
In this application note, we use the comparison of Shuanghuanglian oral drink from two different manufacturers as an example to illustrate the use of the Natural Products Application Solution with UNIFI. The solution combines the ACQUITY UPLC I-Class System, the Xevo G2-S QTof MS, as well as a Traditional Medicine Library, and uses the UNIFI’s binary comparison workflow to compare similar samples for the purpose of quickly identify differences between the two samples.
Using the binary comparison workflow available with the Natural Products Application Solution with UNIFI, this application demonstrates the fast and accurate detection of the differences and similarities of chemical ingredients in Shuanghuanglian oral drink from two different manufacturers. The solution improves researchers’ ability to understand ingredient differences of the same product manufactured by different companies, and it enables more accurate quality control of a product between batches made by a single manufacturer. UNIFI Software provides a simple and effective approach to all binary comparisons among similar samples.
Traditional Medicine is as complex as it is amazing, having gone through development for thousands of years. However, there has not been a well accepted, systematic, and scientific method to evaluate the authenticity and quality of traditional medicine products. For example, there has not been a consistent and effective method for the control of the active ingredients of raw materials in the Traditional Chinese Medicine (TCM). The same is true for methods to evaluate raw materials with different planting techniques and location, or different harvest times, or different processing methods.
In recent years, pharmaceutical researchers have carried out many studies focused on improving Traditional Medicine quality standards, and have already accumulated good amount of valuable experience and methods. Still, generally speaking, binary comparison of Traditional Medicine is time-consuming and expensive. In addition, some methods lack scientific support and can be not very objective. This application note provides a novel approach by incorporating advanced separation and detection technologies with the Natural Products Application Solution with UNIFI.
Similarities and differences in chemical ingredients of raw material from different locations, differences in the same product produced by different manufacturers, and product quality from different batches by one manufacturer all require a comparison of similar samples. The goal is to provide a clear direction for further R&D by identifying any differences via observing the chemical ingredients of each sample. With the constant evolution in analytical technology, the application of high-resolution LC-MS instrumentation such as UPLC/QTof MS has been gaining steadily in acceptance. Application of these technologies can help shorten analysis times and increase separation efficiency. Meanwhile, UPLC/QTof MS offers advantages in wide adaptability, high specificity, low detection limits, small injection volumes, low solvent consumption, highly automated workflows, strong capability for identification, etc. As a result, analytical technologies are playing a more important role for quality control of traditional medicines.
In this application note, we use the comparison of Shuanghuanglian oral drink from two different manufacturers as an example to illustrate the use of the Natural Products Application Solution with UNIFI. The solution combines the ACQUITY UPLC I-Class System, the Xevo G2-S QTof MS, as well as a Traditional Medicine Library, and uses the UNIFI’s binary comparison workflow to compare similar samples for the purpose of quickly identify differences between the two samples; thus providing the scientific basis for better quality control and generating quality standards at later stage. As a result, productivity is greatly improved and the requirements for the operator’s expertise level is significantly reduced.
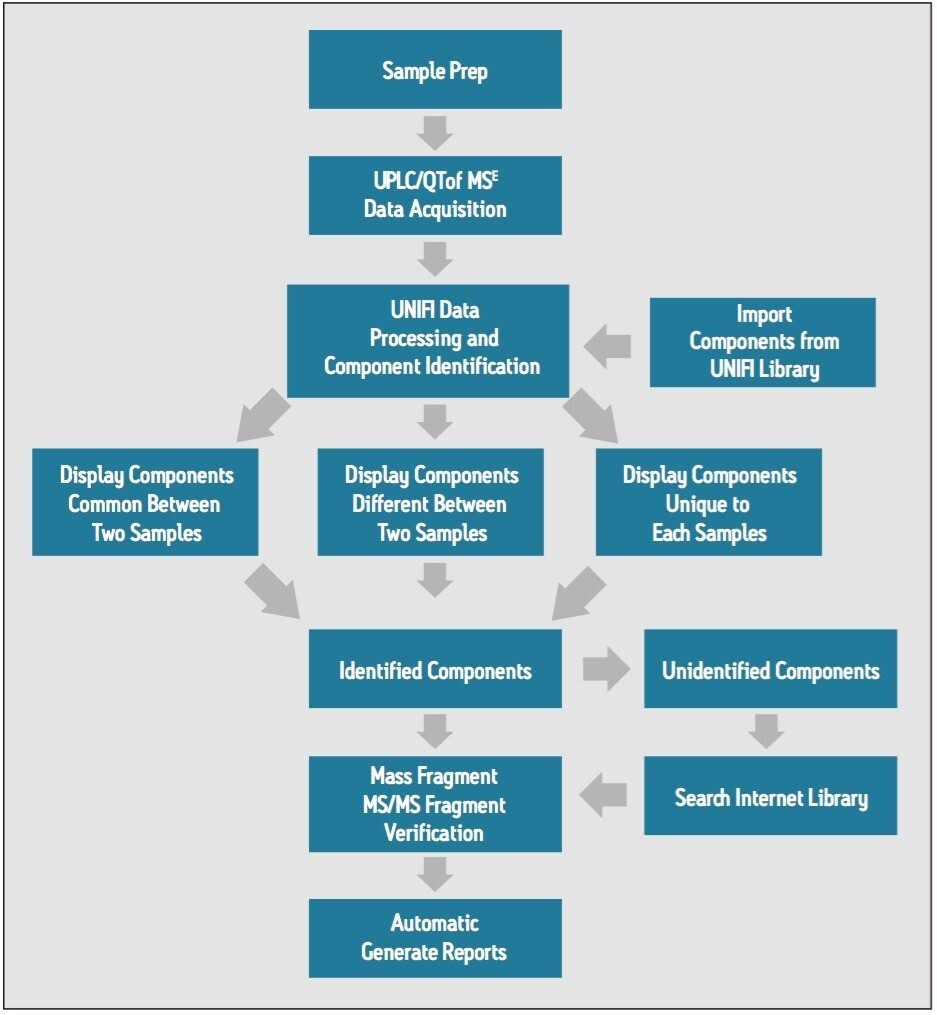
The Shuanghuanglian oral drink contains three herbs: JinYinHua (lonicerae japonicae flos), known as honeysuckle; HuangQin (scutellariae radix), known as scutellaria; and LianQiao ( forsythiae fructus), known as forsythia. The product is used to alleviate symptoms caused by upper respiratory tract infections, tonsillitis, pharyngitis, and bacterial and viral infectious diseases such as viral pneumonia, etc. Within this oral drink, known active ingredients include compounds such as Baicalin, forsythin, and wogonin. Being popular on the market, this product is being made by numerous manufacturers in China. Therefore, we decided to use this representative application example to illustrate the binary comparison workflow provided within the Natural Products Application Solution with UNIFI (Figure 1).
200 μL Shuanghuanglian oral drink was dissolved in 2 mL H2O (effectively 10X dilution) for later use. Injection volume was 1 μL.
|
LC system: |
ACQUITY UPLC I-Class with FTN Sample Manager |
|
Column: |
ACQUITY UPLC HSS T3 2.1 x 100 mm, 1.8 μm |
|
Column temp.: |
40 °C |
|
Sample temp.: |
15 °C |
|
Mobile phase: |
A: water (0.1% formic acid); B: acetonitrile |
|
Time |
Flow rate (mL/min) |
Solvent A(%) |
Solvent B(%) |
Curves |
|---|---|---|---|---|
|
0 |
0.5 |
95 |
5 |
Starting |
|
1 |
0.5 |
90 |
10 |
6 |
|
8 |
0.5 |
55 |
45 |
6 |
|
12 |
0.5 |
45 |
55 |
6 |
|
13 |
0.5 |
5 |
95 |
6 |
|
15 |
0.5 |
95 |
5 |
1 |
|
MS system: |
Xevo G2-S QTof MS |
|
Acquisition range: |
100-1500 Da |
|
Scan time: |
0.1 s |
|
Acquisition mode: |
ESI+, ESI-; resolution mode; MSE |
|
Lock mass: |
Leucine Enkephalin (LE) 1 ppm (scan for 0.3 s, interval: 15 s) |
|
Capillary voltage: |
3 KV (ESI+)/2.5 KV (ESI-) |
|
Cone voltage: |
100 V |
|
Collision energy (eV): |
Low CE: 6/High CE: 20-45 |
|
Source temp.: |
120 °C |
|
Desolvation temp.: |
500 °C |
|
Cone gas flow: |
60 L/h |
|
Desolvation gas flow: |
1000 L/h |
|
Acquisition time: |
15 min |
UNIFI Scientific Information System
UPLC and quadrupole time-of-flight mass spectrometry were used to acquire analytical data of the chemical ingredients in Shuanghuanglian oral drink from two different manufacturers. The binary comparison workflow in Natural Products Application Solution with UNIFI combining with its Traditional Medicine Library was used for data mining. The results obtained have the following information clearly displayed: summary plot of components that are common, summary plot of components that are different, as well as the tables for components that are unique to each sample. Final results indicated that ingredient contents such as forsythiaside A, forsythiaside B, dextrorotation hydroxyl forsythiaside A, arctin, and rutin were higher in manufacturer 1, while ingredient contents such as baicalin was higher in manufacturer 2.
The Natural Products Application Solution with UNIFI includes five preset binary comparison workflow templates, and one report template. As a result, the entire analytical process can be automated from data collection, to peak picking, to library search and structural elucidation (of fragment ions by MassFragment), to producing the final report.
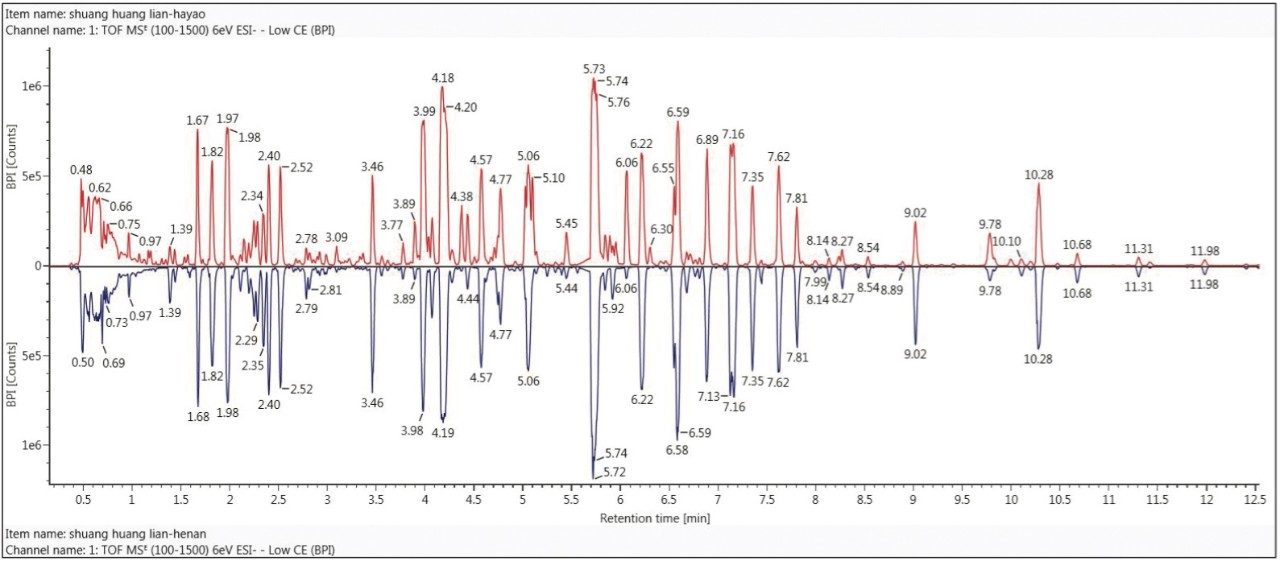
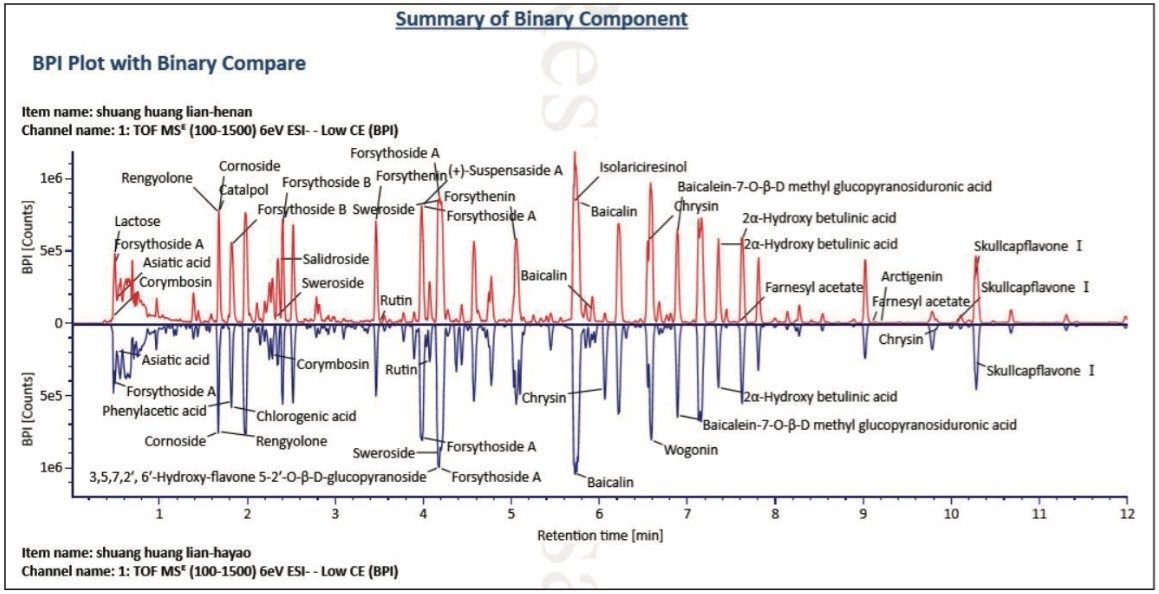
The UPLC/QTof MS base peak ion (BPI) chromatograms of the two Shuanghuanglian oral drink products were compared with the mirror (or, binary) plot, shown in Figure 2. This plot clearly demonstrates the advantages of using UPLC for the analysis of complex natural products. UPLC provides not only a shorter run time (i.e., an effective separation time of 12 min), but also higher separation efficiency and peak capacity. Meanwhile, QTof MS also provides exact mass MS data. In addition, as shown in Figure 2, the chemical ingredients of the two samples are not only complicated, but also very similar. Without the help of an informatics platform to quickly and accurately identify similarities and differences from samples, this is a complex task that is nearly impossible to accomplish.
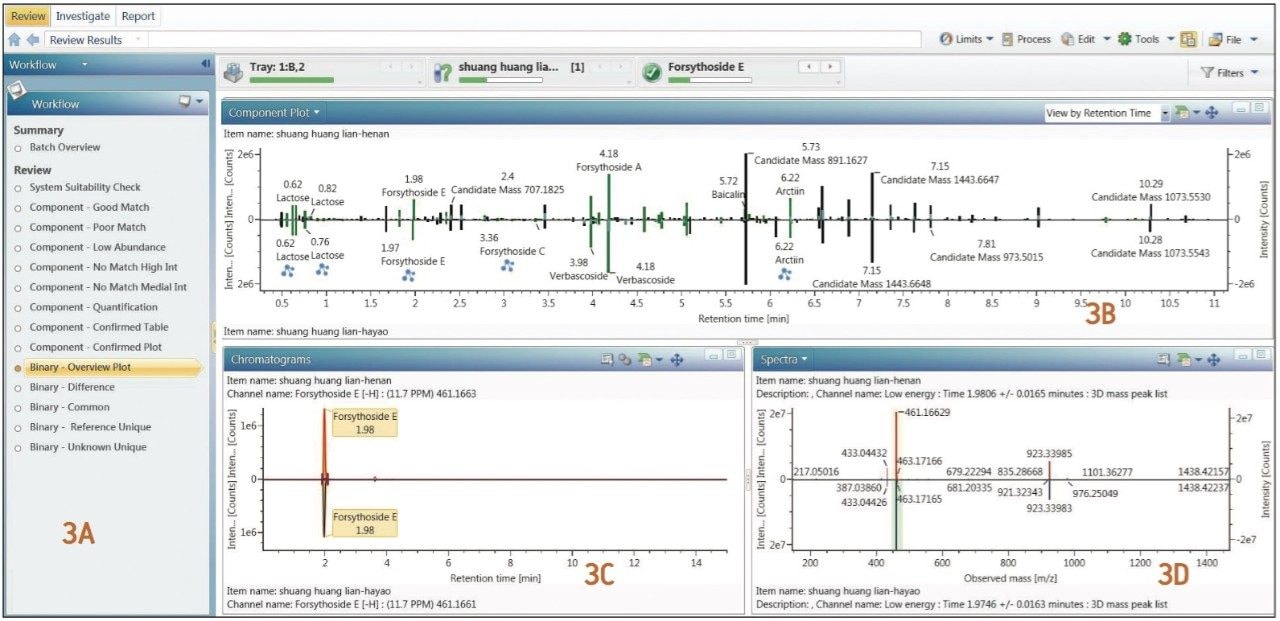
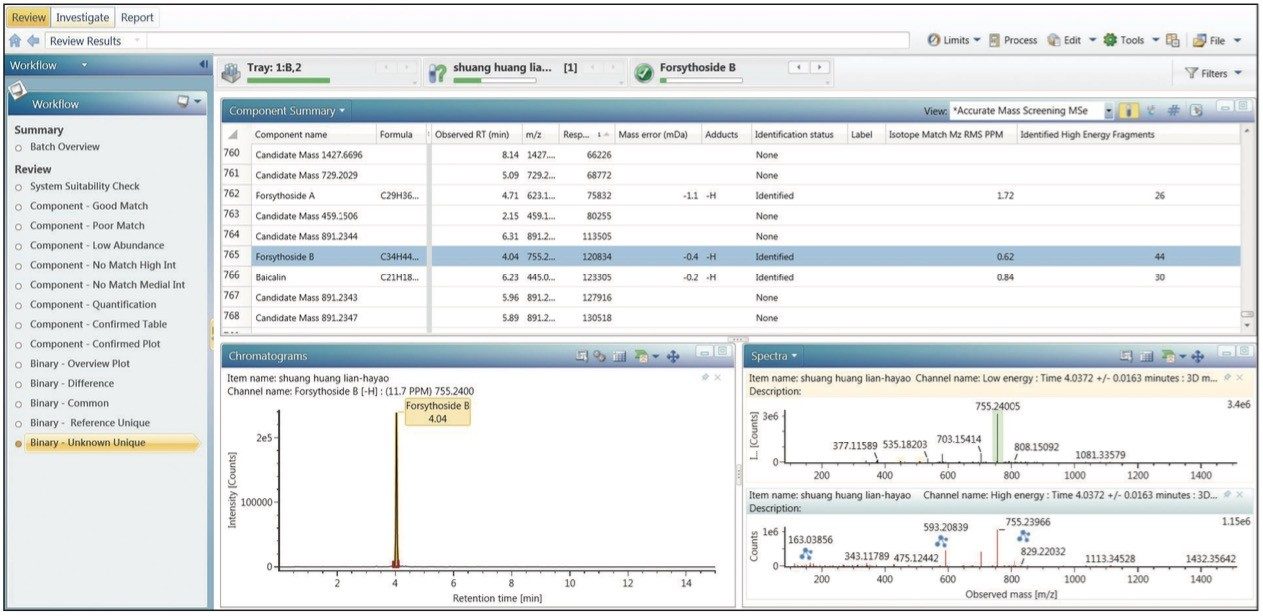
In the natural products field, having a clear understanding of the ingredients in detail is the foundation for all future steps for any research, whether for qualitative identification of chemical ingredients from a single sample, or for component comparison of two different samples. In a previous application note,1 we have described the analytical workflow of ingredient identification for samples with known plants. Following this workflow, all chemical ingredients related to the three herbal plants (honeysuckle, scutellaria, and forsythia) were imported into the component list from the Traditional Medicine Library for data processing. This was then followed by verifying the identified components in a manner described in previous application note.1 Respective results can be easily viewed from the Review window of UNIFI by clicking any of the five preset binary comparison templates. For example, Figure 3 shows the result displayed after clicking the Binary- Overview Plot template.
Figure 3A shows the 14 preset workflow templates in UNIFI. Figure 3B is the comparison of the UPLC/QTof MS component summary plots for samples from two manufacturers. Here, green bars represent ingredients that were matched and automatically identified with the Traditional Medicine Library; the black bars represent ingredients that did not have a match and thus cannot be automatically identified. Figure 3C shows the mirror plot of the selected ion chromatograms (XIC) corresponding to any component that user had clicked from Figure 3B (here, it displayed the XIC mirror plot of forsythoside B at retention time 4.03 minutes). Figure 3D is the mirror plot of the low-energy MS spectra of the ingredient (shown in 3C) from the two samples.
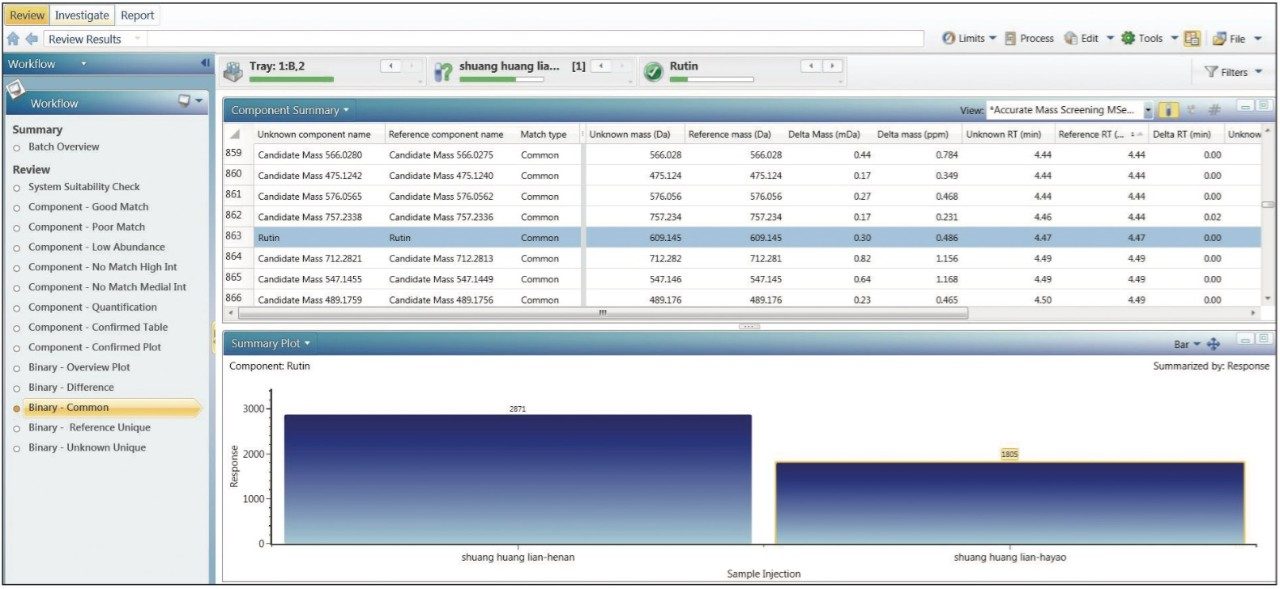
Further understanding of the similarities and differences between these two samples can be obtained by clicking either the Binary-Common workflow or the Binary-Difference workflow. Ingredients that are common or different between the two samples can be directly observed. For example, Figure 4 shows the results provided by the Binary-Common workflow. Figure 4A lists the components that are common in two samples (total of 1978 from preliminary result). Figure 4B shows levels of MS responses in these two samples for the each of the components listed in Figure 4A, which is a reflection of the relative amount of this ingredient in each sample (wogonin in this case). Similar information is presented if the Binary-Difference workflow is clicked, and the results will show ingredients that are different between these two samples (total of 408 from preliminary result).
Using the Binary-Common and Binary-Difference workflow templates, the ingredients that are common and different for these two samples can also be shown in bar chart format, which can be more intuitive. For example, the ingredient difference of these two samples obtained by clicking Binary-Difference can be easily viewed by clicking Component Summary Plot from the Component Summary list (Figure 5). This plot allows researchers to observe the differences between two samples clearly and intuitively.
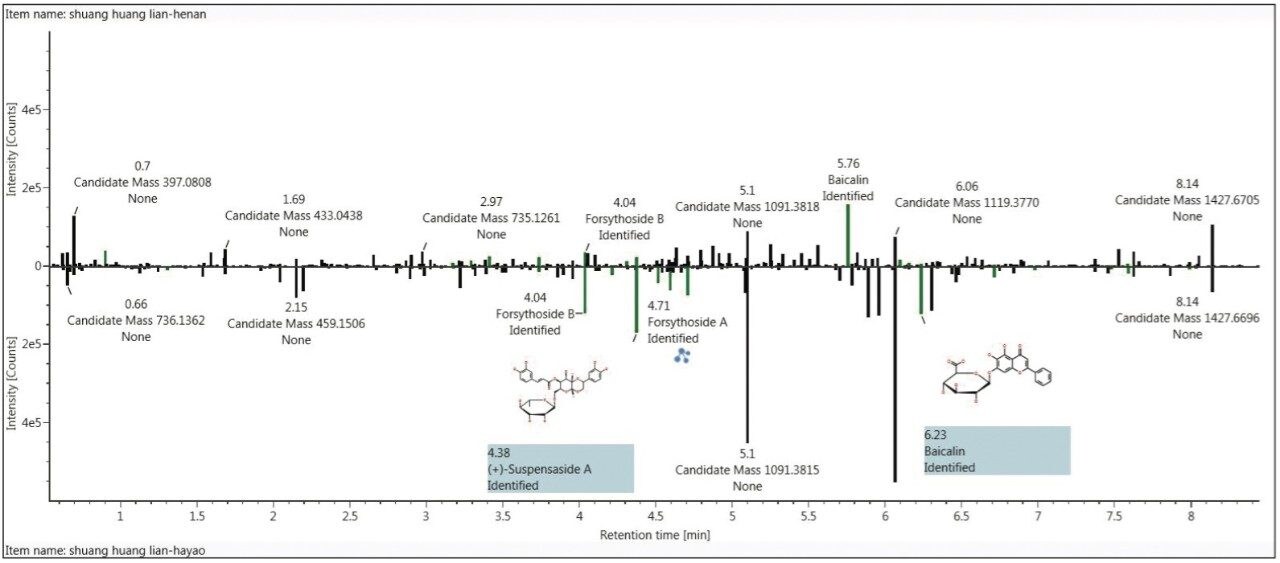
The results of analyzing these two traditional medicine drinks suggested that the baicalin content from manufacturer 2 is significantly higher than that of manufacturer 1, while the content of forsythiaside A, forsythiaside B, dextrorotation hydroxyl forsythiaside A, etc. are much higher in manufacturer 1 than that of manufacturer 2. Figure 5 also clearly shows that there are components that were not identified, being not matched with the Traditional Medicine Library. In this case, we can use UNIFI’s Structural Elucidation Tool for further identification should it be necessary.
Details of the component differences in two samples can be further investigated by applying either the Binary- Reference Unique or Binary-Unknown Unique templates. Components that are unique to each of the samples can then be studied in detail. UNIFI binary comparison requires one sample to be defined as a reference sample (chromatogram or MS on top) and the other as an unknown sample (chromatogram or MS on bottom). In this case, we arbitrarily defined the sample from manufacturer 2 as the reference, and sample from manufacturer 1 as the unknown. To further study the unique ingredients from the manufacturer 1 sample, one only needs to click Binary-Unknown Unique, shown in Figure 6. Results suggest that there are 193 components that are unique in the manufacturer 1 sample.
If satisfied with the confirmed results, the report can be easily obtained and printed by using the NP Binary template. This report presents information such as sample preparation, data collection, and processing method, as well as detailed lists of components common and different from the two samples. Figure 7 shows another part of this report, which is the mirror plot of the BPIs of these two samples, where peak identities are labeled automatically providing results that are visually intuitive.
This application note systematically describes how to apply the binary comparison workflow of the Natural Products Application Solution with UNIFI to compare the ingredient similarities and differences between Shuanghuanglian oral drink from two different manufacturers. This workflow is based on collecting data by UPLC/QTof MS, and using the Traditional Medicine Library as well as automated structure elucidation features within UNIFI. The solution provides a novel and comprehensive approach for the natural products analysis.
This solution is applicable for solving a series of problems, such as comparison of raw materials from different locations, or from the same location but different harvesting times or processing methods; it is also useful for quality control of the same product from different manufacturers, or the same products from the same manufacturer but different batches, etc.
This binary comparison solution contains five preset binary comparison workflow templates and one report template. Used together, UNIFI enables researchers to compare samples effectively and quickly to understand the similarities and differences among samples, providing a foundation for future product research steps. The final result is a significant enhancement of effectiveness and productivity for the research and quality control of traditional medicines.
720004842, December 2013