
This application note demonstrates to provide an easy-to-use methodology for trace level monitoring of contaminants in manufactured ‘clean’ parts and assemblies.
The cleanliness of parts and assemblies is critical to many modern high technology industries, and impacts not only on yield, but also the viability of the products. As surfaces define many of the properties of materials, even trace surface contaminants may dramatically alter the properties and performance characteristics of a material. Low level contamination can therefore be extremely detrimental to the processing or application of the components downstream.
It is vital for these industries to quickly recognize that a component has become contaminated, identify the contaminant, and trace the contamination to the specific part of the process affected. Failure to quickly identify and respond to contamination may lead to product hold, major recall, or loss of revenue and reputation.
Typically the primary source of contamination is from the industrial manufacturing process. Contaminants may originate from moulding, machining, stamping, polishing, etc. which require the use of coolants, lubricants, dispersants, chemical etching, and cleaning agents; or as by-products from chemical reactions used to modify the properties of the material. Once manufactured, the parts undergo rigorous cleaning before entering assembly. Secondary contamination may be from the environment, packaging, or through handling. A final stage of cleaning is therefore required to remove loose particles, organic and ionic contaminants which might be introduced during storage, handling or shipping.
To optimize the cleaning process and troubleshoot problems in the manufacturing process, a rapid and easy sampling method and measurement is required. Here, the Waters ACQUITY UPLC system has been modified to provide direct injection without any separation, which greatly enhances the sensitivity of the method for the detection of low-level contaminants. For these analyses, adopting a chemometric approach can quickly provide information about the similarities and differences within large mass spectral datasets, and may be used to highlight sudden changes, such as the appearance of a new contaminant. Automation of this process greatly reduces the analysis time required and the probability of error in this assessment, while increasing sensitivity. A database of possible contaminants can also be built, so that identified contaminants can be traced to a particular source or process.
Such a workflow may be applicable to many fields where surface contamination and impurities may be an issue, for example in the electronics and packaging industries.

|
LC system: |
ACQUITY UPLC (modified with capillary assembly to provide back pressure) No column |
|
Mobile phase A: |
Water + 0.1% formic acid |
|
Mobile phase B: |
Acetonitrile + 0.1% formic acid |
|
Flow rate: |
0.03 mL/min, isocratic, 50:50 mobile phase A:B |
|
Total run time: |
3.0 min |
|
Injection volume: |
20 μL, full loop, overfill factor 2.0 times |
|
MS system: |
Xevo G2 QTof MS |
|
Ionization: |
ESI positive and negative |
|
Scan mass range: |
m/z 50 to 1200 |
|
Capillary voltage: |
3 kV |
|
Sample cone: |
35 V |
|
Source temp.: |
120 oC |
|
Desolvation gas: |
Nitrogen, 1000 L/hr, 500 °C |
|
Cone gas: |
Nitrogen, 25 L/hr |
|
Lock mass compound: |
Leucine enkephalin (m/z 556.2771 and 278.1141) |
The samples were analyzed using an ACQUITY UPLC System with Xevo G2 QTof MS, and data were acquired using MassLynx Software, v. 4.1. MarkerLynx XS Application Manager was used to process the data.
A dedicated ACQUITY UPLC System and fluidics were used for trace contaminant analysis to ensure that there was no contamination arising from the system or previously run samples. All glassware must be ultraclean, and all solvents and reagents used must be of the highest quality and purity – minimum LC-MS grade solvents were used. In addition, all glassware used were rinsed five times with a soak solution immediately prior to use, and the entire UPLC-MS system was flushed for an hour before use.
For this analysis, sample parts were treated with a detergent containing polyethylene glycol (PEG), a ubiquitous polymer, then underwent complete and incomplete passivation and rinsing.
Samples were prepared in triplicate by soaking individual parts in appropriately sized glass sampling jars (minimizing the volume of solvent used, while completely submerging the component part) for eight hours. The soak solution used was a 50:50 mixture of acetonitrile and water with 0.1% (v/v) formic acid. Sampling jars without parts were prepared in the same manner and analyzed in parallel with the parts as reference blanks. One mL aliquots of the soak solutions were transferred to Waters LC-MS Certified Vial (P/N: 600000668CV), and analyzed by direct-flow injection.
The comprehensive detection and chemical component information obtained using UPLC/ToF-MS can be effective for supporting quality control and the discovery and identification of contaminants. This method allows the cleanliness of otherwise challenging irregular shaped parts and assemblies, and their internal surfaces to be measured. The chemical composition information obtained is processed and visualized by multivariate analysis in order to simplify the interpretation of the results. Any contaminating organic and ionic species identified can then be elucidated using fragment ion information. The workflow for this process is shown in Figure 2.

In monitoring product quality, the material from different production lots, including non-conforming material are measured. Samples are measured in triplicate and accurate masses are automatically extracted and stored. MarkerLynx XS performs Principal Component Analysis (PCA) on the extracted data and separates out the non-conforming material based on differences in their scores.
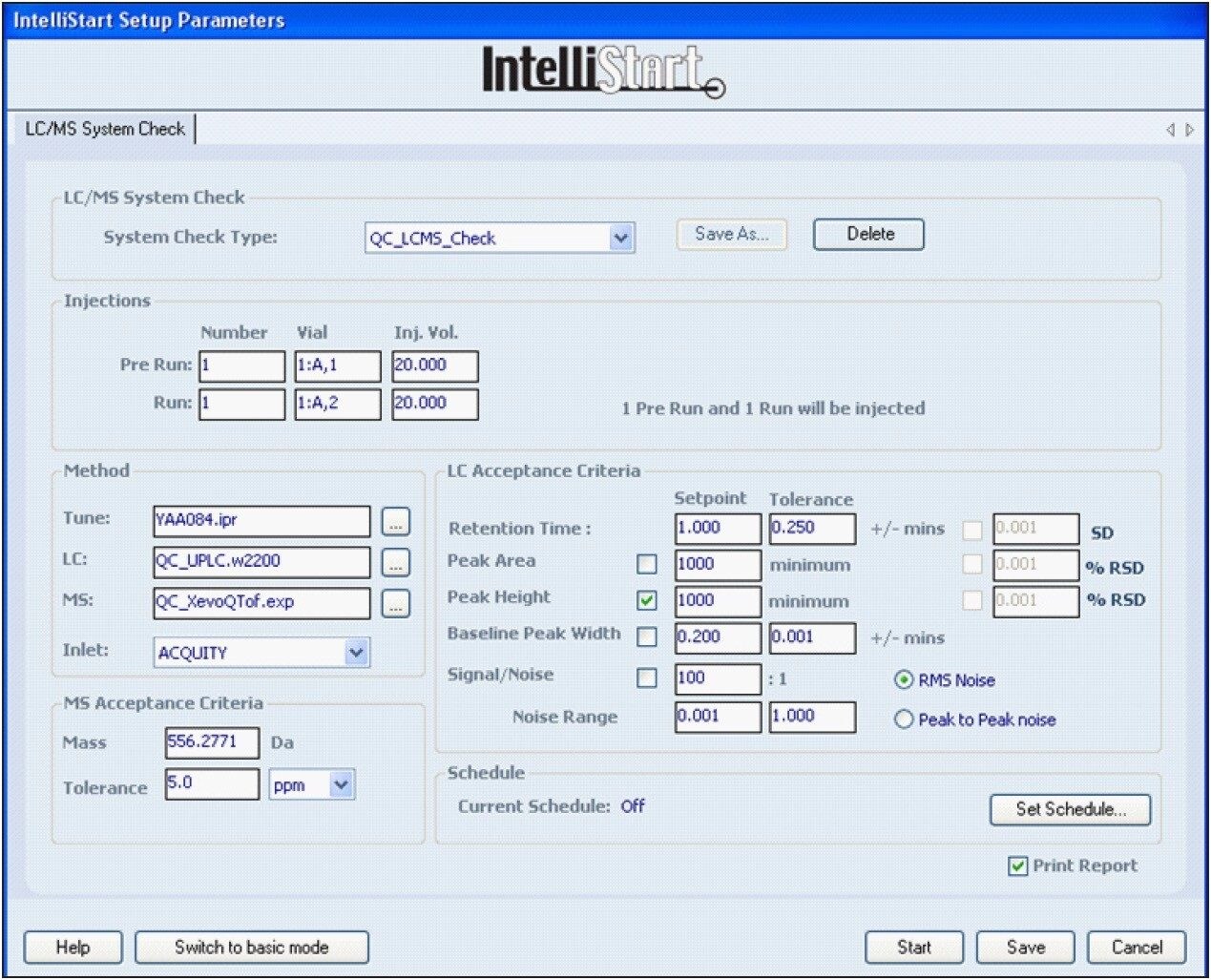
Incorporated into MassLynx Software is Waters IntelliStart Technology, which automates calibration and LockSpray setup. IntelliStart also provides a simple and automated system check procedure to ensure that the LC-MS system is operating optimally. An LC-MS system check, shown in Figure 3, is performed prior to analysis using a series of user-defined criteria, such as mass accuracy, retention time, peak height, etc. for a particular concentration, and injection volume of a reference compound. These can be used for the long-term monitoring of the system’s performance.
A novel feature of Xevo G2 QTof MS is its ability to run methods with multi-point lock mass. For the trace analysis, a two-point lock mass (m/z 278.1141, 556.2771) was utilized to correct for environmental changes in the laboratory over time, which would otherwise result in loss of mass accuracy. Good mass accuracy allows the elemental composition of both the precursor and its fragment ions in MS/MS to be determined with greater confidence and precision, aiding the elucidation of unknown compounds.
Trace contaminant analysis typically relies on manual processing and analysis of data which is time consuming and tedious. The MarkerLynx and Extended Statistics software enable users to overcome the time-consuming problem of identifying patterns in the in LC-MS data sets by showing the similarities and differences between detected MS traces.
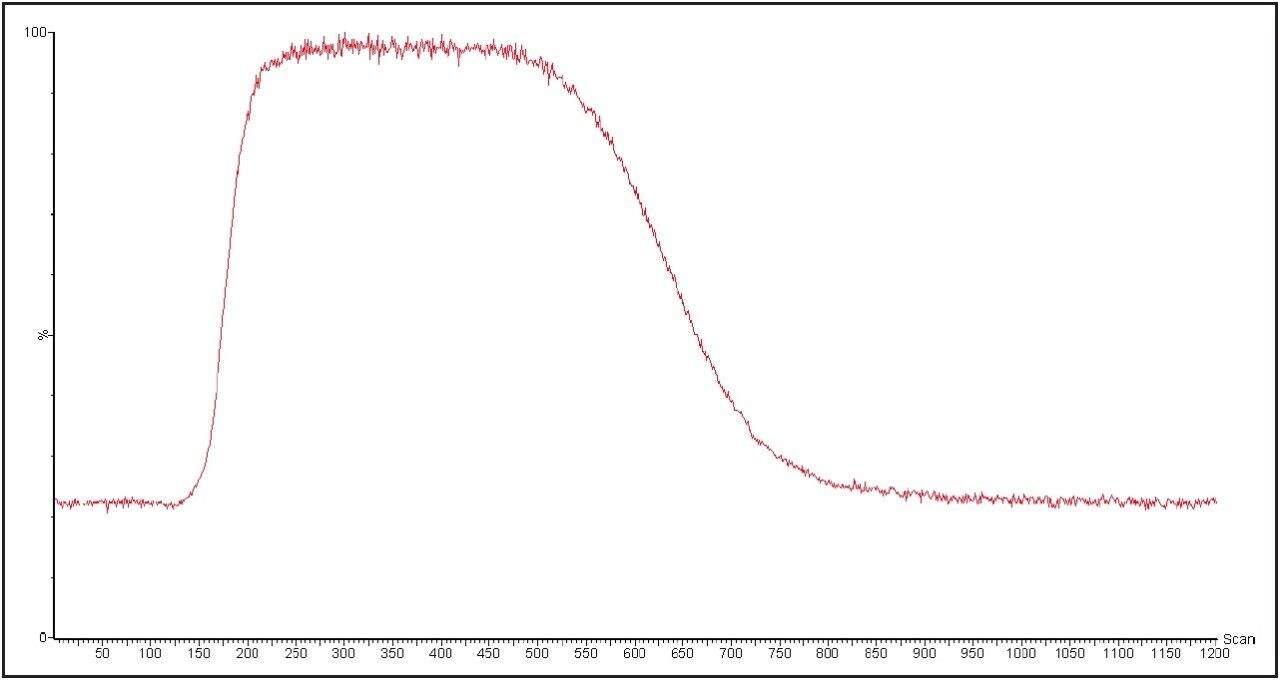
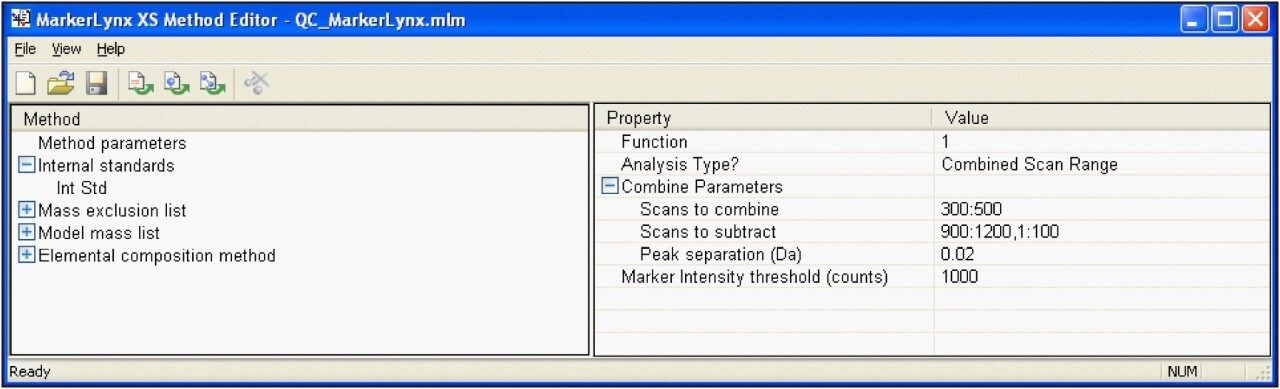
Use of MarkerLynx XS Application Manager eliminates the need to manually process (strip and background subtract) each and every spectrum individually. A 20 μL isocratic LC injection is used to enhance the sensitivity of the method to low-level contaminants, as shown in Figure 4. MarkerLynx XS automatically combines spectra over the selected scan range, subtracting background components present in the mobile phase, as shown in Figure 5.
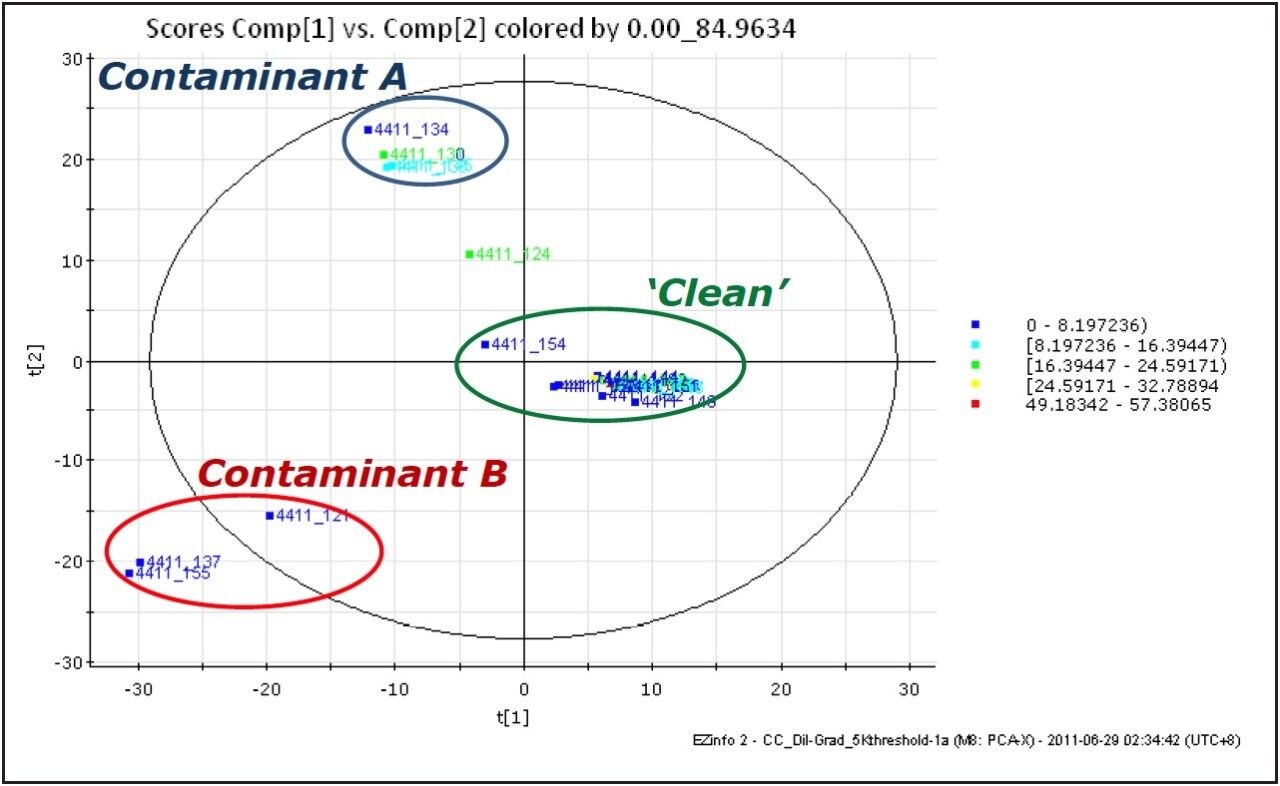
The processed and extracted data are then interrogated using the Extended Statistics chemometrics-based software tool included in the MarkerLynx XS Application Manager. Principal Component Analysis is performed, and the data set is displayed on a scores plot which shows the possible presence of atypical observations, such as contaminated parts and patterns in the data, as shown in Figure 6.
An Orthogonal Partial Least Squares (OPLS) model can then be applied to compare and separate the two groups by their components, enabling extraction of those components responsible for the differences between them. Using the conforming (Clean) group and non-conforming (Contaminant B) group, an S-Plot is obtained, as shown in Figure 7.
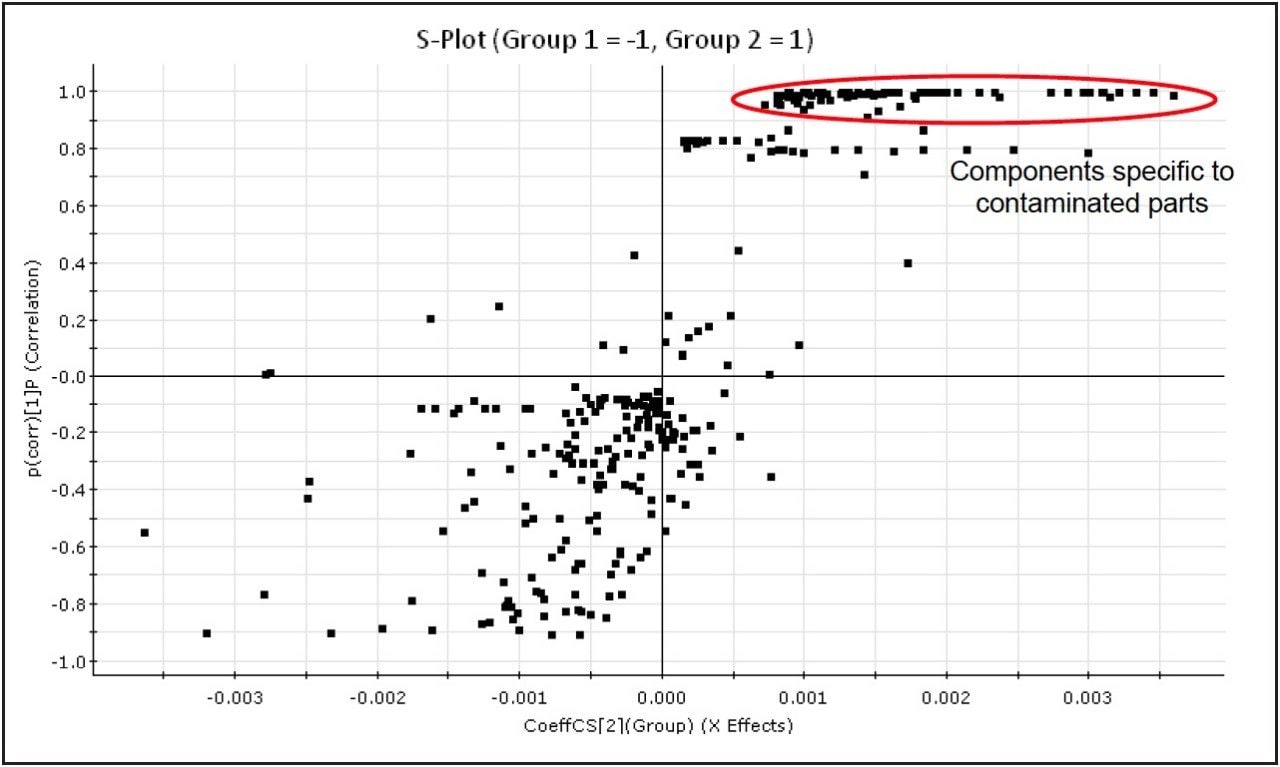
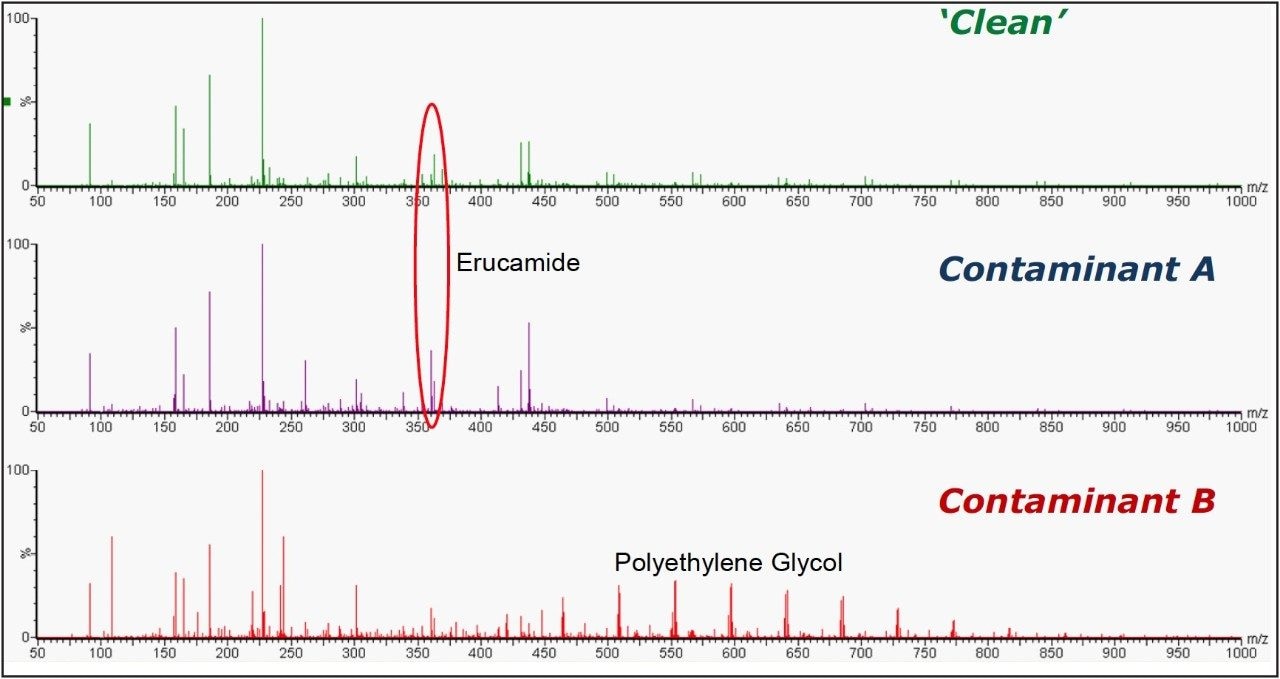
Once the non-conforming components responsible for differences with the clean samples are determined, candidate contaminants can be determined by searching against internal or external databases of known contaminants. In this case, the components specific to Contaminant B were shown to correspond to PEG, in the combined spectrum, shown in Figure 8. The peaks present in both the ‘clean’ and Contaminant A samples were mainly due to sodium formate, formed by formic acid in the soak solution reacting with sodium from the glassware. However, MarkerLynx XS found that the component which differentiated Contaminant A from the ‘Clean’ samples was m/z 360.32, corresponding to erucamide [M+Na]+, a common slip agent used in plastics manufacturing.
The high mass accuracy and accurate isotope patterns provided by Time-of-Flight mass spectrometry (ToF MS) allow elemental compositions to be generated with high confidence for unknown compounds. MS/MS can also be used to provide structural information in order to help identify and confirm contaminants. After contaminated components and contaminants have been identified, they can be used to trace the source (manufacturing or processing step) where the contaminant(s) may have been introduced, and the issue can be addressed. MarkerLynx XS is sufficiently selective to separate identify differences in manufacturing and processing contaminants on the different cleaning validation piece parts.
720004030, July 2011