In this application note, we introduce a simple, 3-step SPE protocol using Oasis HLB in μElution Plates and 1 cc cartridges. This protocol saves an average 40% processing time, 70% solvent consumption, and provides a simple method that works for a wide range of acidic, neutral, and basic analytes. This simplified, 3-step protocol consists of load, wash, and elution, thus eliminating the conditioning and equilibration steps.
In bioanalysis, solid phase extraction (SPE) is usually chosen as one of the best options available for sample preparation to extract analyte of interest from complex samples. However, due to its perception of being time consuming, costly and complicated, SPE is often the last option selected. Admittedly, SPE can provide substantial benefits in bioanalysis including low matrix factors, high sample cleanliness, and concentration of low level analytes/metabolites. The standard SPE protocol consists of 5 steps and includes conditioning, equilibration, sample loading, washing, and eluting which is time consuming and complicated. Therefore, a simplified SPE protocol is desired that can be used for a wide range of pharmaceutical drug compounds and their metabolites.
In this application note, we introduce a simple, 3-step SPE protocol using Oasis HLB in µElution Plates and 1 cc cartridges. This protocol saves an average 40% processing time, 70% solvent consumption, and provides a simple method that works for a wide range of acidic, neutral, and basic analytes. This simplified, 3-step protocol consists of load, wash, and elution, thus eliminating the conditioning and equilibration steps. The key to being able to perform this simplified protocol is to use the water-wettable Oasis HLB sorbent that does not de-wet under vacuum, which is crucial for recovery of analytes during SPE. To demonstrate this unique capability, Oasis HLB (a water-wettable sorbent), will be compared to a silica-based C18 and a competitive polymer-based sorbent using typical compounds found in drug discovery and development spiked into urine and plasma. These compounds include antidepressants, antiretrovirals, and non-steroidal anti-inflammatory drugs. The µElution and 1 cc cartridge formats were evaluated using this simple protocol in terms of recovery, reproducibility, and matrix factors. In addition, long-term method robustness and batch-to-batch consistency of the Oasis HLB product were demonstrated with sorbent lots manufactured between the years 2005 and 2013. The results will show that simplified, 3-step protocol gives the same high analyte recoveries, low variability, and low matrix factors as the standard 5-step SPE protocol while reducing processing times and overall cost.
|
LC system: |
ACQUITY UPLC I-Class |
|
Column: |
ACQUITY UPLC CSH C18, 2.1 x 100 mm, 1.7 μm (p/n: 186005297) |
|
Mobile phase A: |
0.1% formic acid in water |
|
Mobile phase B: |
0.1% formic acid in acetonitrile |
|
Flow rate: |
500 μL/min |
|
Column temp.: |
40 °C |
|
Sample temp.: |
10 °C |
|
Strong needle wash: |
70/30 ACN/water with 2% formic acid |
|
Weak needle wash: |
70/30 ACN/water with 2% formic acid |
|
Injection mode: |
Partial loop with needle overfill |
|
Injection volume: |
1 μL |
|
Time(min) |
%A |
%B |
Curve |
|---|---|---|---|
|
0.0 |
80 |
20 |
— |
|
0.3 |
80 |
20 |
6 |
|
3.0 |
70 |
30 |
6 |
|
6.5 |
30 |
70 |
6 |
|
6.6 |
80 |
20 |
6 |
|
7.0 |
80 |
20 |
6 |
|
MS system: |
Xevo TQ-S |
|
Ionization mode: |
ESI+ |
|
Capillary voltage: |
2.5 kV |
|
Desolvation temp.: |
500 °C |
|
Cone gas flow: |
150 L/Hr |
|
Desolvation gas flow: |
1000 L/Hr |
|
MRM transition monitored: |
See Table 2 |
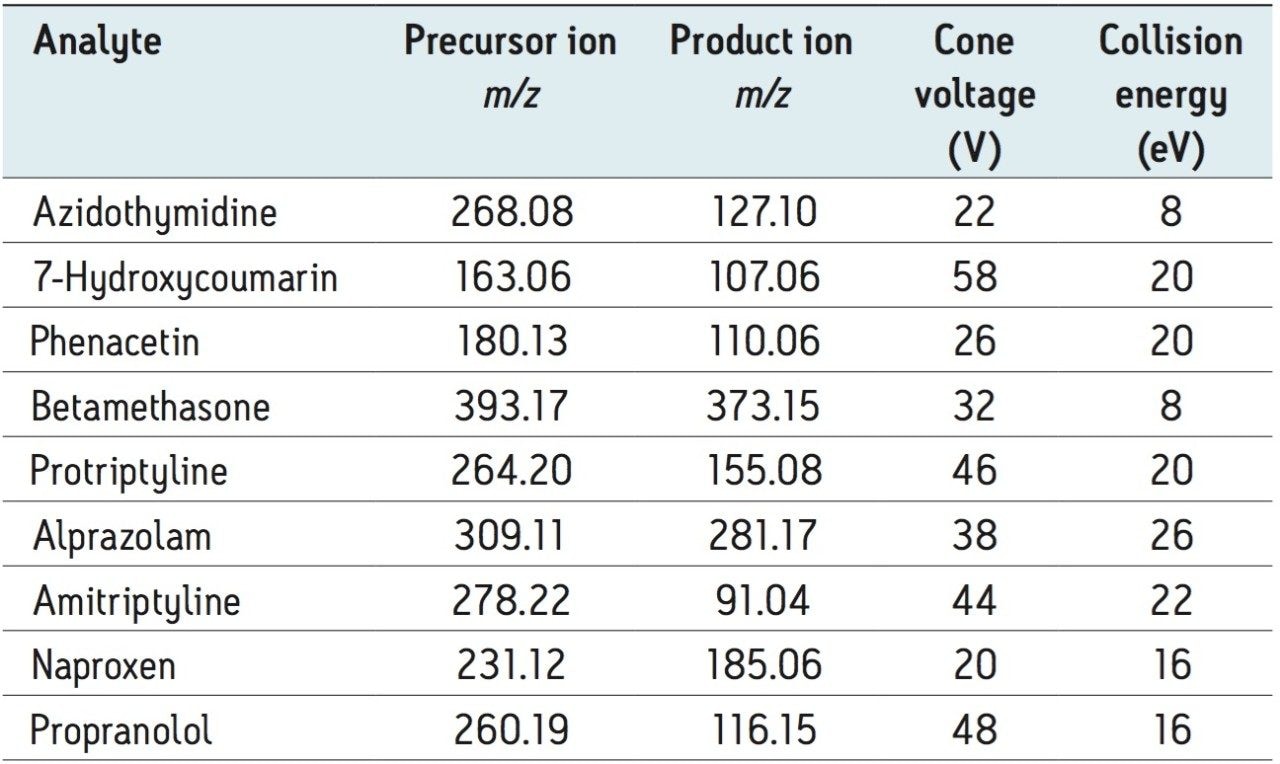
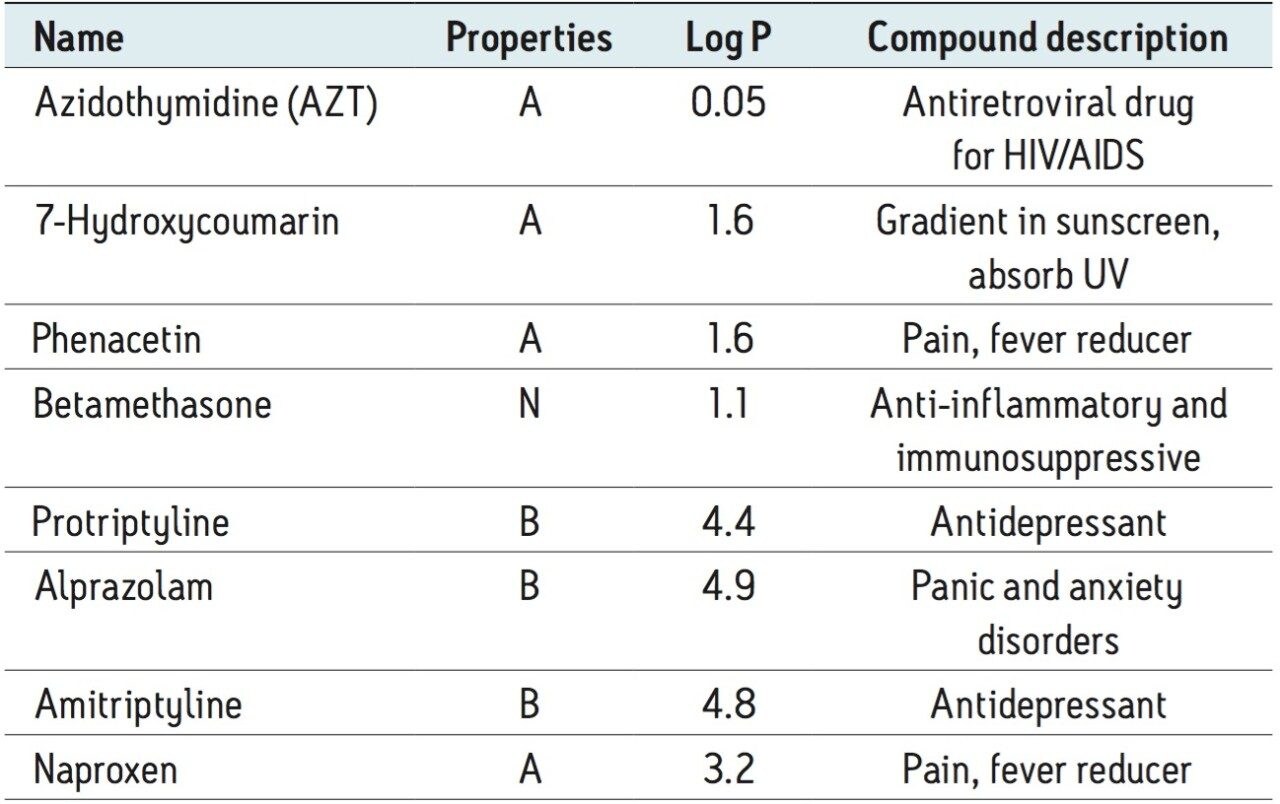
Table 1 lists the tested compounds with their acidic (A), basic (B) or neutral (N) properties, logPs (indication of hydrophobicity) and compound description. Stock solutions were prepared in 75% acetonitrile and a working solution containing a mixture of the analytes was prepared in 40% acetonitrile. The concentration for working solutions is 0.2 μg/mL, except Azidothymidine (AZT), which was 3 µg/mL. 10 µL of each working solution was spiked into 240 µL plasma, the spiked plasma was then diluted with 4% H3PO4 at 1:1 ratio before loading onto the SPE devices. Instead of using the working solution for the blank plasma samples, a 40% acetonitrile solution was used as the spike. Similar sample pretreatment was applied to urine sample prior loading.
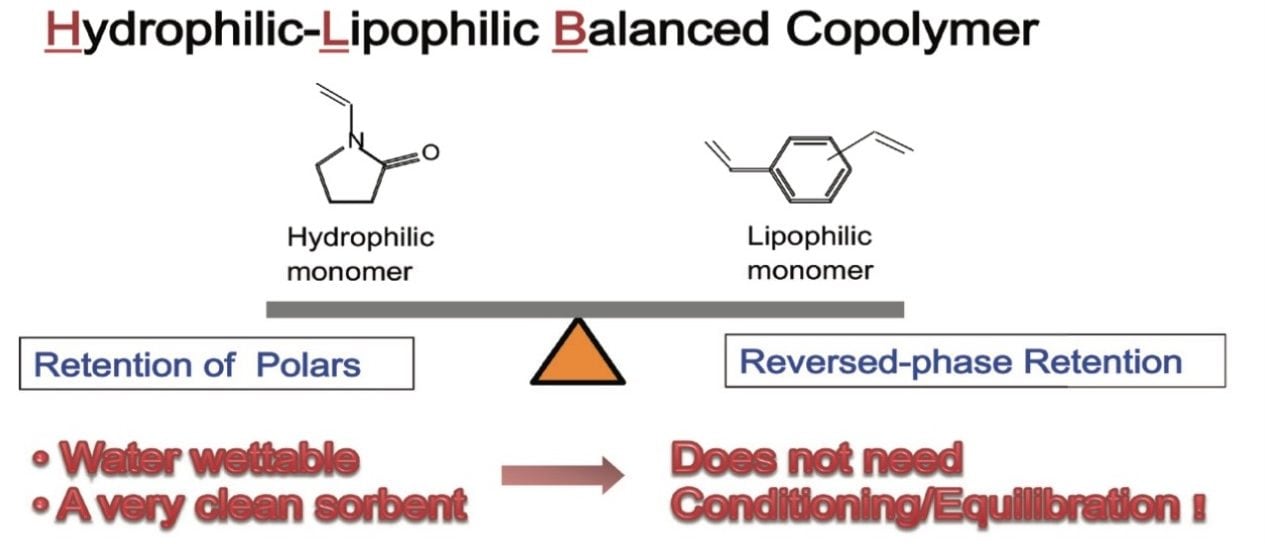
For the Oasis HLB µElution plate (p/n: 186001828BA), the standard sample preparation protocol consists of 5-step and includes conditioning, equilibration, sample loading, washing, and eluting. Conditioning and equilibration steps are used to solvate and then remove the solvent, respectively, so that analytes can interact with the sorbent. These steps are necessary for sorbents that are not water-wettable, like silica-based C18 and hydrophobic polymer-based sorbents. Since Oasis HLB is a water-wettable sorbent, the analytes interact with the sorbent and are retained even when directly loaded in an aqueous sample solution, thus allowing for the elimination of the conditioning and equilibration steps and reducing the number of processing steps from 5 to 3. This results in an average reduction in solvent consumption of 70% and a 40% saving in sample preparation time. These benefits are due to the balanced nature of the hydrophilic/lipophilic surface (Figure 1).
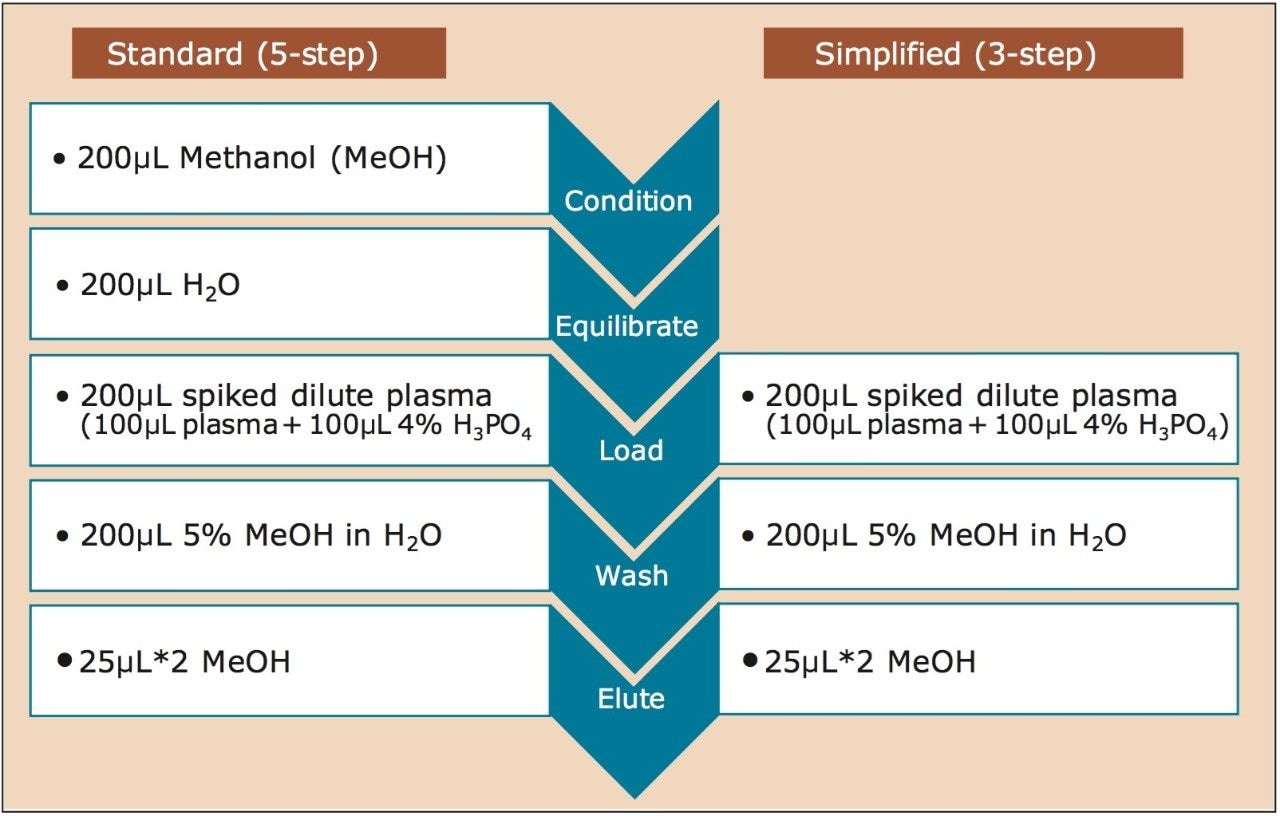
For example, it takes 1 hour to process a 96-well plate with standard, 5-step SPE protocol. Using the simplified 3-step protocol shown here, it takes only 36 minutes to process this same 96-well plate. In an eight hour work day, instead of 8 plates, 13 plates can be easily processed with this simplified protocol. Figure 2 shows the comparison for the 5-step and 3-step protocols for the µElution plate. For 1 cc, 30 mg cartridges (p/n: 186001879), all steps are the same for each protocol, except that the loading, washing and elution volumes, are 1 mL each.
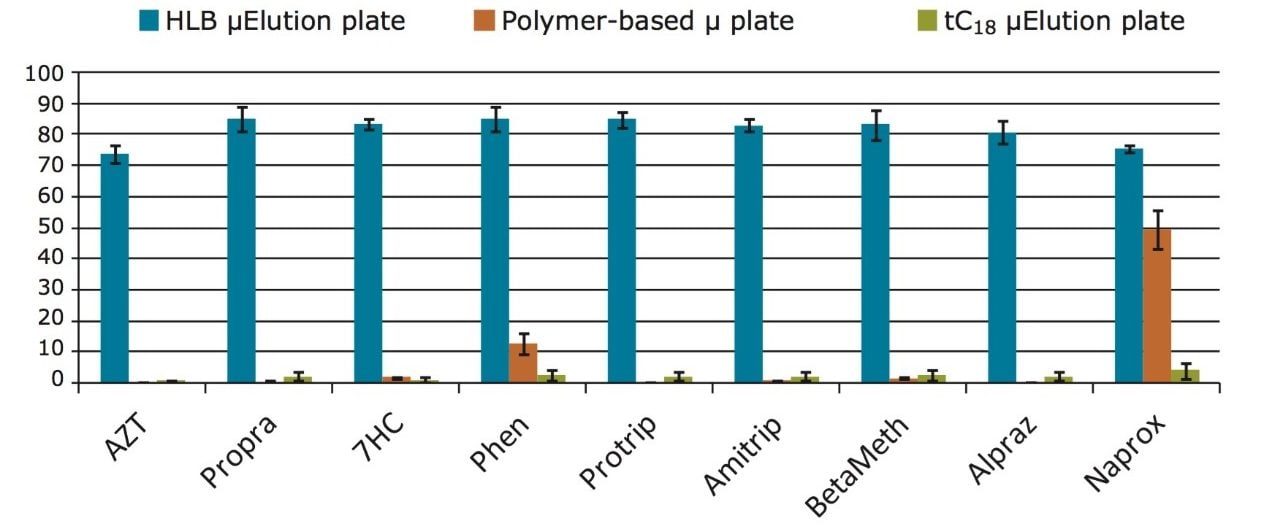
The water-wettable property of the Oasis HLB sorbent allows scientists performing bioanalysis to simplify the SPE protocol by eliminating the conditioning and equilibration steps. These steps cannot be eliminated when using most types of SPE sorbents. To prove this, recoveries were compared between Oasis HLB, a silica-based C18 sorbent, and a competitive polymer-based sorbent in the micro-elution plate format using rat plasma samples. With the simplified protocol, the silica-based C18 and the competitive sorbents showed very low recoveries (Figure 3). In contrast, the Oasis HLB sorbent performed very well with this 3-step protocol, indicating that non-water-wettable or partially water-wettable sorbents cannot be used for simple sample preparation in bioanalytical studies.
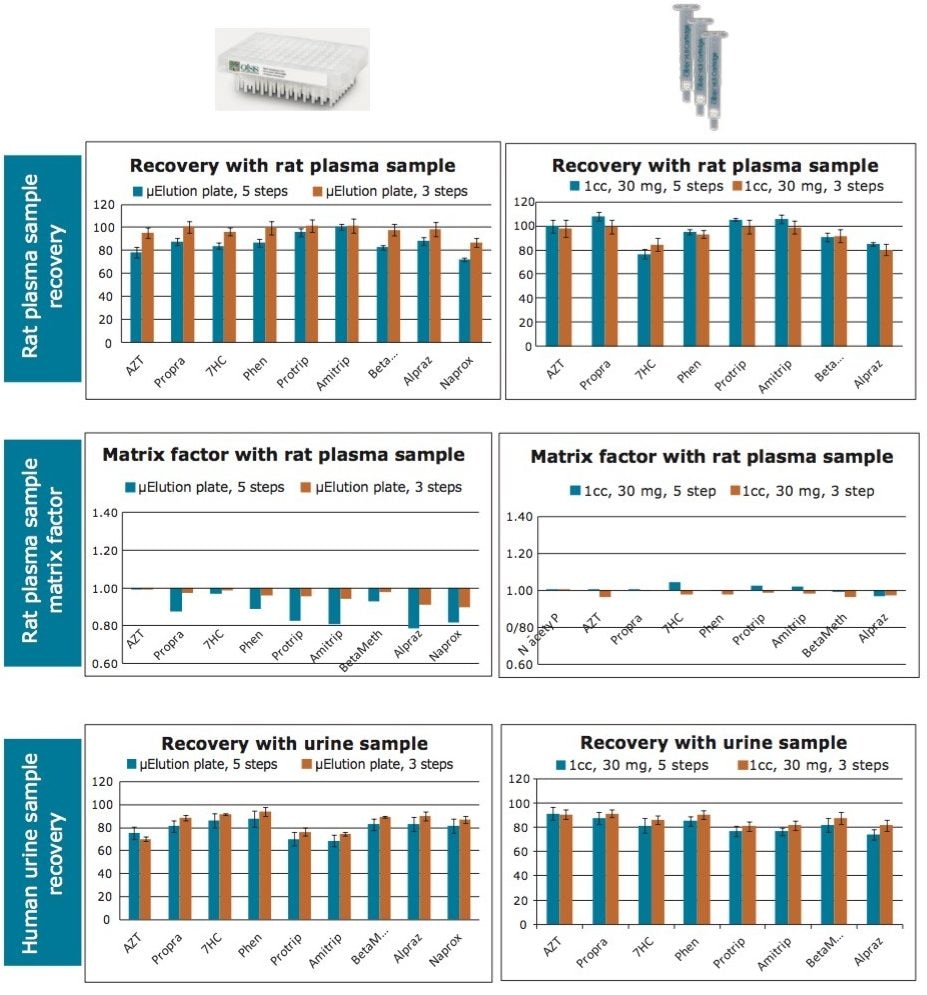
Next, recovery and matrix factors were calculated for both the standard and simplified SPE protocols with Oasis HLB for both rat plasma and human urine samples in both the µElution and 1 cc cartridge formats. The percent recovery was obtained by the ratio of the peak area of a pre-spiked, extracted sample to the peak area of a post-spiked, extracted sample. The matrix factor was calculated as the ratio of the peak area in the presence of matrix (measured by analyzing blank matrix spiked after extraction with analytes) to the peak area in absence of matrix (solvent standard solution of the analytes).
Comparable recoveries were obtained for both rat plasma and human urine, with the average recovery greater than 80% and standard deviation less than 5%. Equivalent matrix factors were obtained in both biological samples (rat plasma and human urine) using both protocols. Matrix factors of 1.00±0.15 were obtained with the simplified protocol. Recoveries and matrix factors are summarized in Figure 4. These data show that independent of device format, the simplified, 3-step protocol on Oasis HLB can be used successfully for a wide range of analytes for routine bioanalytical studies. The µElution plate is strongly recommended when there is limited sample volume available or analytes need to be concentrated (up to 15X concentration) without the need for evaporation or reconstitution. Cartridges can be used individually and are suggested when extracting a smaller number of samples that do not need concentration, or need limited concentration.
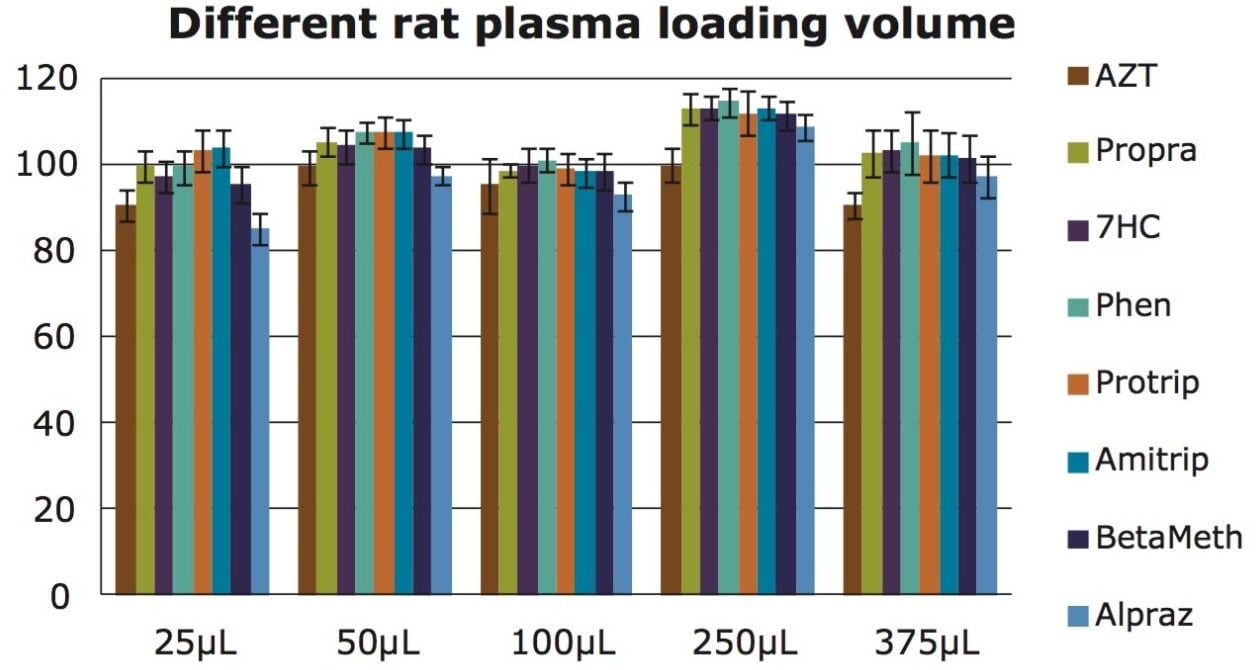
One potential concern with using a simplified protocol could be the volume loading capacity. In order to prove the 3-step protocol can be used with commonly used volumes in bioanalysis, we tested the simplified protocol with plasma volumes between 25 and 375 µL (µElution plate format). As shown in Figure 5, the simplified protocol provides 80–110% recoveries with standard deviations of less than 5%, which confirms that the loading capacity is acceptable and does not require pre-conditioning and equilibration.
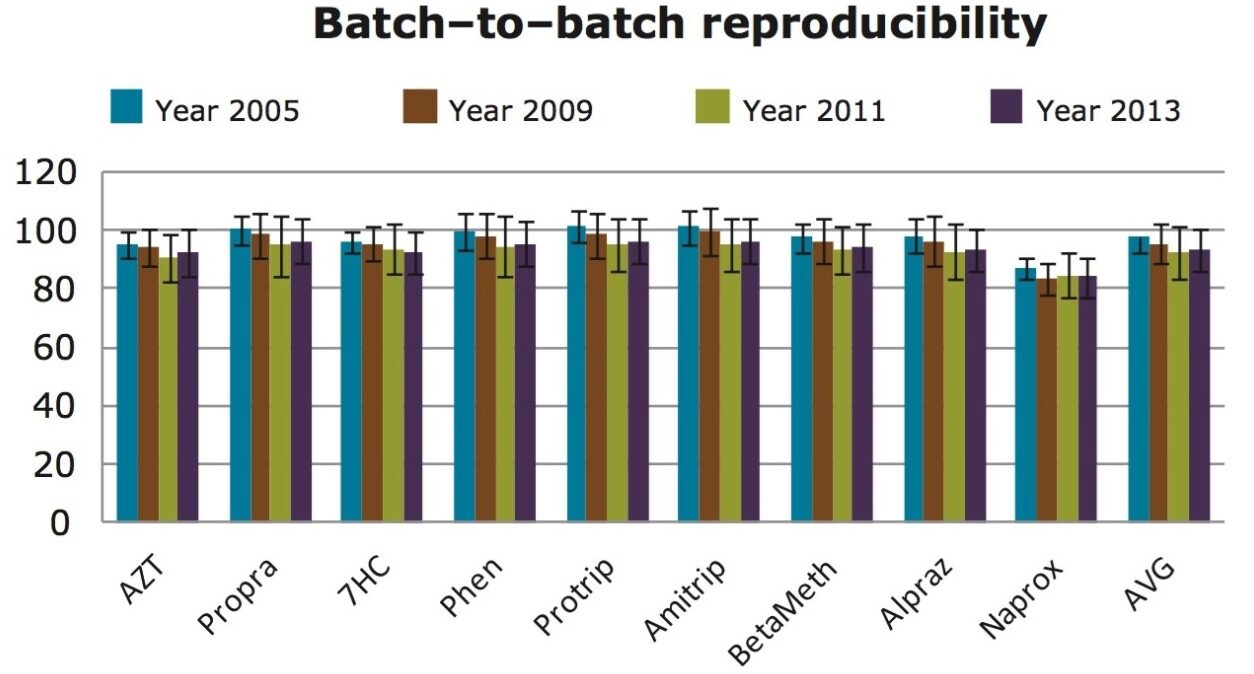
A critical attribute of any SPE method in bioanalysis is the ability to obtain the same results over a long period of time (life of drug). Therefore, it is important to evaluate the batch-to-batch reproducibility of the SPE method. To prove the long term viability of the simplified SPE approach on Oasis HLB, an experiment was performed with µElution plates from four different batches of Oasis HLB produced between the years 2005 and 2013. The recoveries were between 80–100 % with less than 6% standard deviation (n=4) (Figure 6). These data show that the results with a simple, 3 step SPE protocol give the reproducibility needed for a bioanalytical assay that spans the life of a drug while saving 40% of the time and 70% of the solvent compared to a 5 step protocol.
A simplified, 3-step protocol for SPE in bioanalysis using Oasis HLB enables:
720005140, September 2014