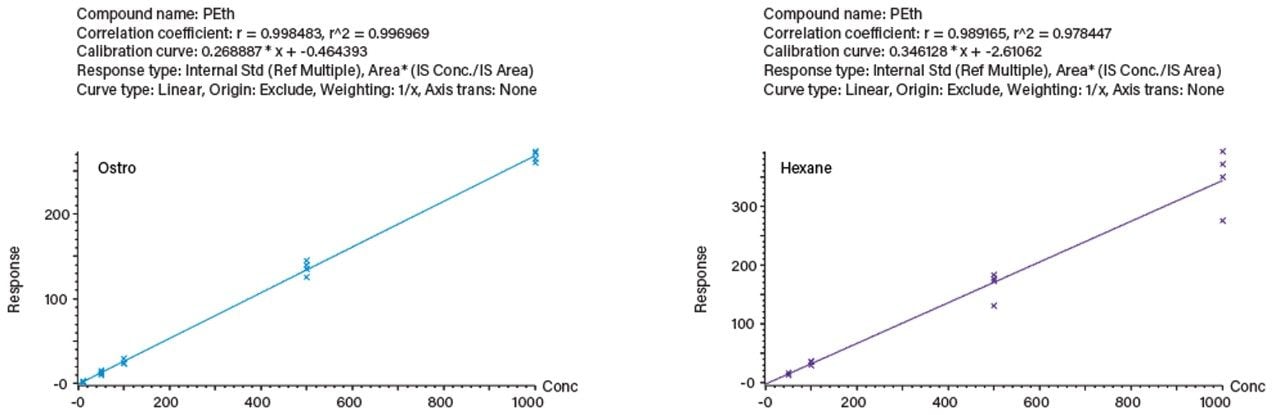
Figure 2 shows the calibration lines for four blood samples per concentration prepared by both techniques; even with this small sample set, better reproducibility was observed with the Ostro Plate.
Carryover was evaluated by monitoring the response obtained for blank samples injected immediately following the analysis of high concentrations (3000 ng/mL) of PEth – no carryover was observed for either extraction method.
Preparation time for a batch of 24 samples was significantly shorter (1 hour vs. 3 hours) with the Ostro Plate procedure owing to the requirement for additional transfer steps (vial to glass tubes) and larger volumes to dry down with the LLE method.
The use of the Ostro Protein Precipitation & Phospholipid Removal Plate as trapping material provided cleaner extracts, a lower LLOQ, and improved reproducibility. Sample preparation time was significantly shorter compared with a commonly-used LLE procedure, the Ostro Plate format also offers potential for automation. The use of the ACQUITY UPLC I-Class/Xevo TQD System allows analytically sensitive detection of PEth in whole blood for forensic toxicology analysis. The total run time was 6.3 minutes and included an extended washing step to eliminate the use of strong additives to prevent carryover.
720006059, July 2017