This study investigates the use of both BEH C18 and CSH C18 columns for preparative peptide separations. Based on loading studies with analytical bore columns, the use of 5-μm BEH130 C18 and 5-μm CSH130 C18 shows significant promise for facilitating preparative peptide separations with either TFA or HOAc containing mobile phases. Both BEH130 C18 and CSH130 C18 column chemistries have useful attributes. The following results also show that both BEH C18 and CSH C18 can yield narrower target peaks at high mass loads with optimized HOAc mobile phases than those containing 0.1% TFA.
Peptides have proven to be very useful as both therapeutic agents and markers in research. It is common practice to purify peptides for these purposes by preparative reversed-phase (RP) chromatography. In most instances, it is imperative that the purification process yield peptides of high purity. Contaminants can render results from biological assays ambiguous, and be a serious concern when present in active pharmaceutical ingredients. As a result, there is a need for high chromatographic resolution to minimize the co-elution of impurities that are often closely related chemically to the target peptide. There is also a need for column chemistries with excellent loadability to ensure that throughput and productivity are optimized. Typically, peptides are separated using mobile phases containing strong ion pairing agents, such as trifluoroacetic acid (TFA). However, when mobile phases containing TFA are employed, additional preparation steps are required. It is necessary to remove or exchange trifluoroacetate (TFA salt), because of its inherent toxicity.1 Less toxic counter ions, notably acetate, are preferred. In fact, most peptide pharmaceuticals are either acetate salts or acetate-containing liquid formulations.2-3 It would seem advantageous to avoid TFA mobile phases when possible, instead using acetic acid (HOAc) mobile phases. It has been previously shown that a peptide in a TFA solution, such as a crude synthetic peptide,4-5 is more than partially converted to an acetate form after using HOAc mobile phases and isocratic RP chromatography.6 Meanwhile, gradient separations with short wash steps involving a high concentration of an acetate buffer can be employed for more complete conversion of salt forms.7 In the end, a streamlined purification process involving HOAc mobile phases would facilitate obtaining the desired peptide and counter ion with fewer steps.
This study investigates the use of both BEH C18 and CSH C18 columns for preparative peptide separations. BEH C18 is an organo-silica C18 stationary phase, based on ethylene bridged hybrid (BEH) technology, and is noted for its robustness and pH stability. Charged Surface Hybrid (CSH) C18 is an evolution of BEH C18 as it is modified to contain a low level positive surface charge under acidic conditions. The following data demonstrates that each of these stationary phases is amenable to high mass load peptide separations with both TFA- and HOAc-modified mobile phases. The following results also show that both BEH C18 and CSH C18 can yield narrower target peaks at high mass loads with optimized HOAc mobile phases than those containing 0.1% TFA.
MassPREP Peptide Mixture (p/n 186002337, shown in Table 1) was reconstituted with either 0.1% TFA or 0.1% HOAc (depending on the mobile phase employed) to a total peptide concentration of either 0.6 or 2.0 mg/mL (depending on the sample load). A low purity (<70%) preparation of the synthetic peptide, DFVGYGVKDFVGVGVK, was reconstituted in 0.1% TFA/0.1% HOAc to a concentration of either 1 or 4 mg/mL.
|
LC conditions |
|
|---|---|
|
System: |
ACQUITY UPLC H-Class Bio System with a 20-cm Column Heater |
|
Detection: |
ACQUITY UPLC TUV Detector with 500-nL Analytical Flow Cell Xevo G2 Q-Tof Mass Spectrometer |
|
Wavelength: |
214 and 250 nm |
|
Scan rate: |
2 Hz (filter time constant, 1 s) |
|
Columns: |
XBridge BEH130 C18 4.6 x 100 mm, 5 μm, Porous, 130Å (p/n 186003579) XSelect CSH130 C18 4.6 x 100 mm, 5 μm, Porous, 130Å (p/n 186007077) |
|
Column temp.: |
40 °C |
|
Sample temp.: |
10 °C |
|
Injection volume: |
50 to 1000 μL, sample loads noted below |
|
Flow rate: |
1 mL/min (split post-UV detector to ca. 20 μL/min for infusion into the MS source) |
|
Mobile phases: |
See gradient tables |
|
Vials: |
LCGC Certified Clear Glass 12 x 32 mm Screw Neck Qsert Vial (p/n 186001126C) |
MassPREP Peptide Mixture
A: 0.1% (v/v) TFA in water
B: 0.1% (v/v) TFA in 90:10 acetonitrile (ACN)/water
or
A: 0.1% (v/v) HOAc in water
B: 0.1% (v/v) HOAc in 90:10 ACN/water
|
Time(min) |
%A |
%B |
|---|---|---|
|
0 |
99.5 |
0.5 |
|
1 |
99.5 |
0.5 |
|
61 |
40.0 |
60.0 |
|
62 |
10.0 |
90.0 |
|
65 |
10.0 |
90.0 |
|
66 |
99.5 |
0.5 |
|
85 |
99.5 |
0.5 |
Focused Gradients for DFVGYGVKDFVGVGVK
A: 0.1% (v/v) TFA in water
B: 0.1% (v/v) TFA in 90:10 ACN/water
|
Time(min) |
%A |
%B |
|---|---|---|
|
0.0 |
90 |
10 |
|
3.0 |
90 |
10 |
|
4.0 |
80 |
20 |
|
24.2 |
60 |
40 |
|
29.2 |
10 |
90 |
|
32.2 |
10 |
90 |
|
33.2 |
90 |
10 |
|
52.0 |
90 |
10 |
A: 0.1% (v/v) HOAc in water
B: 0.1% (v/v) HOAc in 90:10 ACN/water
or
A: 99:1 (v/v) water/HOAc – 1% HOAc
B: 90:9:1 (v/v) ACN/water/HOAc – 1% HOAc
|
Time(min) |
%A |
%B |
|---|---|---|
|
0.0 |
90 |
10 |
|
3.0 |
90 |
10 |
|
3.3 |
87 |
13 |
|
23.5 |
67 |
33 |
|
29.2 |
10 |
90 |
|
32.2 |
10 |
90 |
|
33.2 |
90 |
10 |
|
52.0 |
90 |
10 |
|
Mass spectrometer: |
Xevo G2 Q-Tof |
|
Ionization mode: |
ESI+ |
|
Analyzer mode: |
Resolution |
|
Capillary voltage: |
3.00 kV |
|
Cone voltage: |
25 V |
|
Source temp.: |
120 °C |
|
Desolvation temp.: |
350 °C |
|
Cone gas flow: |
0.0 L/h |
|
Desolvation gas flow: |
800 L/h |
|
Calibration: |
NaI, 1 μg/μL from 50 to 2000 m/z |
|
Acquisition: |
50 to 1990 m/z, 2 Hz scan rate |
MassLynx Software v4.1
The application of CSH130 C18 and BEH130 C18 to analytical peptide separations, such as peptide mapping, has been extensively discussed in previous work.8-9 Briefly, CSH130 C18, with its novel positive surface charge, has been found to provide improved peak shapes and loadability compared to other peptide reversed-phase column chemistries. Significant increases in peak capacity, up to 90%, have been observed for analytical applications, particularly when mobile phases with little to no ion pairing are used. The positive surface charge of CSH130 C18 also provides unique selectivity and less retentivity compared to BEH130 C18, which makes the two sorbents excellent companion column chemistries for peptide separations.
To investigate the performance of CSH130 C18 and BEH130 C18 in preparative separations, loading studies were performed with a number of different peptides and mobile phases that are typically used in manufacturing, namely those containing either TFA or HOAc. Analytical (4.6 mm I.D.) columns packed with 5 μm particles were employed for these method development experiments.
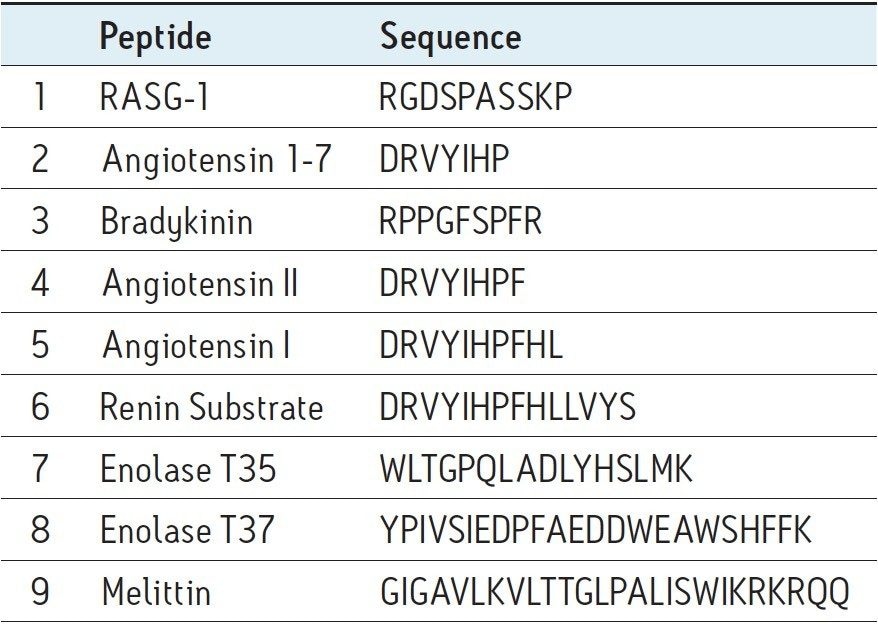
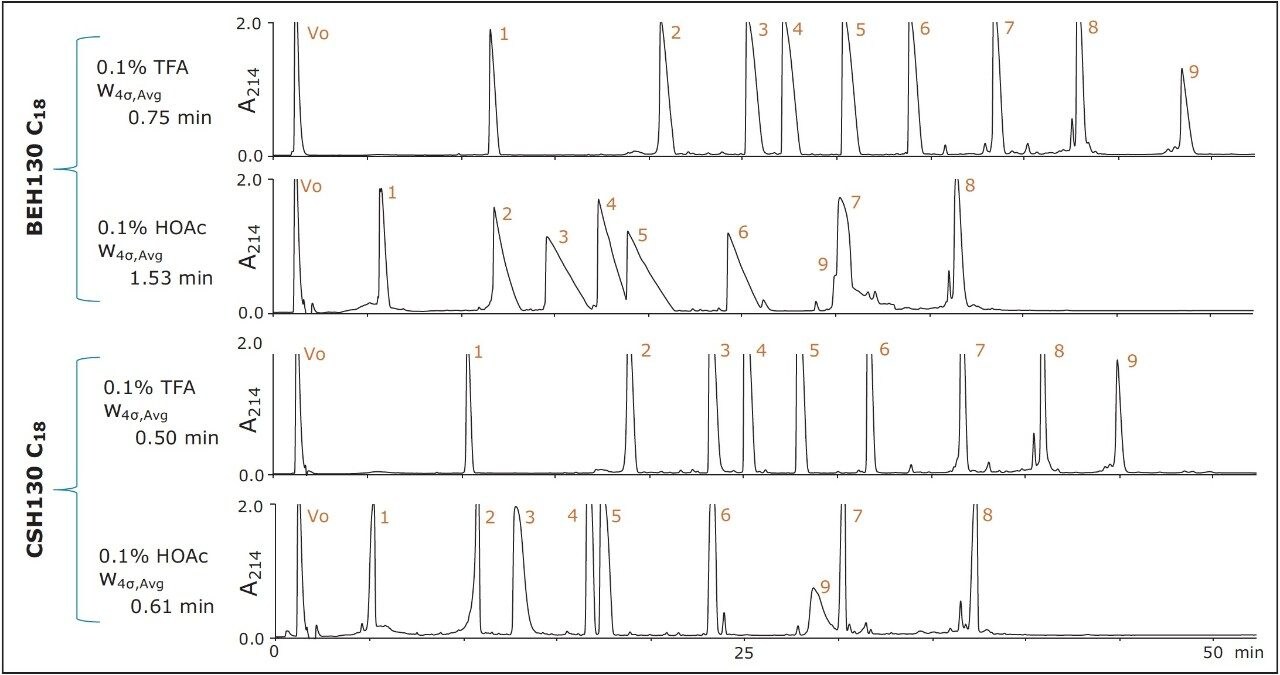
The MassPREP peptide mixture, containing nine different peptides shown in Table 1, was the first sample used to interrogate these columns. Figure 1 displays a set of chromatograms obtained for this mixture at semi-preparative loads using BEH130 C18 and CSH130 C18 as well as two different mobile phases, one containing 0.1% TFA and the other 0.1% HOAc. With 0.1% TFA, the average 4σ peak width for the BEH column was 0.8 minute. With 0.1% HOAc, this average peak width increased nearly two-fold to 1.5 minutes. HOAc is a much weaker acid than TFA, producing a less acidic mobile phase with significantly less ionic strength and ion pairing ability. Accordingly, peak shape for most C18 columns would be expected to be much worse when using HOAc instead of TFA. This assumption holds true for semi-preparative sample loads with BEH130 C18, as shown in Figure 1. However, peak shapes were remarkably well maintained with the CSH130 C18 column when TFA was exchanged for HOAc. The average 4σ peak widths observed using the CSH130 column with 0.1% TFA and 0.1% HOAc mobile phases were 0.5 and 0.6 minutes, respectively. These peak width data are summarized in Figure 2, where individual peak widths for the peptides in the mixture are plotted according to column type and mobile phase condition. In addition to the data for the semipreparative loads, data obtained for analytical sample loads are shown. This figure highlights that BEH130 C18 and CSH130 C18 produce similar peptide peak shapes under some conditions, including analytical sample loads with 0.1% TFA. However, under other conditions, such as semi-preparative loads with 0.1% HOAc, CSH130 C18 yields much narrower peaks. As has been demonstrated before,8-9 CSH130 C18 tends to deliver markedly better peak shapes for peptides in acidic mobile phases with little to no ion pairing. These data also demonstrate that this becomes even more evident for sample loads 20 times greater than those routinely used for analytical separations.
Preparative separations must often be performed at sample loads up to and sometimes more than 1000 times greater than those used for analytical separations. Sample loads below and within this regime were investigated using a synthetic, low purity peptide of the sequence DFVGYGVKDFVGVGVK, a neutral peptide (pI = 6) with a molecular weight of 1.7 kDa. Separations were performed on both the BEH and CSH columns using focused gradients to reduce run times, and low sensitivity wavelength (250 nm) detection to assess full peak shapes.
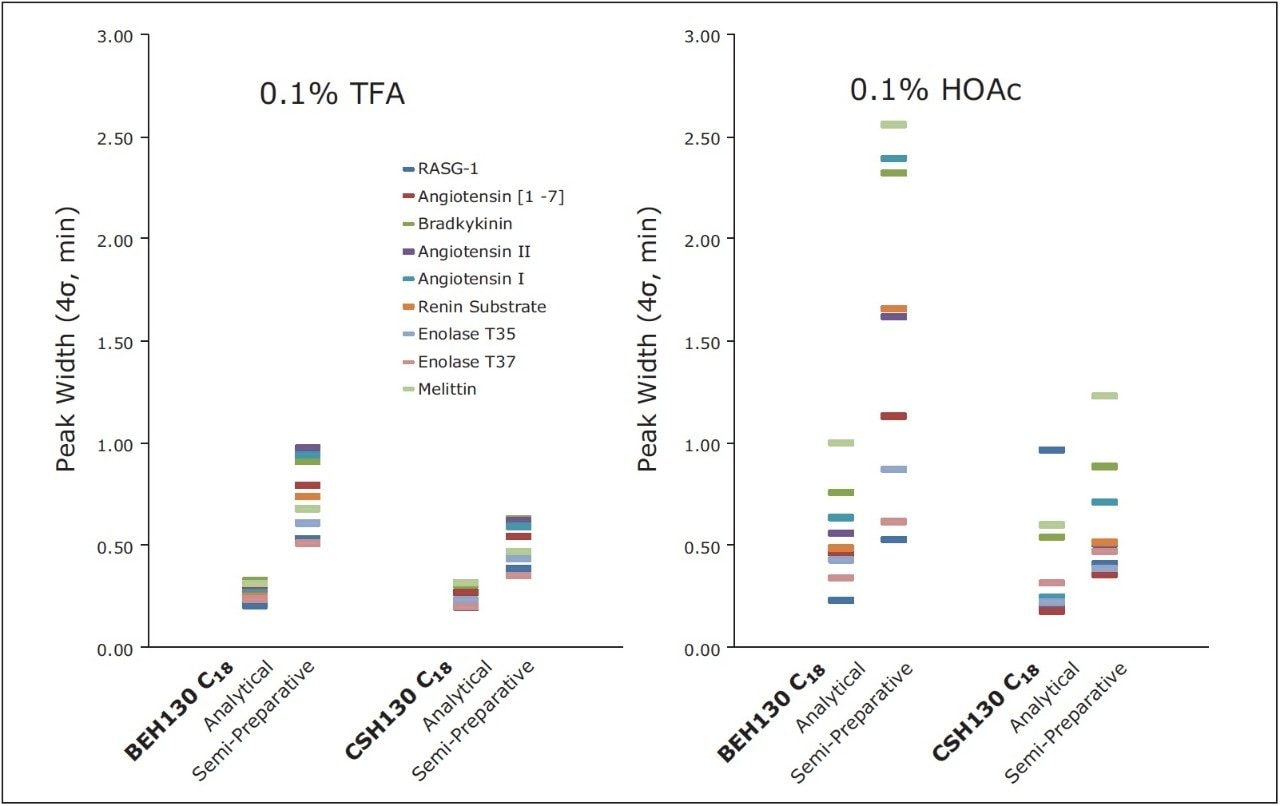
Semi-preparative and preparative sample loads with 0.1% HOAc modified mobile phases were analyzed first, as shown in Figure 3. A semi-preparative load (50 μg) on the BEH column produced the target peptide as a peak with the pronounced tailing that is consistent with commonly observed Langmuirian isotherms. Conversely, at a preparative load (1 mg), the target peptide eluted as a slightly fronting peak, typified by an anti-Langmuirian isotherm. Anti-Langmuirian isotherms are known to occur when peptides are present in zwitterionic form.10 Consequently, the 0.1% HOAc modified mobile phase was not acidic enough to fully protonate the carboxyl groups of this synthetic peptide. The peptide was likely present in both cationic and zwitterionic forms. The relative amount of the zwitterion would be expected to increase with sample load, particularly when the concentration of the target peptide exceeds the protonation/buffering capacity of the mobile phase. This explains the dramatic change in peak shape with increasing load on the BEH column.
Interestingly, 0.1% HOAc, as seen in Figure 3, appears to be ideal for obtaining a narrow target peptide peak with BEH130 C18 at the preparative sample load. Under these conditions, the BEH column yielded a narrower target peptide peak than the CSH column. In fact, CSH130 C18 yielded fronting peaks at both sample loads with 0.1% HOAc. This is not surprising given that its positive surface charge minimizes the extent of tailing that occurs for peptides.8-9 Thus, the fronting peak shape is more readily apparent. Because there is no peak tailing, the width of the target peak at the preparative sample load is actually greater on the CSH column than the BEH column.
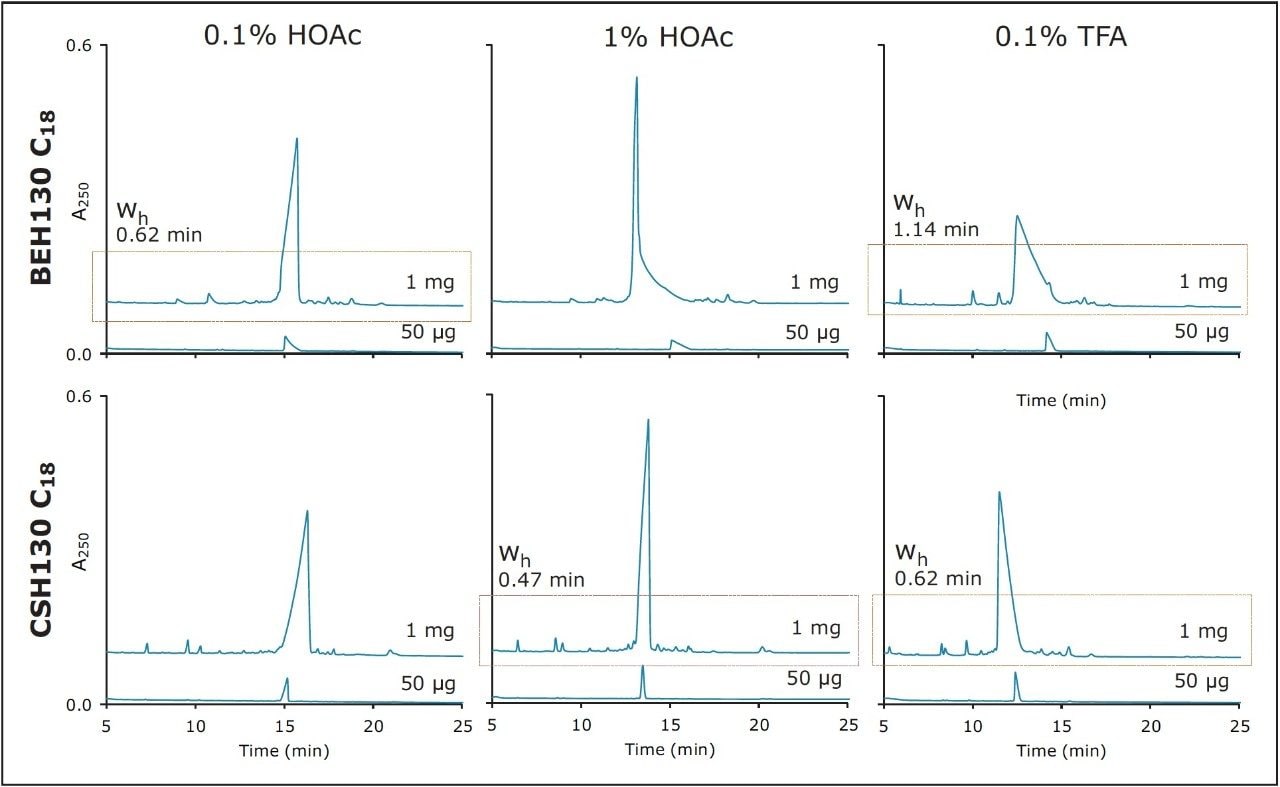
Based on this, CSH130 C18 and BEH130 C18 would be expected to produce optimal peak shapes under different mobile phase conditions. To this end, separations were also performed with a mobile phase modified with 10 times more acid (1% HOAc). Intermediate concentrations were not evaluated, although they may be of value for purification development. This change in the mobile phase composition significantly improved the peak shape for the CSH column, but actually worsened peak shape for the BEH column, as shown in Figure 3. With their optimal HOAc mobile phases, both the BEH (0.1% HOAc) and CSH (1% HOAc) columns produced narrow target peptide peaks (half height widths of 0.5 and 0.6 minutes, respectively). To benchmark these results, separations were also performed with 0.1% TFA as an ion-pairing agent, as seen in the right panel of Figure 3. With TFA, target peptide peak widths were 0.6 minute on CSH130 C18 and 1.1 minutes on BEH130 C18. Regardless of column chemistry, the optimized HOAc mobile phases yielded significantly narrower peptide target peaks than those containing TFA. These results demonstrate that acetic acid mobile phases have utility for peptide preparative chromatography.
Narrower target peptide peaks often coincide with greater resolution of impurities and, in turn, provide the opportunity to collect higher purity fractions. The effect of column chemistry and mobile phase additive on the preparative loads of DFVGYGVKDFVGVGVK is shown again in Figure 4, where the baselines of the chromatograms and the capability of each separation to resolve a number of MS-identified impurities from the target peak are the focus. As previously noted, narrower target peaks were observed with the HOAc mobile phases. Figure 4 also demonstrates that co-elution of the monitored impurities was likewise minimized through the use of the HOAc mobile phases. In addition, it is apparent that through the use of the different mobile phase additives and the two different column chemistries, chromatographic selectivity between the target peptide and the impurities was dramatically altered. For the parameters screened in this loading study, the CSH column with a 1% HOAc mobile phase appeared to provide both the narrowest target peptide peak and the least co-elution with monitored impurities. Nonetheless, a comparable separation could be achieved with the BEH column and a 0.1% HOAc mobile phase. The availability of column chemistries with different selectivity and optimal additive concentrations can be of benefit when developing challenging preparative separations.
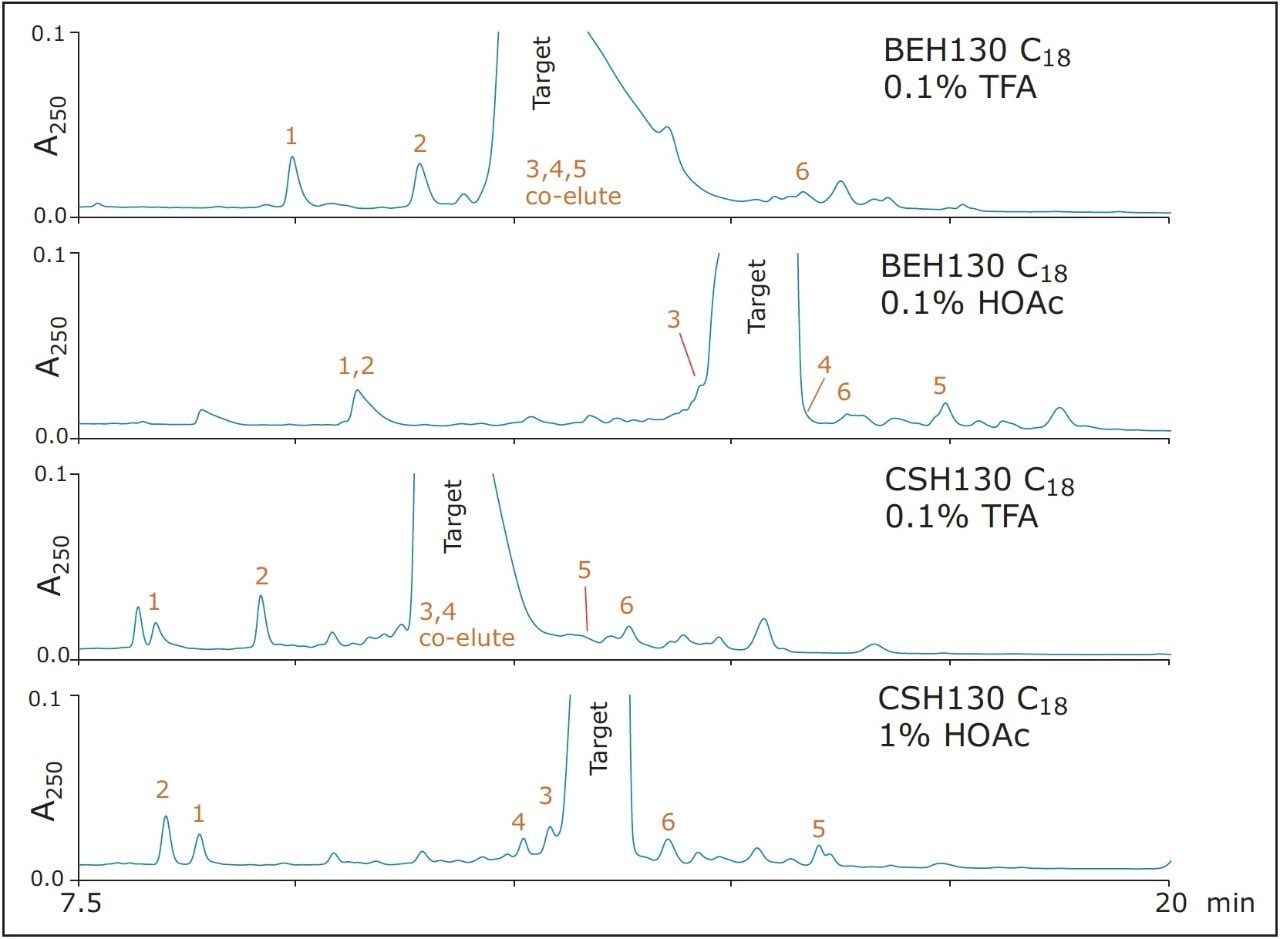
Thus far, results for only a moderate preparative sample load (1 mg) of the synthetic peptide, DFVGYGVKDFVGVGVK, has been discussed. Chromatograms acquired for 4 mg loads of this peptide are shown in Figure 5. This sample load would correspond to 0.5 g of material and, thus, very high productivity per injection on a larger 50-mm I.D. column. From these data, it is clear that both the CSH and BEH columns are amenable to high sample loads. It is notable, nevertheless, how strikingly consistent the peak profiles are for the CSH column from semi-preparative (50 μg) to preparative (4 mg) loads. Such predictability may prove useful when needing to develop an isolation method without consuming large amounts of sample.

Based on loading studies with analytical bore columns, the use of 5-μm BEH130 C18 and 5-μm CSH130 C18 shows significant promise for facilitating preparative peptide separations with either TFA or HOAc containing mobile phases. Both BEH130 C18 and CSH130 C18 column chemistries have useful attributes. Under acidic conditions, CSH130 C18 tends to exhibit improved loadability and generally narrower target peaks compared to BEH130 C18. Lower volume fractions can, therefore, be obtained with CSH130 C18, which could be exploited to aid subsequent purification and solvent removal steps. BEH130 C18, on the other hand, is perfectly suited to neutral/basic pH preparative chromatography, due to its longer term and higher temperature stability at such conditions. Finally, CSH130 C18 and BEH130 C18 also exhibit unique selectivity, making them useful companions for resolving challenging impurity/target peptide profiles.
Perhaps more interesting than these aforementioned attributes is the fact that each stationary phase optimizes with different concentrations of mobile phase additive and yields best peak shapes for preparative loads of an example synthetic peptide with optimized HOAc mobile phases, rather than those containing 0.1% TFA. This suggests an opportunity to leverage these hybrid particle C18 columns to streamline purification processes, since using HOAc mobile phases would mean that peptides with a pharmaceutically acceptable counter ion could be obtained in fewer steps.
720004674, April 2013