UPLC Glycan Separation Technology columns, when used in conjunction with the ACQUITY UPLC System and its Fluorescence (FLR) Detector, provide a highly-resolving, reproducible, and rapid method for profiling glycans.
Glycosylation is a post-translational modification of proteins that occurs in all eukaryotic cells. The sugar chains on glycoproteins can mediate biological activity, play a role in receptor-mediated recognition, increase solubility, regulate half-life and exert a stabilizing influence upon conformation. Specific glycan structures are, therefore, associated with safety and efficacy attributes of many protein drugs. Correct glycosylation is essential if a glycoprotein is to achieve and maintain the structure with full biological activity. The measurement of glycans, therefore, is important in biopharmaceutical development projects.The relative amounts of the individual glycan structures are monitored during process development to establish the stability of the growth and purification steps of manufacturing. The same measurements are required in the development of formulations and stability testing.
Waters provides a glycan analysis solution to meet the needs of these applications. The new ACQUITY UPLC BEH Glycan Separation Technology column separates the released glycans of biopharmaceuticals as their 2-aminobenzamide (2-AB) derivatives. This column is used with a Waters ACQUITY UPLC instrument with fluorescence detection (FLR) to ensure resolution, sensitivity and speed.

N-linked glycan mixtures were purchased as 2-AB labeled derivatives (ProZyme, San Leandro, CA) or released from glycoproteins with PNGase F. The released glycans were labeled according to the procedure of Bigge et al.1 Prior to analysis the samples were diluted to produce a 1 pmol/μL solution in 50% Buffer A/50% acetonitrile. Glycans are minimally soluble in higher acetonitrile concentrations which may lead to sample loss over time
|
LC System: |
Waters ACQUITY UPLC System |
|
Column: |
ACQUITY UPLC BEH Glycan, 2.1 x 150 mm, 1.7 μm |
|
Column Part Number: |
186004742 |
|
Column Temp: |
60 ˚C |
|
Sample Temp: |
15 ˚C |
|
Flow Rate: |
500 μL/min except during aqueous wash |
|
Mobile Phase A: |
100 mM ammonium formate, pH 4.5 |
|
Mobile Phase B: |
Acetonitrile |
|
Weak Needle Wash: |
90/10 acetonitrile/water (v/v) |
|
Strong Needle Wash: |
10/90 acetonitrile/water (v/v) |
|
Seal Wash: |
50/50 methanol/water (v/v) |
|
Injection volume: |
1.5 μL, Partial Loop; 2 μL, Full Loop |
|
Detection: |
Fluorescence (FLR) λex 330 nm λem 420 nm |
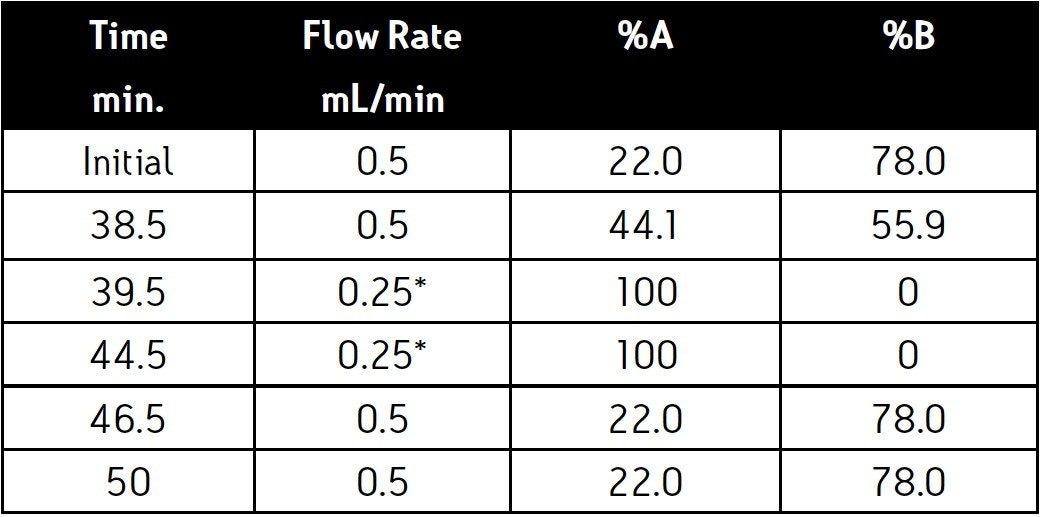
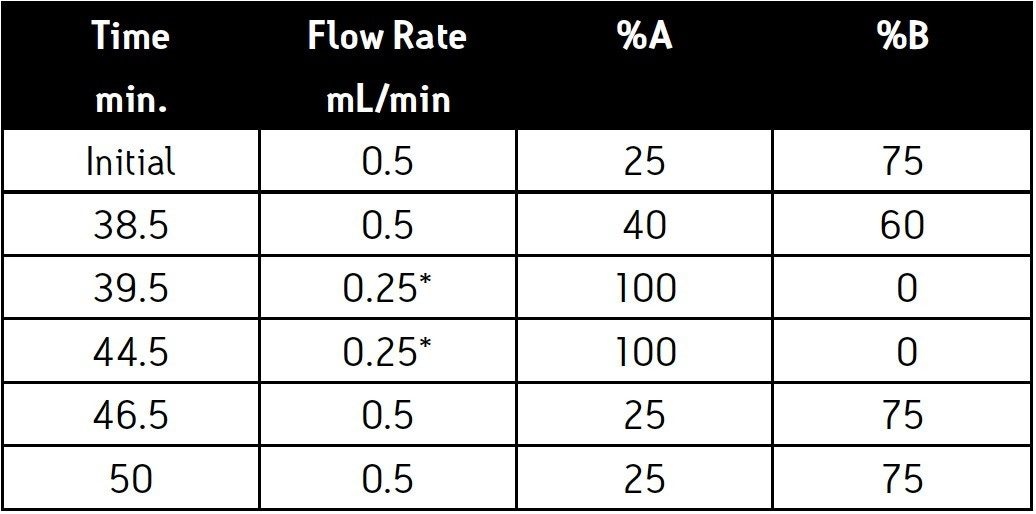
Alternative needles and mixers are available for peptide mapping. (P/N 205000507, Peptide Needle Kit; P/N 205000403, ACQUITY UPLC High Sensitivity Filter Mixer) The installation of the peptide mapping needle or mixer will not adversely effect glycan analysis. Chromatograms 1 and 2 were collected with an ACQUITY UPLC instrument fitted with the peptide needle and mixer. Chromatograms 3 and 4 were collected with an unmodified ACQUITY UPLC instrument.
Human IgG Glycans and dextran ladder
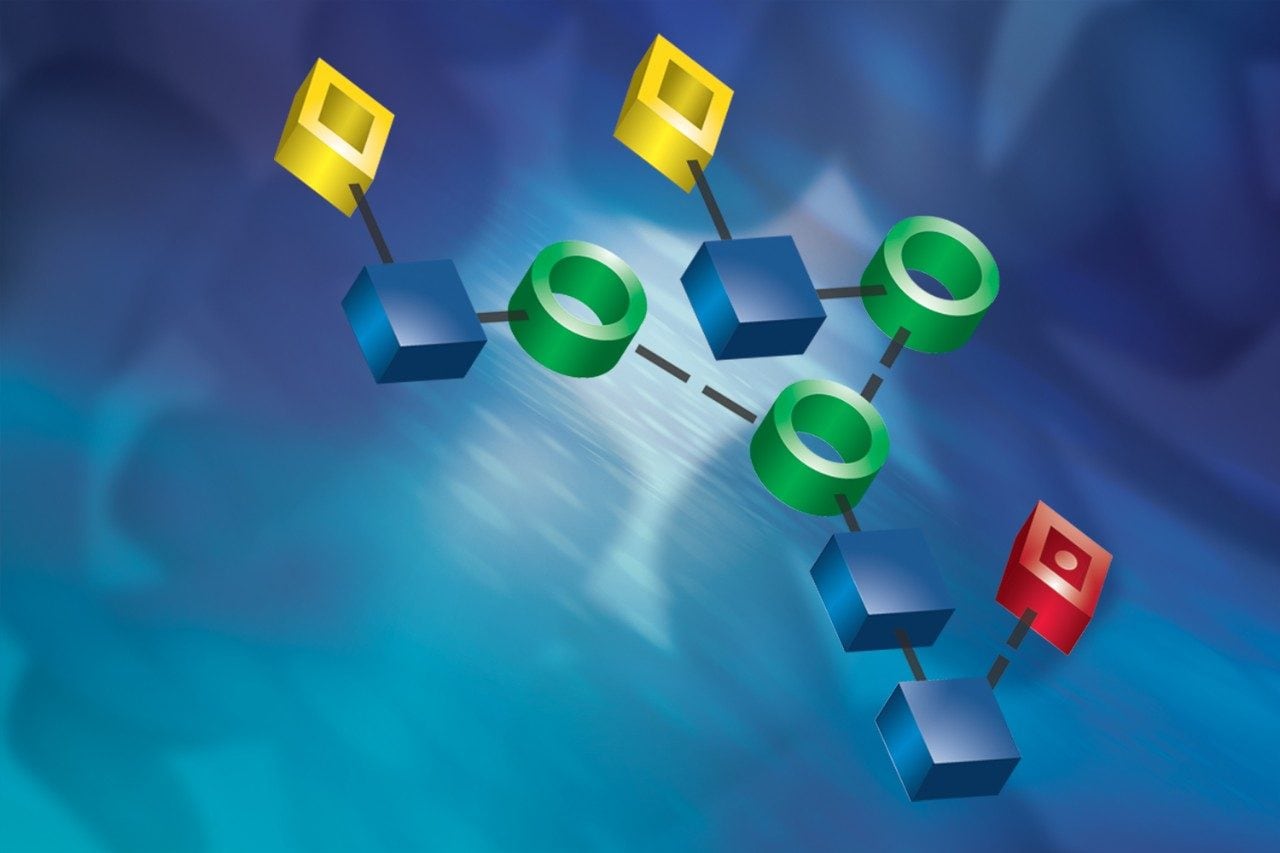
The design and development of the UPLC Glycan Separation Technology Column was targeted to the analysis of the glycans that are commonly found or expected on biopharmaceutical proteins. The array of oligosaccharides should, therefore, include high mannose, complex, hybrid, and sialylated glycans as shown in Figure 1. To establish a common detection for all the species that might be observed in a single sample, the glycans were labeled with 2-aminobenzamide. These fluorescent derivatives separate best on an amide-bonded phase. Since the labeled oligosaccharides are very hydrophilic, the preferred mode of separation is Hydrophilic Interaction Chromatography (HILIC). Since the set of glycans includes many closely related structures, it is necessary to use the selected chemistry under conditions that provide the highest possible resolution. The column was, therefore, developed for use on Ultra Performance Liquid Chromatography instruments with sub 2-micron particles. The BEH Technology hybrid material is used as the base for the 1.7 μm particles for maximum physical and chemical stability. The column is optimized for use with the ACQUITY UPLC System with FLR detection to take advantage of the minimum band-spreading. When running the system at the flow rate of 0.5 mL/min, higher than HPLC, a side effect of the small particles is higher back pressure that is accommodated by the ACQUITY UPLC System.
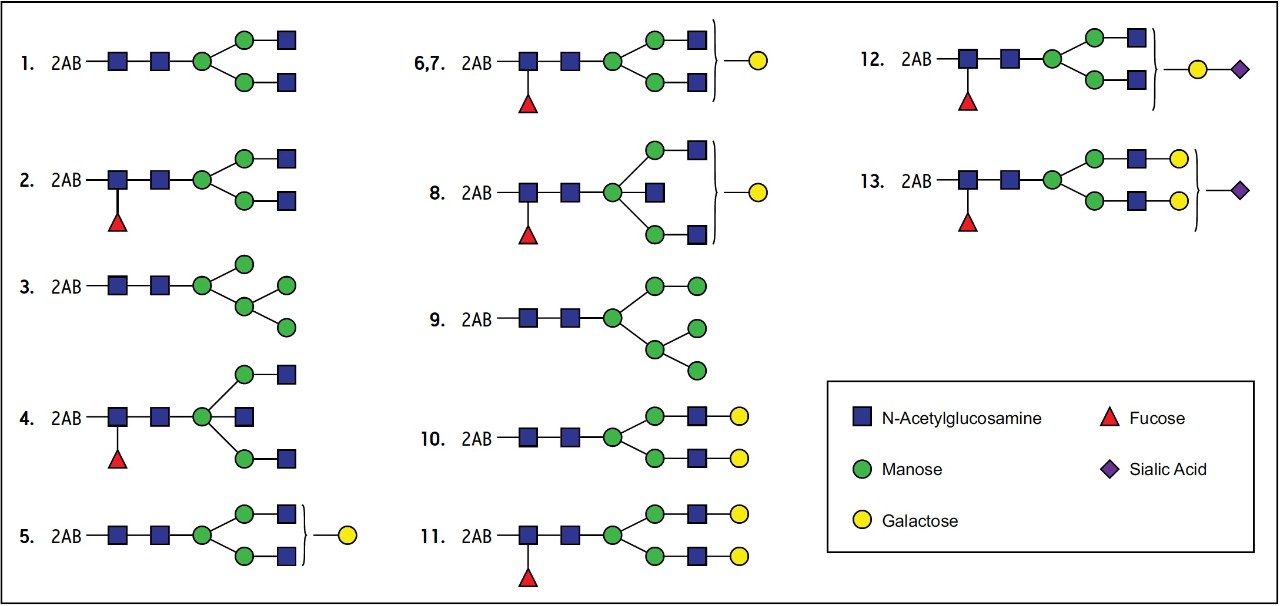
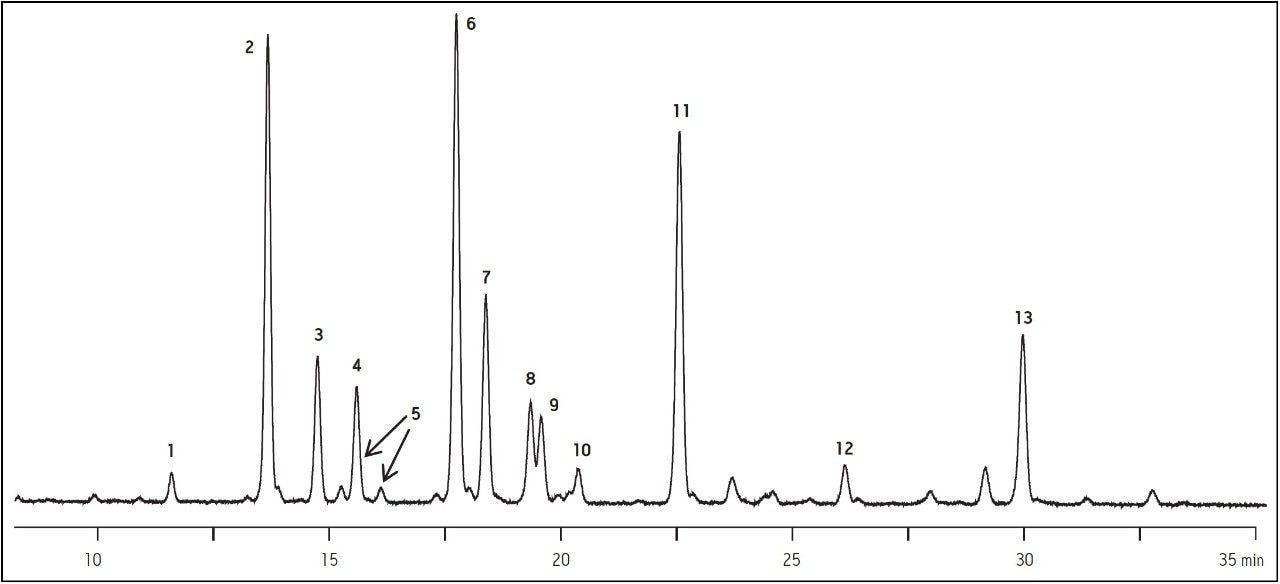
The column was evaluated during the development process with a sample of 2-AB labeled human IgG glycans, and a typical chromatogram is shown in Figure 2. This sample includes high mannose, complex, hybrid, and sialylated glycans as an index of the chemical suitability of the bonded phase. The final selected column composition, as shown in Figure 2, is particularly useful because of the excellent resolution of peak 2 (G0F) and peak 3 (Man5). Numerous isomers are present that are substituted on one of the two or three branches of the glycan. For example, the isomers shown as Peaks 6 and 7 are consistently separated. For quantitative analysis of the mixture, the areas of each of the peaks are compared. Peaks that are well resolved typically produce reproducible and reliable relative quantitation.
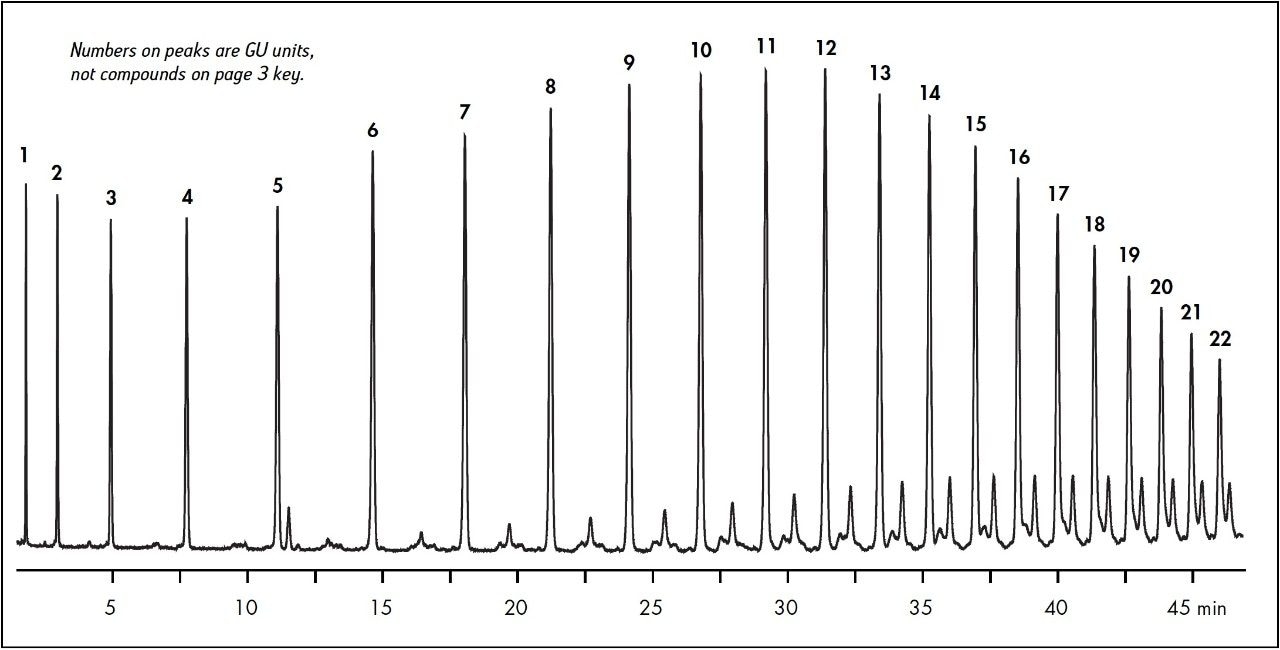
The column has also been evaluated for the separation of other significant glycans. The homo-polymeric series of glucose oligomers shown in Figure 3 extends from 1 to 22 sugars (Glucose Units, GU) with retention for the smallest species and good resolution for the largest. Measuring retention times relative to the glucose units provides standardization to estimate the effect of individual monosaccharide additions and deletions. It also provides a meaningful reference over column lifetime, and to compensate for any or batch-to-batch reproducibility. GU values can also be useful when characterizing unknown glycan structures.2
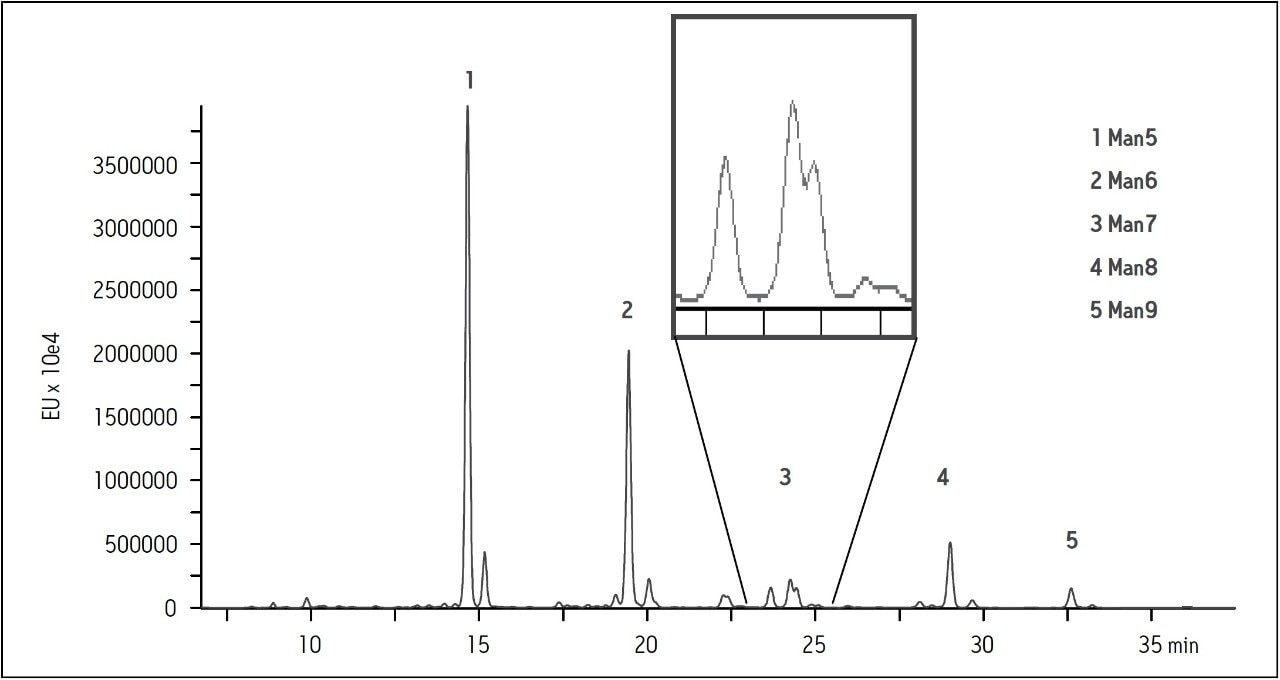
The resolving power of this column was tested with several samples that include neutral and acidic oligosaccharides, branched variants, and linkage isomers. The mixture of high mannose glycans that is present on bovine ribonuclease b contains many such isomers. In particular the Man7 glycan is known to contain three isomers that are substituted on one of the two or three branches of the glycan. A complex of several peaks eluting just before 25 minutes in Figure 4 shows the separation of isomers of the Man7 glycan
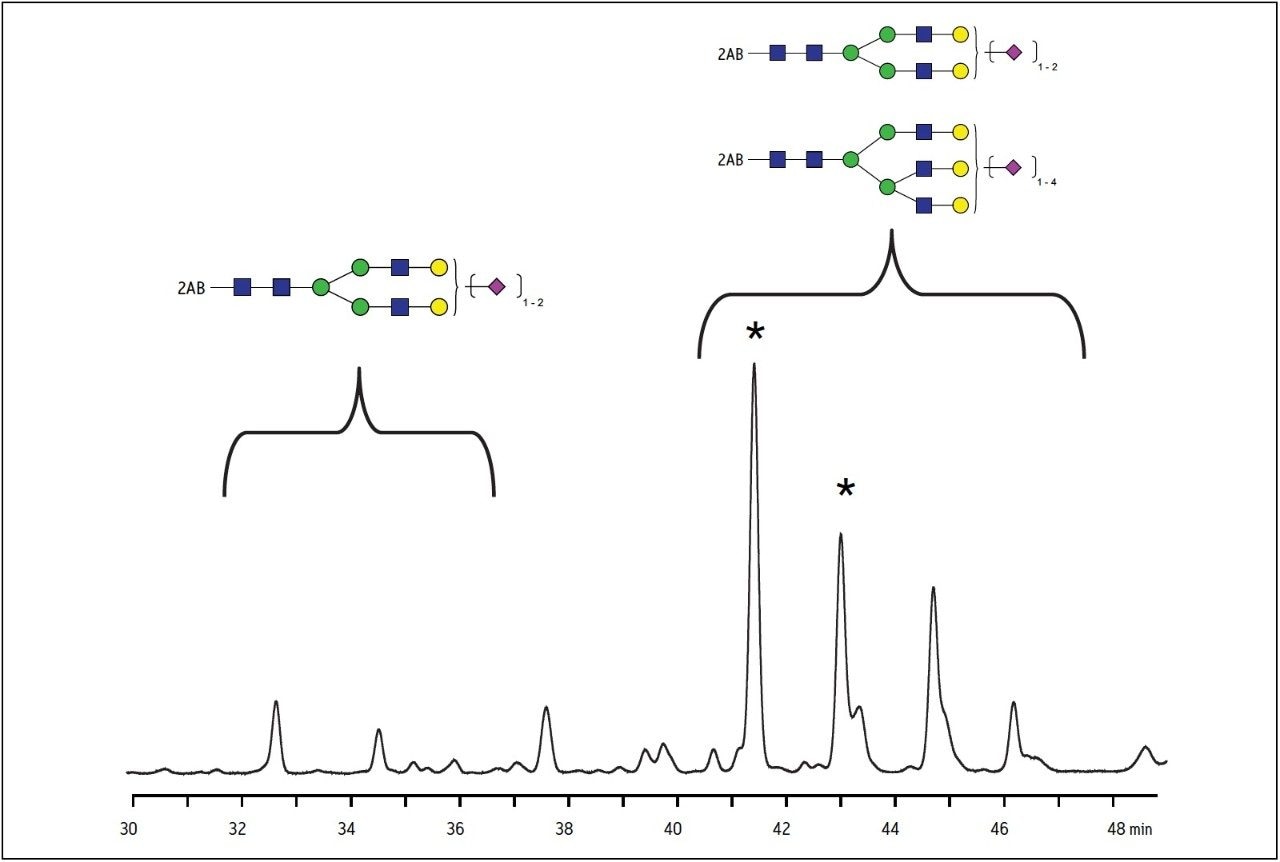
This separation chemistry is also suitable for charged, acidic glycans. Bovine fetuin yields a mixture of mono-, di-, tri-, and tetra-sialylated species that present a challenge to analysis. With larger glycan structures, the number of isomers increases. When sialic acid residues are added to glycan termini they can be attached on the different branches, and the linkage position on the galactose residue may be on the 2, 3, 4, or 6 position. The resolution of these structures is shown in Figure 5. A pair of isomers that are triantennary glycans with two sialic acid residues elute at 41.4 and 43.1 minutes
The Waters UPLC Glycan Separation technology columns, when operated with the ACQUITY UPLC instrument and FLR detector provide a highly-resolving, reproducible, rapid method for profiling glycans.
720003112, June 2009