This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates to evaluate chromatographic retention time and system reproducibility of Waters ionKey/MS System.
ionKey/MS: Enhanced MS with the turn of a Key. More sensitivity, more information from the same sample, more robust analysis. All with less solvent usage and less complexity to the user.
Chromatographic reproducibility is an essential requirement in any analytical laboratory environment. Parameters such as chromatographic retention time (RT), peak width (PW), peak tailing, and area counts are often monitored during system suitability testing before assays can begin on any liquid chromatography system. A critical component to obtaining highly reproducible LC-MS results is the consistent performance of the separations devices utilized. Chromatographic retention time reproducibility is not only dependent on the quality and consistency in the manufacturing of the separations devices, but is also effected by gradient formation, system pressure, flow rate and temperature control. Historically, these later variables, controlled by system hardware, have been shown to be a source of poor performance and reproducibility of results for LC-MS systems that operate in the low nL-μL/min flow rate ranges.
The ionKey/MS System, shown in Figure 1, integrates the UPLC separation directly into the source of the mass spectrometer. The system combines the ACQUITY UPLC M-Class and the Xevo TQ-S Tandem Quadrupole Mass Spectrometer with the iKey Separation Device. The iKey consists of the ceramic-based separations channel, packed with UPLC grade particles, an integrated emitter, fluidic connections, electronics, ESI interface, heater, and e-cord. Once inserted into the source, all fluidic connections are made through the turn of a handle thereby creating a true “plug and play” capillary LC-MS instrument.

Figure 2 illustrates the separation of a peptide standard utilized in the evaluation of the system. As can be observed in Figure 2, the overlay of fifteen different analysis of the peptide standard illustrates the excellent reproducibility of the ionKey system. The data from this evaluation is further displayed in Table 1. The data illustrates average retention times of 6.84 minutes for the peptide standard with 0.1 % RSD. In this evaluation, the peptide standard data was acquired over a course of 500 injections of rat plasma prepared via protein precipitation, utilizing a 15 minute gradient equating to five days of continuous operation.
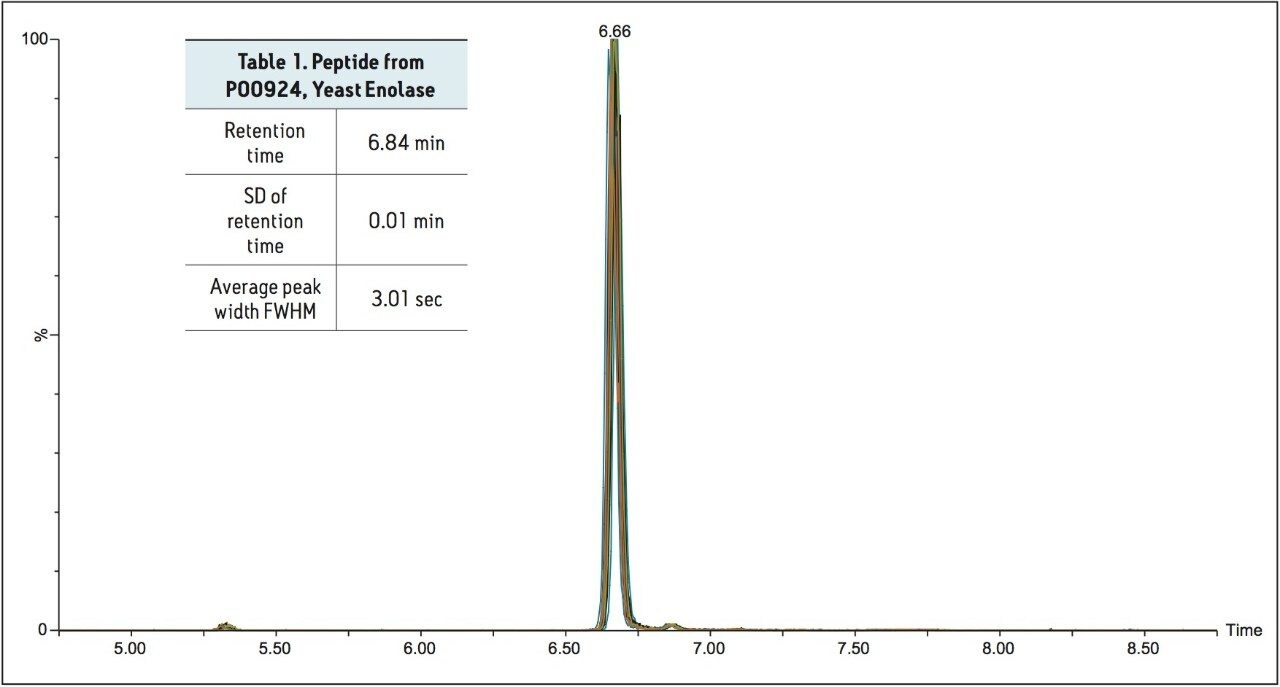
This same peptide standard and methodology was then run on 18 different iKeys, on 5 independent ionKey/MS Systems operated by 5 different scientists. The data from these tests is displayed in Table 2. As can be observed in this table, retention time reproducibility values for the 18 iKeys tested, had an average retention time of 6.69 minutes with 1.6 % RSD. This data clearly illustrates the reproducibility of each of the individual iKey Separation Devices as well as the reproducible control of system pressure, flow rate, gradient formation and temperature control afforded by the individual ionKey/MS Systems utilized in the evaluation.
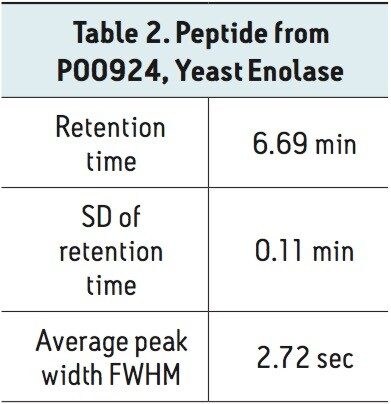
The data shown here demonstrates the excellent chromatographic retention time reproducibility for a peptide standard injected on multiple iKeys during robustness testing of protein precipitated rat plasma. Moreover, the evaluation illustrated the excellent reproducibility between multiple ionKey/MS Systems operated by 5 independent scientists.
720004951, March 2014