
A novel microflow technique using the ACQUITY UPLC M-Class System and IM-MS that leverages both positive and negative ionization has been developed to analyze the extracts of Passiflora edulis, alata, caerulea, and incarnata. The profiles determined from the extracts can be used to confirm food commodity authenticity.
Legislative focus has resulted in the expansion of method development to support the analysis of active compounds in and authenticity of functional food products and dietary supplements. For example the European Union’s Directive 2004/24/EC came into full effect on 30 April 2011. Hundreds of traditional herbal remedies were banned, as the EU directive aims to protect consumers from possible damaging side-effects of over-the-counter herbal medicines. Recent regulations allow only long-established and quality-controlled products to be sold. Manufacturers have to prove that their products have been made to strict standards and contain a consistent and clearly marked dose.
The ionKey/MS System ion mobility mass spectrometry (IM-MS) can provide a route to specific and unambiguous identification at low detection levels. This technique offers some unique advantages for profiling complex mixtures. IM-MS combines high resolution mass spectrometry and high efficiency ion mobility based measurements, with enhanced sensitivity. IM-MS is a rapid orthogonal gas separation phase technique that allows another dimension of separation to be obtained within an LC timeframe. Compounds can be differentiated based on size, shape, and charge.
A novel microflow technique using the ACQUITY UPLC M-Class System and IM-MS that leverages both positive and negative ionization has been developed to analyze the extracts of Passiflora edulis, alata, caerulea, and incarnata, shown in Figure 1. The genus Passiflora consists of approximately 450 species, a few of which are commercially exploited in functional food products such as teas and juices. These species contain flavonoids, one of the largest and most widespread classes of compounds which possess diverse pharmacological/biological properties. The target marker flavonoids ionize efficiently in positive and negative modes, enabling a positive/negative mode on the ionKey/MS System comparison to be performed. Collision cross section measurements (CCS) can be used to produce routine unequivocal identification of marker flavonoid isomers in complex samples.1 The profiles determined from the extracts can be used to confirm food commodity authenticity.
|
LC system: |
ACQUITY UPLC M-Class |
|
Mobile phase A: |
Water (0.1% Formic acid) |
|
Mobile phase B: |
Acetonitrile (0.1% Formic acid) |
|
Flow rate: |
2.0 μL/min |
|
Injection volume: |
5 μL (full loop) |
|
Separation device: |
iKey BEH C18 PCA Separation Device, 130A, 1.7 μm, 150 μm x 50 mm (p/n 186007580) iKey BEH C18 Separation Device, 130A, 1.7 μm, 150 μm x 50 mm (p/n 186007256) |
|
Separation device temp.: |
40 °C |
|
Time (min) |
Flow rate |
%A |
%B |
|---|---|---|---|
|
0.0 |
2 |
99.0 |
1.0 |
|
1.0 |
2 |
99.0 |
1.0 |
|
3.0 |
2 |
90.0 |
10.0 |
|
5.0 |
2 |
70.0 |
30.0 |
|
13.0 |
2 |
1.0 |
99.0 |
|
15.0 |
2 |
1.0 |
99.0 |
|
15.1 |
2 |
99.0 |
1.0 |
|
17.0 |
2 |
99.0 |
1.0 |
|
MS system: |
SYNAPT G2-Si |
|
Ionization mode: |
ESI+ and ESI- |
|
Capillary voltage: |
3 kV (+) and 2.6 kV (-) |
|
Sample cone voltage: |
30 V |
|
Lock mass and CCS: |
Leucine enkephalin, [M+H]+ =556.2766 and [M-H]- =554.2620 |
|
Acquisition range: |
50 to 1,200 m/z |
|
Acquisition rate: |
10 spectra/sec |
|
Collision energy ramp: |
30 to 70 eV |
|
Resolution: |
20,000 FWHM (Res mode) |
|
IMS T-Wave velocity ramp: |
Start: 1,000 m/s, End: 300 m/s |
|
IMS T-Wave pulse height: |
40 V |
|
IMS gas flow: |
90 mL |
For positive ion electrospray, an iKey Separation Device (p/n: 186007256) was used. The iKey Separation Device, shown in Figure 2, incorporates a 1.7 μm, ACQUITY UPLC BEH C18, stationary phase in a 150 μm diameter separation channel. The eluent from the separation channel flows directly to an integrated ESI emitter. All microfluidic, gas, and electrical connections are automatically engaged when the iKey Seperation Device is inserted into the source enclosure and locked into place. For negative mode, the Post Column Addition (PCA) iKey Separation Device (p/n: 186007580) was used. The PCA iKey Separation Device incorporates an additional channel, enabling post column addition of solvent. The make up solvent was configured to be delivered from channel A of the MS system fluidics for this study.

The ionKey/MS System ion mobility screening was performed in positive and negative modes to analyze the hydromethanolic extracts of P.incarnata, P.edulis, P.caerulea, and P.alata. In a previous study using UPLC and ion mobility, the extracts were diluted 40:1.2 For the analysis performed it has been possible to dilute the samples further to 400:1. Also separate aqueous high purity standard solutions (0.1 pg/μL to 10 pg/μL) of isovitexin, vitexin, isoorientin, and orientin were used to determine sensitivity and linear response. The iKey Separation Device gradient employed initial conditions at 99% aqueous. Using a post column addition solvent (IPA), enabled a single voltage of 2.6 kV to be employed throughout the chromatographic gradient, while maintaining stable spray conditions and stable ionization of the analytes exiting the iKey Separation Device. In Figure 3 the complexity of the samples analyzed is illustrated. The base peak ion chromatogram illustrates the conventional view of the complexity of the sample profiled. A series of flavonoid isomers have been selected to clearly illustrate the chromatographic performance obtained using both iKey Separation Device designs, as shown in Figure 4. The ionKey/MS System’s positive and negative mode extracted exact mass chromatograms for a series of isomeric flavonoids determined to be present in 400:1 diluted Passiflora edulis extract are presented. The chromatographic profile obtained is shown to be comparable using the iKey Separation Device and PCA iKey Separation Device.
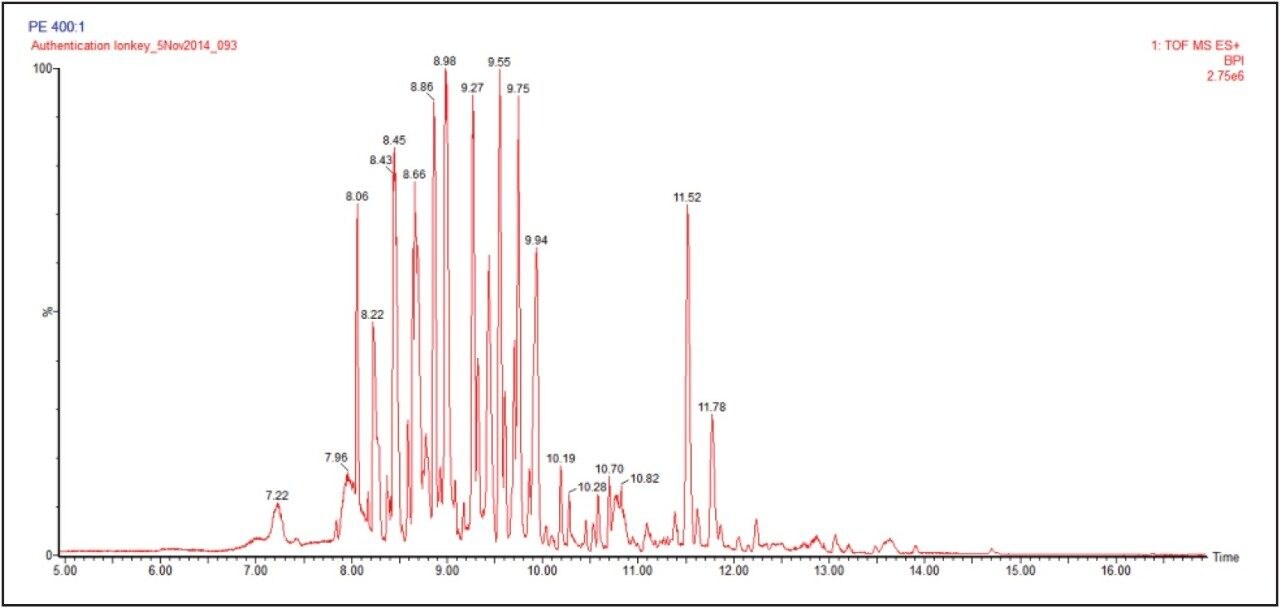
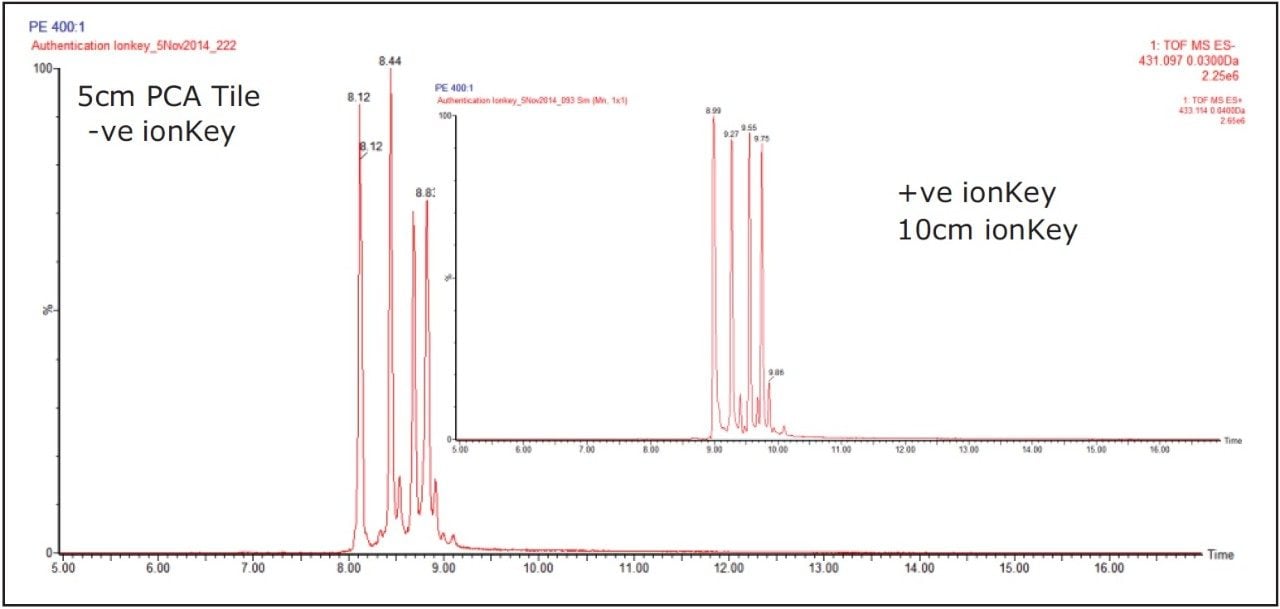
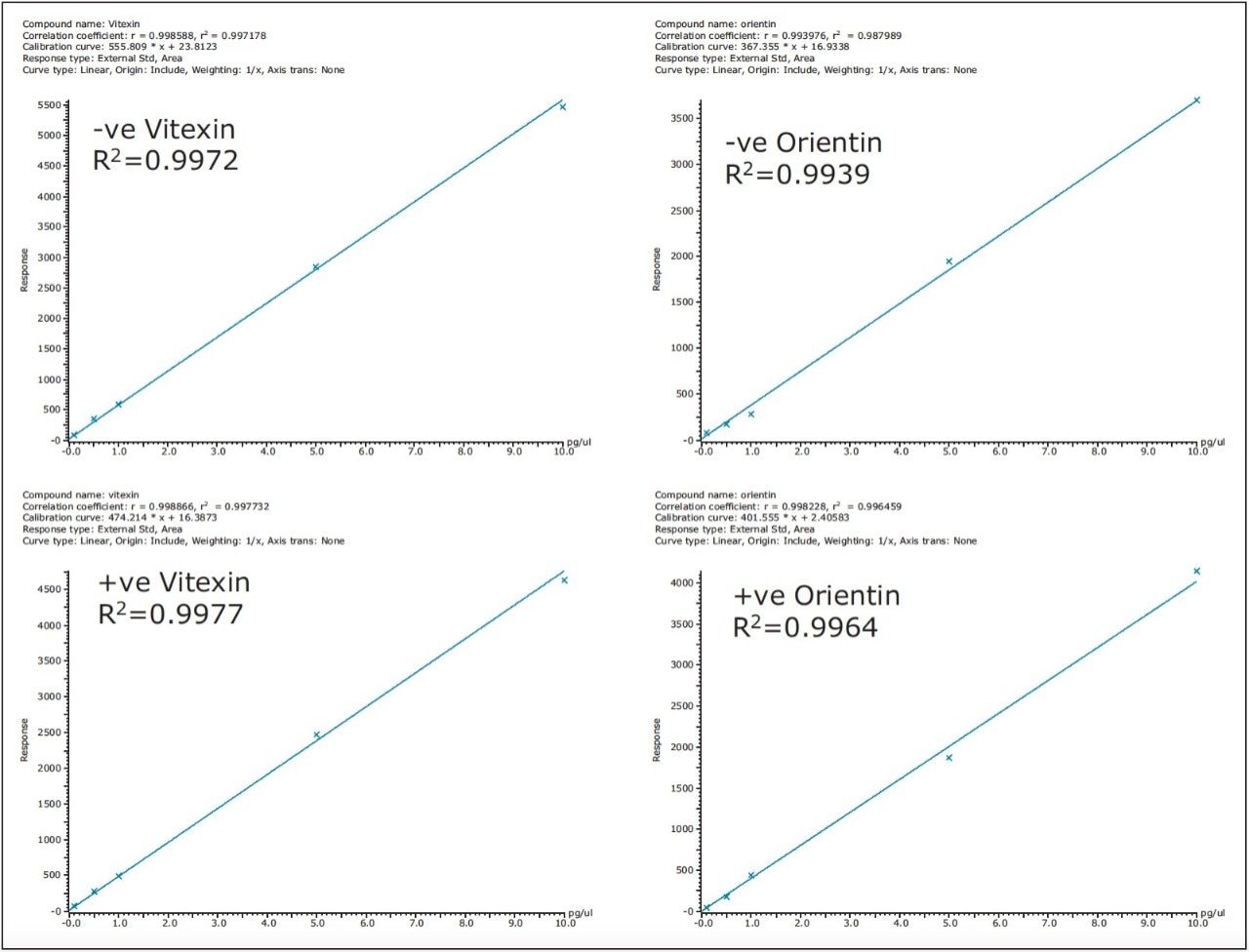
Figure 5 shows that the linear response obtained for orientin and vitexin solvent standards (0.1 pg/μL to 10 pg/μL) using positive and negative mode on the ionKey/MSE System is equivalent. Excellent correlation coefficients of R2>0.99 have been acquired over three orders of dynamic range. The data shows the potential of using the ionKey/MS System with benefits of full spectral acquistion attained using time-of-flight mass spectrometry at low detection levels; in this case at 100 fg/μL.
Utilizing the extended functionality of the SYNAPT G2-Si, it shown in Figures 6 and 7 that using positive and negative mode the ionKey/MS System ion mobility IM-MS,E it is also possible to retain sensitivity as illustrated for isovitexin solvent standard over the concentration range 0.1 pg/μL to 10 pg/μL.
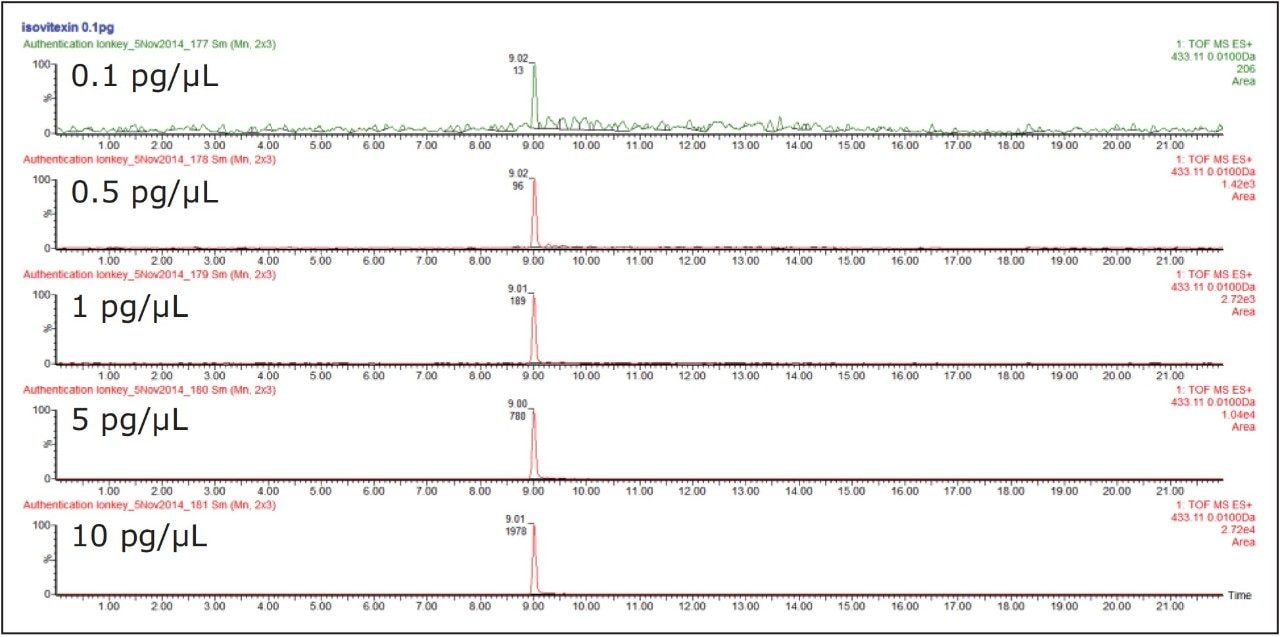
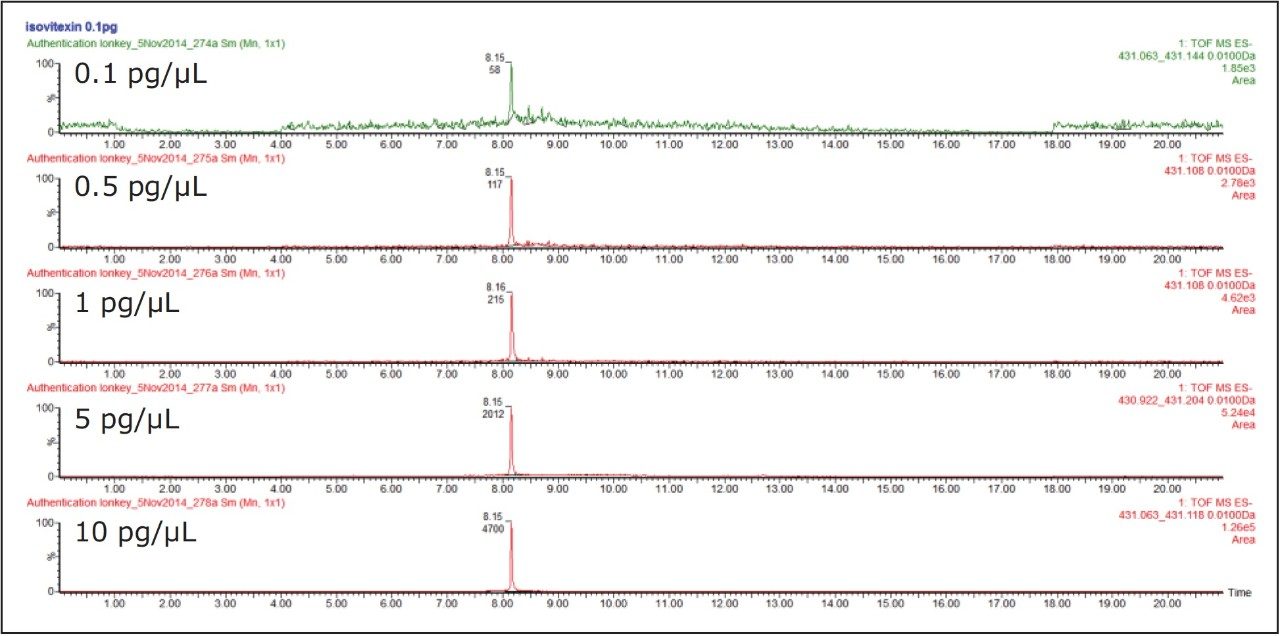
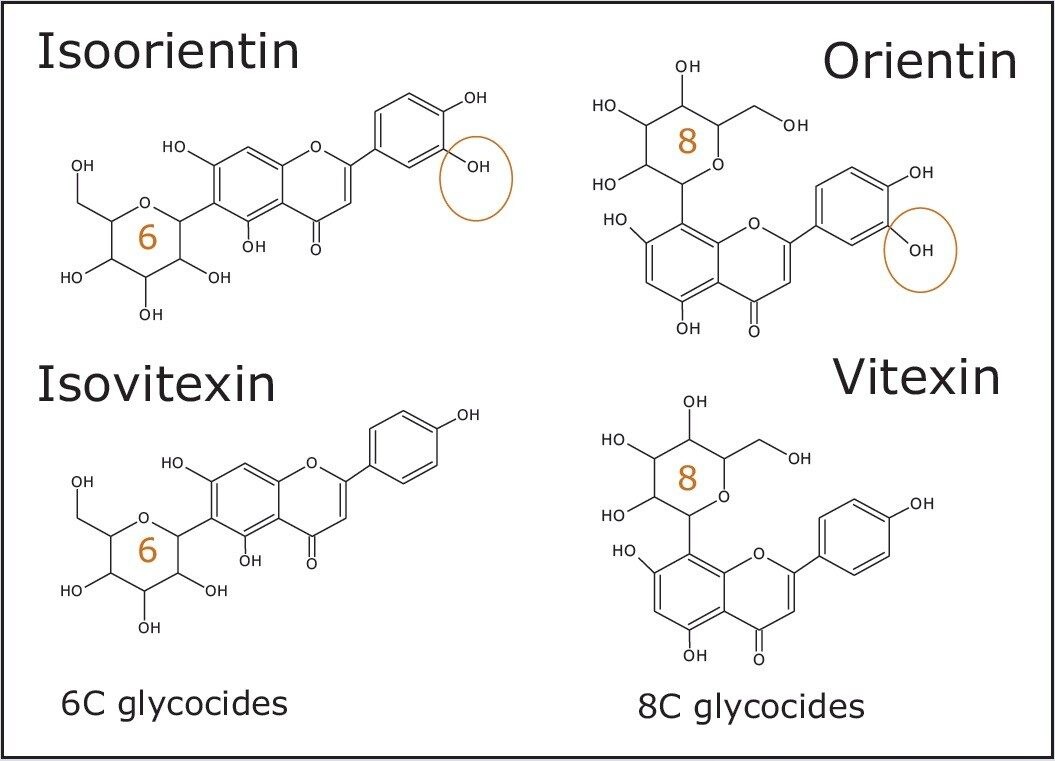
Under the chromatographic conditions used, vitexin and isovitexin coelute. In negative ion mode both the 6C glycoside isomers (isoorientin/orientin) and the 8C glycoside isomers (isovitexin/vitexin), can be separated using ion mobility. They can also be identified from their CCS values obtained using nitrogen based travelling ion wave mobility (TWCCSN2).
Hence it is possible to generate the individual calculated concentrations for two isomeric species (isovitexin/vitexin) that coelute at the same retention time using ion mobility, as shown in Figure 8. For the first time, two coeluting isomeric species have been quantified using precursor ion selection. This has been acheived because the flavonoids are ion mobility separated, which could not be acheived using only the selectivity of mass accuracy. In addition, the UNIFI Software Component Summary is presented where a mass accuracy of <1 ppm has been obtained, as well as CCS measurements within 0.21% of the expected TWCCSN2 values.
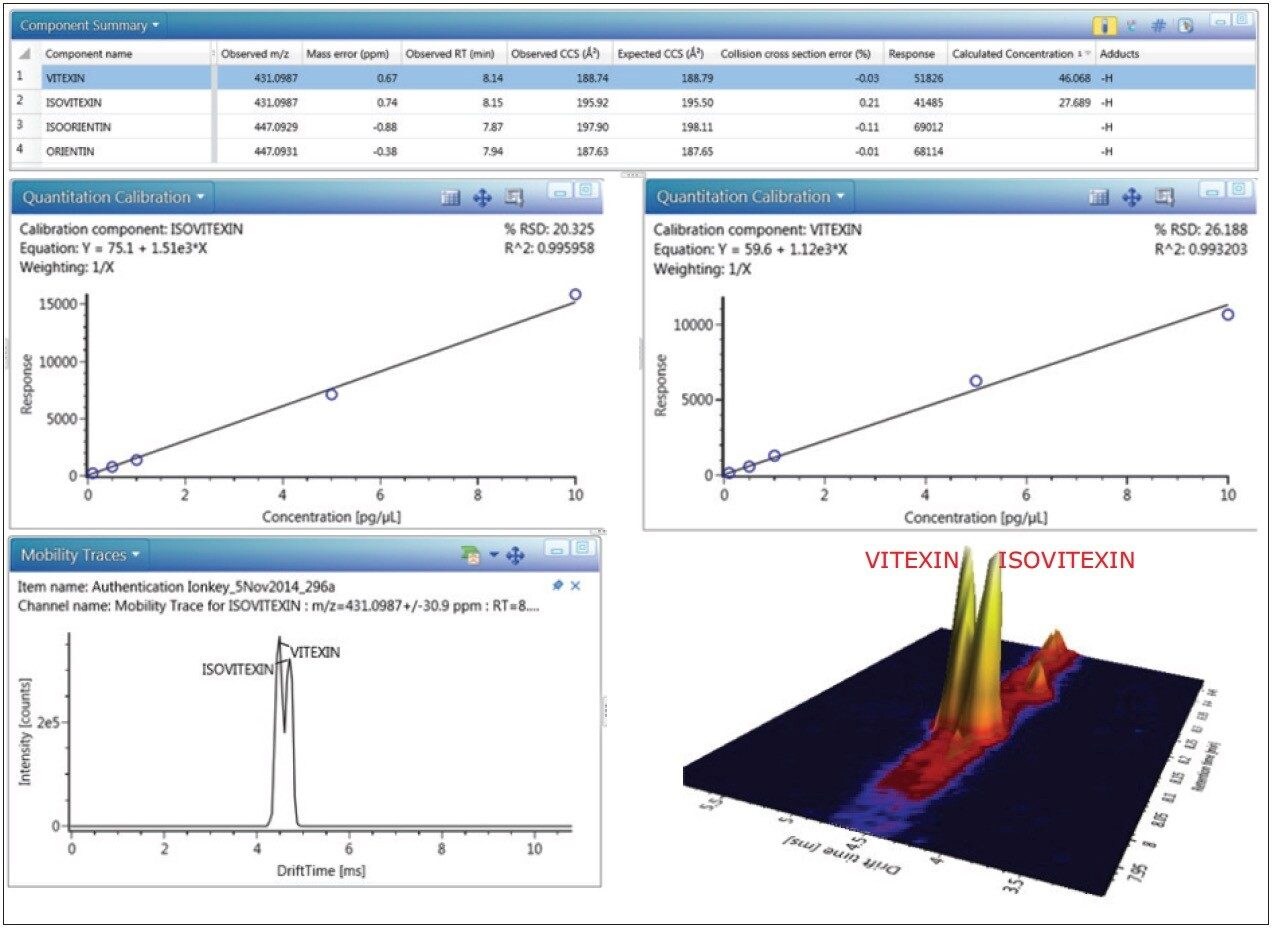
The flavonoid library was created within the UNIFI Scientific Library 17 months before this assay was performed, illustrating the reproducibility and precision that can be obtained using ion mobility. The combination of the ionKey/MS System and ion mobility offers the potential of high selectivity, specificity, and sensitivity, as well as practical aspects such as reduced solvent and sample consumption. Using TWCCSN2 as an additional identification parameter also has the potential to reduce the reliance on retention time based confirmations and hence the need to purchase expensive high purity standards (1 mg vitexin 98% pure= £144.50, and 1 mg isovitexin 98% pure= £144.50).
720005425, January 2016