This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the improved long-term stability of the ACQUITY UPLC BEH200 SEC, 1.7 μm column with the use of a guard column.
The combination of the BEH200 SEC analytical column and guard column allow for a stable analysis and quantitation of a monoclonal antibody in the presence of excipients.
Size-exclusion chromatography (SEC) is commonly used to measure aggregates and other size variants in protein-based therapeutics. Of particular interest are soluble aggregates, which are thought to play a role in immunogenicity. Studies have found a variety of chemical and physical conditions can affect the amount of aggregates in biotherapeutics. To minimize these effects, excipients or stabilizers are typically added to protein-based therapeutics. Additives can decrease long-term column stability in SEC, resulting in inaccurate quantitation due to changes in retention, peak shape or spacing between peaks. While a variety of cleaning protocols may be used to try to restore the column, this approach can be time consuming and ultimately ineffective. As a last resort, column replacement is often required, resulting in higher costs and further delays in analysis time. With the introduction of the ACQUITY UPLC BEH200 SEC, 1.7 μm guard column, long-term stability for the SEC column can be increased, thereby saving time and money.
The UPLC SEC separation for biomolecules combines the ACQUITY UPLC H-Class Bio System with a sub-2 μm BEH SEC column. While the ACQUITY UPLC BEH200 SEC packing material is resistant to chemical and mechanical degradation for hundreds of injections, various sample components may adversely affect the column. With the use of a guard column in combination with the BEH200 SEC column, the column stability can be extended for hundreds of injections in the presence of these excipients.
In this set of experiments, protein standards, uracil and a murine monoclonal antibody (mAb) were analyzed on both a BEH200 SEC, 1.7 μm, 4.6 x 150 mm column alone and a BEH200 SEC, 1.7 μm, 4.6 x 150 mm column equipped with a guard column with the same packing material (4.6 x 30 mm). The mAb was diluted in a representative biotherapuetic formulation containing polysorbate 80, sucrose and sodium phosphate. The samples were analyzed on a single BEH200 SEC column over the course of 500 injections and on a BEH200 SEC column equipped with a guard column for over 900 injections. The guard column was replaced approximately every 200 injections. Evaluation of the mAb monomer efficiency on the BEH200 SEC column alone shows a decrease in plate count (from 5000 to 3000) after 550 injections. The same sample tested on the column and the guard combination shows no significant change in mAb monomer efficiency over 900+ injections (Figure 1). Any drops in monomer efficiency were restored after replacement of the guard column.
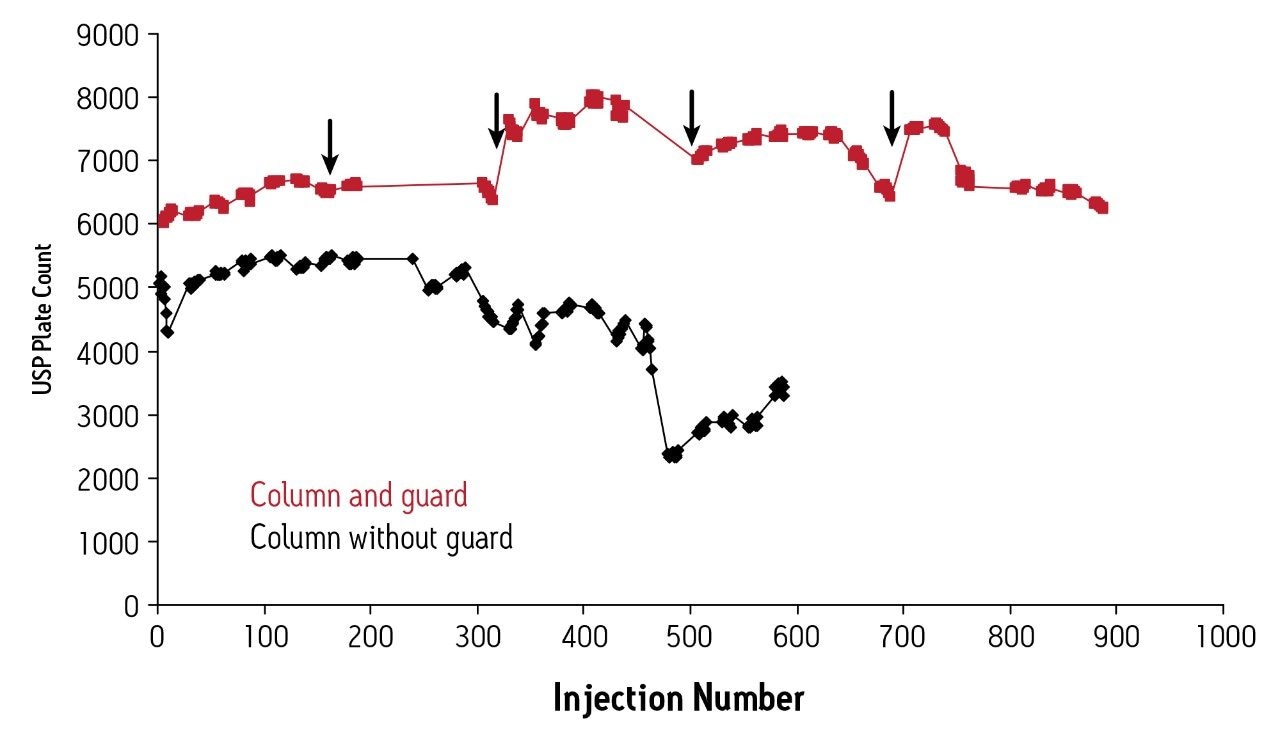
Before replacement of each guard column, the BEH200 SEC, 1.7 μm column was tested alone to check column efficiency. Comparison of this test to the initial column testing demonstrates the long-term stability achievable with a guard column (Figure 2). The mAb separation on the column alone shows a decrease in peak height and an increase in peak tailing over the course of 480 injections. Furthermore, the mAb monomer/dimer USP resolution decreases 26%. In contrast, the separation on the column protected by a guard shows no significant change in peak height or peak tailing over the course of 900 injections. The mAb monomer/dimer USP resolution remains relatively unchanged, demonstrating the effectiveness of the guard column.
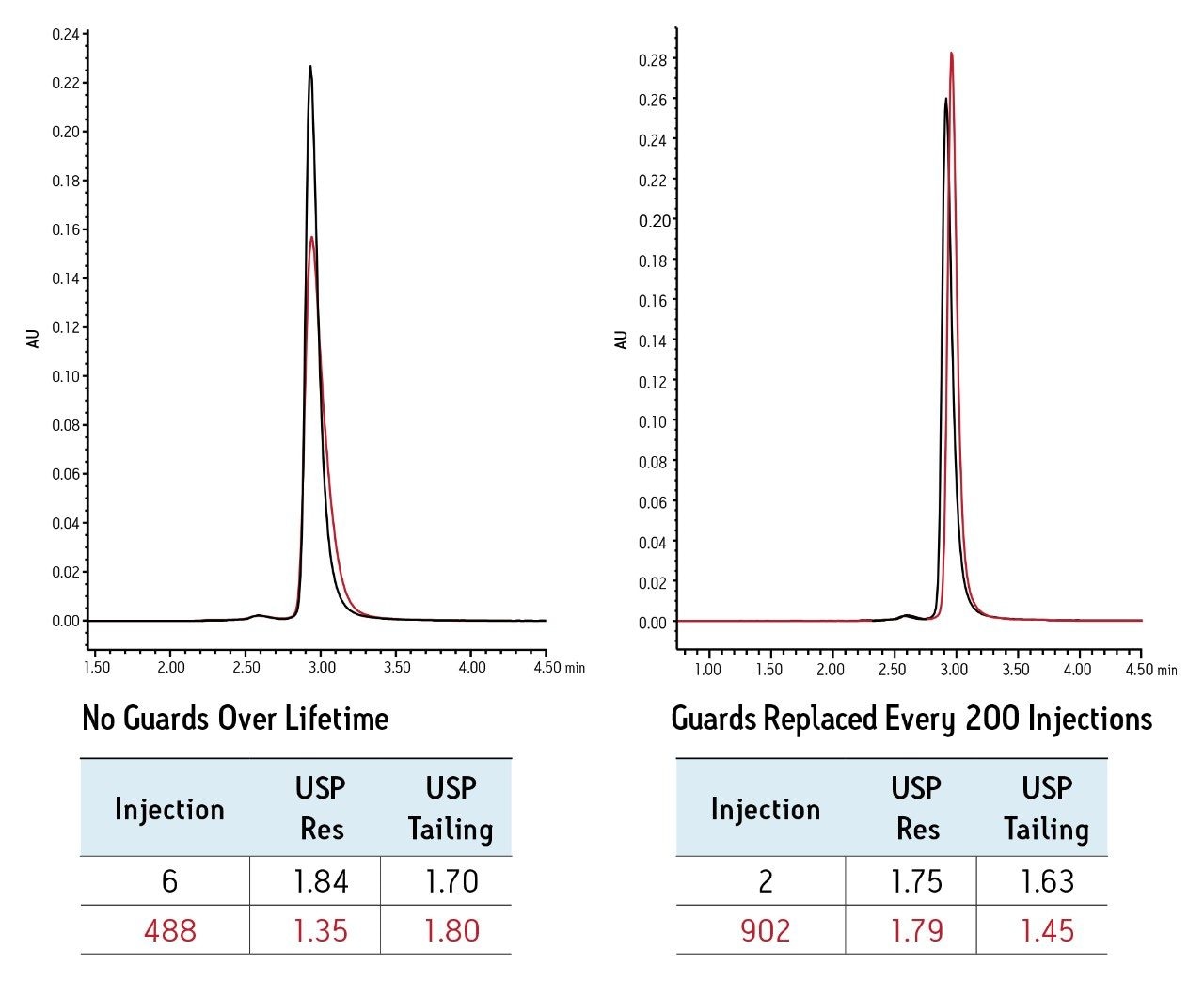
The combination of the BEH200 SEC analytical column and guard column allow for a stable analysis and quantitation of a monoclonal antibody in the presence of excipients. While the presence of excipients or other matrix components may adversely affect a SEC separation over time, the regular replacement of the guard column preserves the performance of the analytical column for over 900 injections. This allows the biochemist to reduce costs and minimize down time, thereby resulting in more stable analyses for monoclonal antibodies and their soluble aggregates.
720004034, July 2011