For research use only. Not for use in diagnostic procedures.
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates a highly selective solid-phase extraction (SPE) technique with Oasis MAX μElution Plates for the extraction of testosterone from serum samples for LC-MS/MS analysis in clinical research.
The Oasis MAX μElution Plate enables optimized analytical sensitivity and selectivity for the analysis of testosterone in serum for clinical research.
To determine the pharmacokinetic, bioavailability, and safety of new formulations of testosterone gels in clinical research, accurate and precise quantification of testosterone at low physiological concentrations is required.1
Historically, testosterone has been analysed by direct immunoassay methods. However, it has been reported that these immunoassay methods can suffer from a lack of specificity due to the cross reactivity of structurally similar steroid hormones i.e., dihydrotestosterone. This results in greater imprecision and inaccuracy, most notably at the lower concentrations of testosterone found in serum samples. LC-MS/MS provides a solution to this problem by providing a platform to discern between similar steroids through the use of chromatographic separation and selective MRM mass spectrometry.
Additional selectivity can also be obtained through the use of sample preparation prior to analysis. The use of sample preparation can provide orthogonal selectivity to the LC-MS/MS dimension of the method. Potential interferences found in the MRM trace of the analyte of interest can be removed with selective sample preparation techniques such as mixed mode SPE.
Serum samples were diluted with internal standard, ammonia, zinc sulphate, methanol, and water. Following centrifugation, sample supernatant was loaded onto the 96-well Oasis MAX μElution Plate (PN:186001829) following conditioning and equilibration. A high pH wash was performed with ammonia in 20% methanol and samples were eluted with methanol followed by water.
20 μL of each extracted sample was injected on an ACQUITY UPLC I-Class/Xevo TQD utilizing mobile phases consisting of 2 mM ammonium acetate and 0.1% formic acid in water and methanol. Separation was performed on an ACQUITY UPLC HSS C18 SB Column (PN:186004118). The MRM transitions used for this analysis were m/z 289.2 for the testosterone precursor ion and m/z 96.9 and 109.0 for the quantifier and qualifier product ions respectively. Testosterone-13 C3 was used as the internal standard.
In this technology brief, an Oasis MAX μElution Plate was successfully employed for the selective extraction of testosterone from serum. This chemistry provides mixed-mode interaction which utilizes both hydrophobic and anion exchange functionality. The added selectivity of this sorbent is more proficient at removing interferences than conventional reversed-phase SPE.
The additional selectivity provided by the Oasis MAX chemistry compared to other extraction techniques, provides enhanced analytical sensitivity for ESI MS analysis of testosterone. This is observed in the extraction and quantification of testosterone in serum using optimized Oasis MAX μElution, Oasis HLB μElution, and protein precipitation extraction protocols. The largest S/N ratio observed for the quantifier ion of testosterone was obtained using the Oasis MAX μElution Plate for the sample preparation (Figure 1).
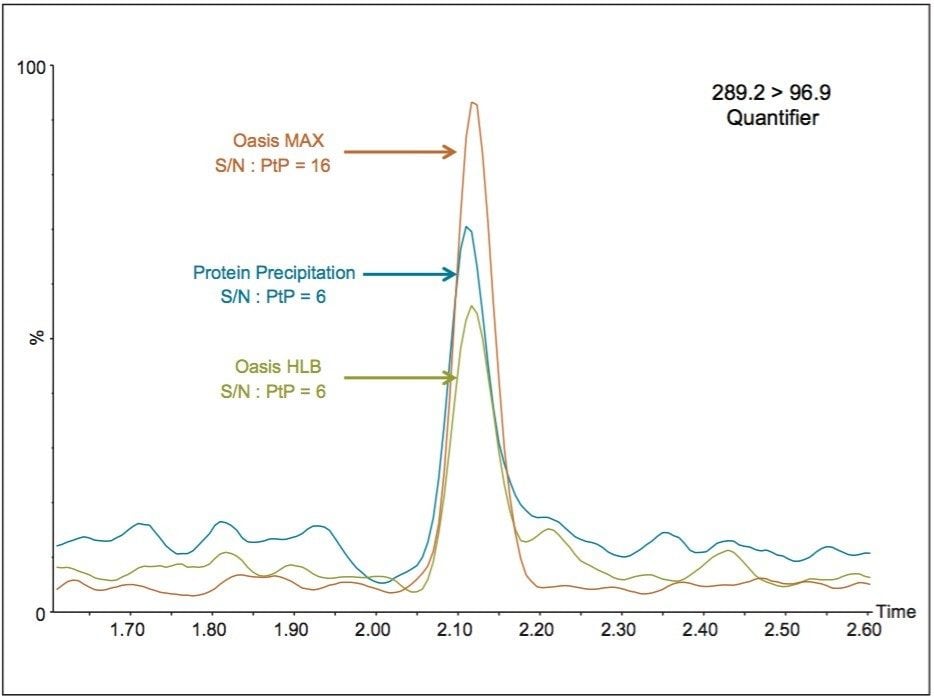
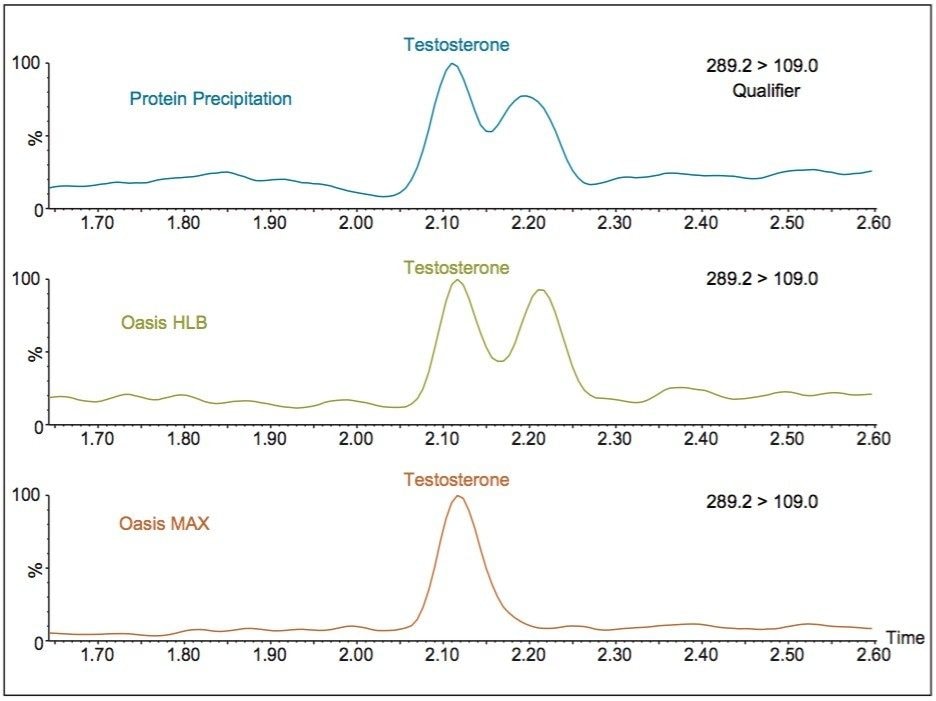
The Oasis MAX μElution Plate also removes an interference clearly present in the qualifier trace of the same serum samples, as can be seen in Figure 2. The same interference is also evident in Figure 1. This selectivity improves the accuracy of the ion ratios observed between the quantifier and qualifier ions as well as peak integration reproducibility.
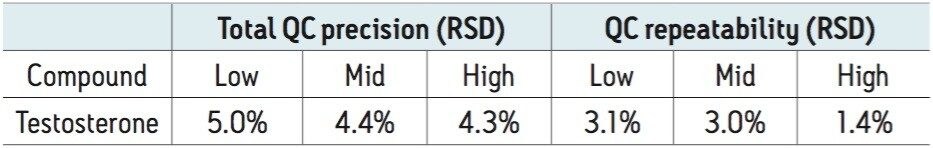
Over a calibration range of 0.17–51 nmol/L for testosterone the precision of the method was determined by extracting and quantifying three replicates of tri-level QC material on two occasions per day over five separate days (n = 30). The results of these experiments are seen in Table 1, where total precision and repeatability is ≤5.0% RSD.
A LC-MS/MS method for the analysis of testosterone for clinical research has been developed. Sample preparation was performed using an Oasis MAX 96-well μElution Plate for the extraction of testosterone from serum. The benefits of this method include:
720005224, December 2014