Integrating LC–MS Spent Media Analytics with DoE for Rational Feed Optimization in mAb Manufacturing
Richard Jeske, Fang Lu, Yun Alelyunas, Julie Wushensky, Chandler Przybylski, Rui Chen
Waters Corporation, Immerse™ Lab Delaware, United States
Published on March 18, 2026
Abstract
Optimizing amino acid supplementation in fed-batch Chinese hamster ovary (CHO) cultures is critical for maximizing monoclonal antibody (mAb) productivity while minimizing accumulation of inhibitory metabolites. In this study, high-resolution LC-MS based spent media analysis was used to quantitatively track amino acid depletion and accumulation during a 14-day fed-batch mAb production process, revealing nutrient limitations not apparent from biomass or titer measurements alone. These data guided selection of aspartic acid, cystine, and asparagine for targeted supplementation using a stoichiometrically balanced, glucose-linked feeding strategy evaluated via a resolution IV fractional factorial design. Study results showed that cystine and aspartic acid supplementation improved yields by 7.6% and 3.6% respectively with a 10.6% increase when aspartic acid and cystine were supplemented in combination. In contrast, asparagine supplementation increased cumulative biomass by ~7.3% but promoted ammonium (10–15 mM) and lactate accumulation (>2 g/L), reducing specific productivity and final mAb yield by ~19.1% on average. These results demonstrate that LC–MS enabled spent media analysis provides a powerful, data-driven framework for identifying amino acid–specific metabolic bottlenecks and designing rational feed strategies, enabling optimization of mAb production while maintaining metabolic health in CHO cell cultures.
Benefits
- By enabling simultaneous, time-resolved measurements of multiple media components, LC–MS based spent media analysis supports identification of true nutrient limitations, discrimination between beneficial and detrimental supplementation strategies, and mechanistic interpretation of productivity outcomes.
- When integrated with statistically designed experiments, LC–MS data reduce empirical trial-and-error, accelerate feed optimization, and generate structured datasets that are well suited for model-based and predictive process control applications in biomanufacturing.
Introduction
Recombinant protein manufacturing is commonly performed using fed-batch cultures to achieve high cell densities, extend production duration, and reduce overall cost. Batch and feed media formulations typically contain more than 50 components, including amino acids, vitamins, and nucleosides, to support cellular energy demands and biomass formation. Compounding this complexity, CHO cell lines exhibit distinct metabolic preferences, and amino acid utilization varies with both cell line and mAb product, complicating identification of productivity-limiting nutrients.1,2
While depletion of essential nutrients can directly limit productivity, accumulation of excess feed components or metabolic intermediates can also impair cell growth and protein secretion. Feed strategies must therefore be both stoichiometrically balanced and dynamically matched to cellular demand.
LC–MS- based spent media analysis enables quantitative, molecularly resolved measurements of nutrient consumption and metabolite accumulation throughout a production process. By resolving individual media components across a wide dynamic range, LC–MS supports mechanistic interpretation of nutrient limitations that cannot be inferred from biomass or productivity metrics alone.
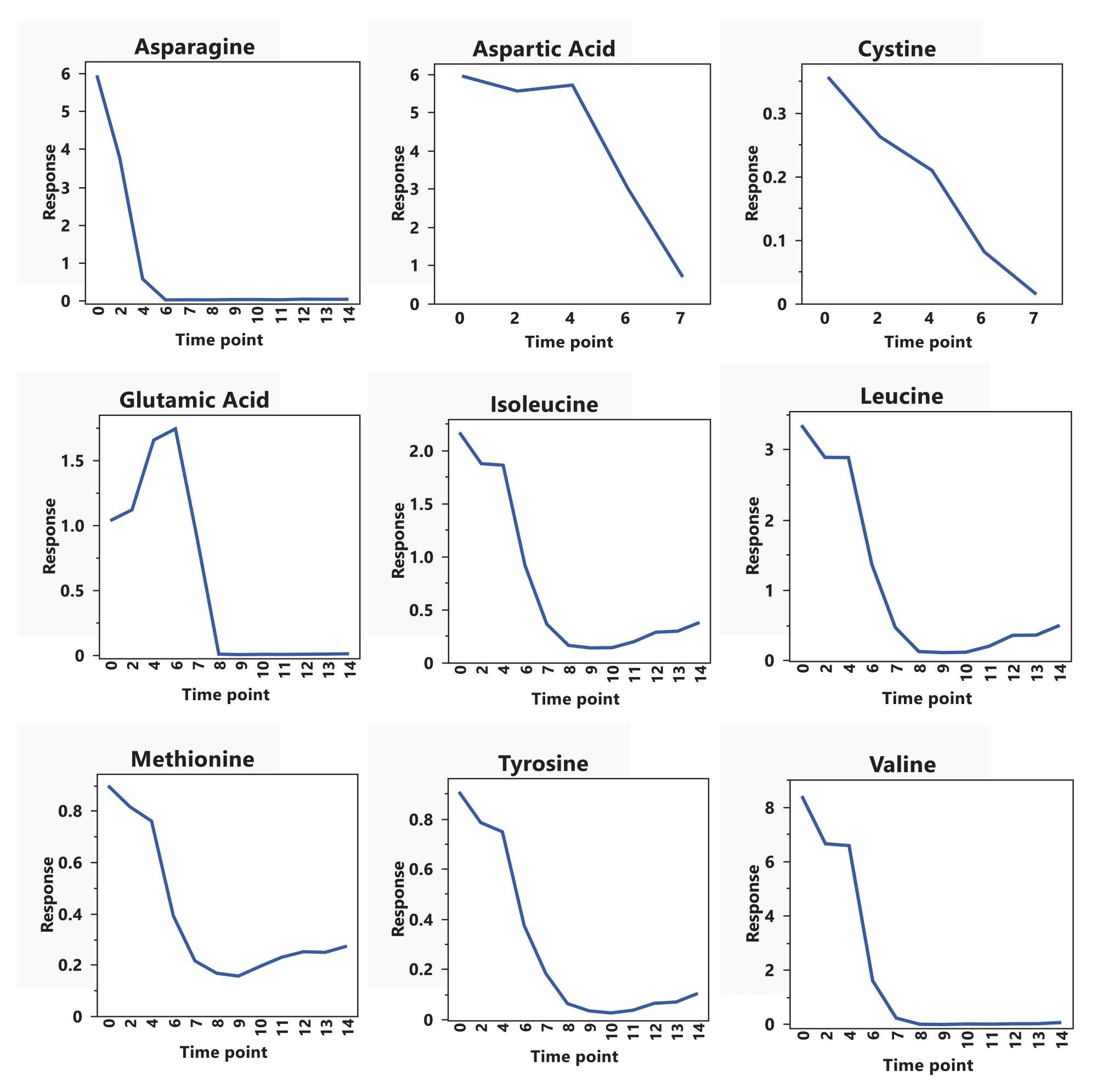
In our previous work, LC–MS analysis of spent media collected during a 14-day fed-batch mAb production process enabled direct quantification and ranking of nutrient depletion, facilitating prioritization of candidate amino acids for supplementation based on cellular demand. Nine amino acids were identified as depleted to below 15% of their initial concentration by day 7 (Figure 1). In order to efficiently screen the impact of feeding back these nine depleted amino acids, a preliminary study (data not shown) was carried out using a Resolution III fractional factorial Design of Experiments (DoE). Culture performance was evaluated by comparing viable cell density, mAb titer, and waste metabolite accumulation against a vehicle control. Supplementation with aspartic acid, asparagine, valine, and cystine showed distinct impacts on one or more of the evaluated responses. However, Resolution III designs are unable to distinguish between main effects and potentially active two-way interactions. Therefore, this follow up study was designed using a Resolution IV fractional factorial DoE to ensure reported main factor responses were not a result of aliased two-factor interactions. This study evaluated whether replenishing depleted amino acids in stoichiometric proportion to glucose consumption would improve mAb productivity while minimizing accumulation of inhibitory waste metabolites. By integrating LC-MS based spent media analytics with statistically designed experimentation, this work demonstrates a broadly applicable framework for rational feed optimization and generates quantitative datasets suitable for model-based and predictive process control applications.
Materials and Methods
LC-MS Based Approach for Balanced Amino Acid Feed Design
High-resolution LC-MS was used to develop a stoichiometrically balanced feed strategy as it allows for simultaneous quantification of components while achieving sufficient sensitivity and dynamic range for resolving near-complete media depletion. Spent media samples from a historical 14-day fed-batch mAb production run (Figure 1) were analyzed and asparagine, aspartic acid, cystine, and valine consumption were quantified. Glucose concentrations were measured using a BioProfile® FLEX2 cell culture analyzer (Nova Biomedical), and glucose consumption over the same period was determined from these measurements. LC-MS derived amino acid consumption data were normalized to glucose consumption to calculate the molar ratio of each amino acid consumed per mole of glucose. These consumption rates were then used to define daily bolus feed quantities according to Equation (1).
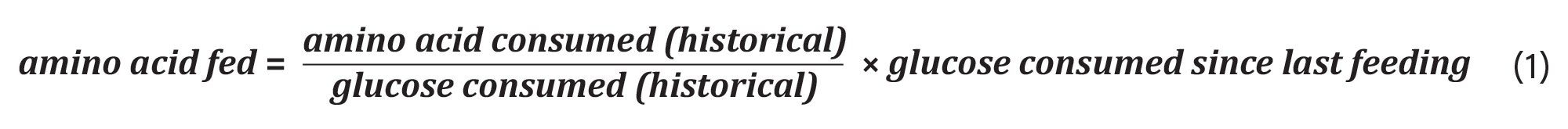
Because glucose was measured daily in the process, it was used as a reference nutrient to guide amino acid replenishment while minimizing the risk of overfeeding. Anchoring amino acid supplementation to glucose consumption, informed by LC–MS-derived consumption ratios, provided a practical framework for translating analytical data into actionable feed strategies. Amino acid feed stocks were prepared in 1x DPBS (10x DPBS stock solution: MilliporeSigma, cat. D1408), adjusted to pH 7.2, and sterile-filtered through 0.22 µm PVDF filters (MilliporeSigma, cat. SLGVM33RS).
Experimental
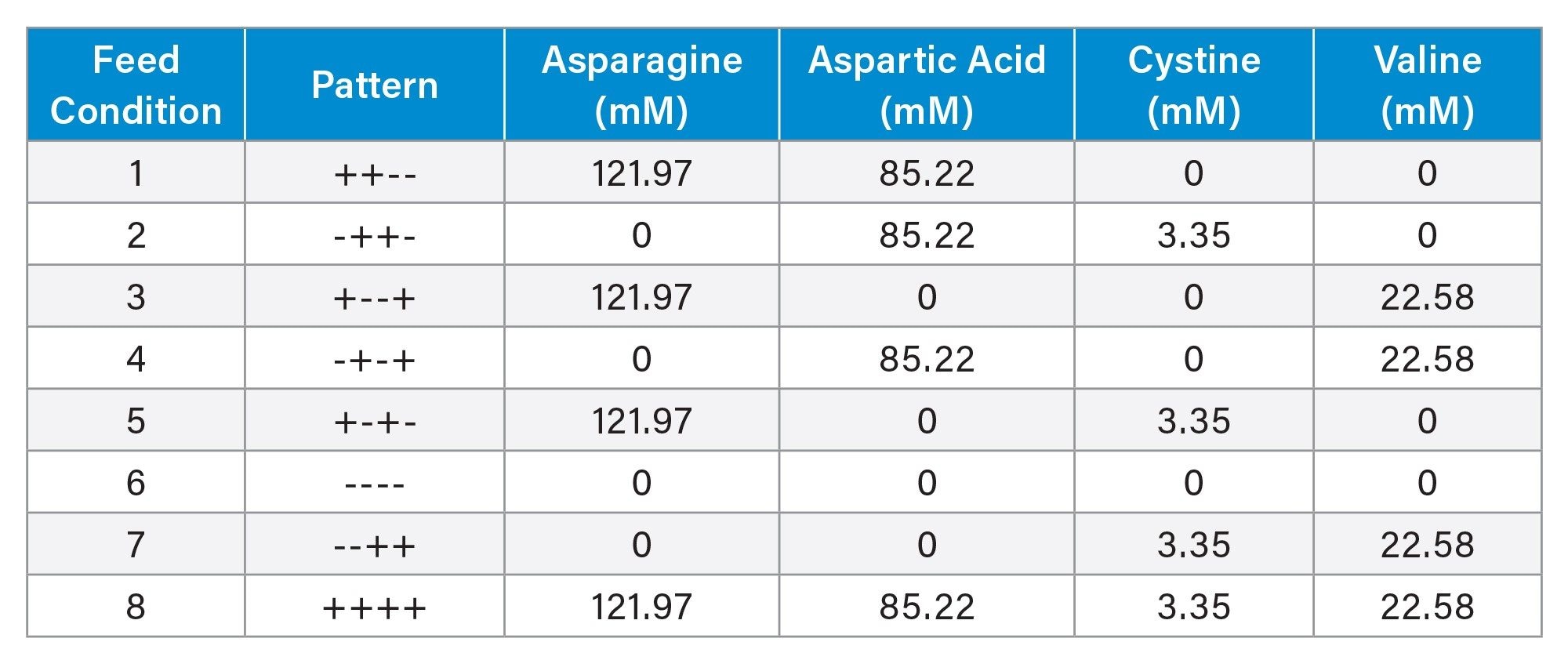
A (DoE) approach was employed to evaluate the impact of amino acid supplementation while capturing potential interactions between feed components. A Resolution IV fractional factorial design consisting of eight feed conditions was generated using JMP Software (Table 1). Feed concentrations shown in Table 1 correspond to the concentrations in the experimental amino acid feed solutions. Bolus feed volumes were adjusted daily to maintain stoichiometric feeding relative to glucose consumption. Conditions 5–8 were performed in duplicate.
Seed Train and 14-day Fed-Batch Shake Flask Culture
A commercially available CHO cell line producing mAb was thawed and cultured in 30 mL of EX-CELL® CD CHO Fusion medium (MilliporeSigma, cat. 14365C) in a 125 mL shake flask at 37 °C, 135 rpm (25 mm orbital throw), 80% relative humidity, and 5% CO2. Cells were passaged every 3 - 4 days by dilution into fresh medium and seeded at 0.3 - 0.4x106 cells/mL.
For fed-batch cultures, cells were inoculated at 0.5 × 10⁶ cells/mL in EX-CELL® Advanced CHO Fed-batch medium (MilliporeSigma, cat. 14365C). Starting on day 3, a 400 g/L glucose solution (MilliporeSigma, cat. G7021) was fed daily to bring the final glucose concentration to 6 g/L, and EX-CELL Advanced CHO Feed 1 (MilliporeSigma, cat. 24368C) was added at 2.5% (v/v). Daily feeding of experimental amino acid feeds started on day 5. All feeds were administered after sampling.
Glucose, ammonium, and lactate concentrations were measured using a BioProfile® FLEX2 (NOVA Biomedical). Cell count and viability were measured using a Cellaca PLX System (Revvity) and ViaStain™ AOPI Staining Solution (Revvity, part# CS2-0106). For sample clarification, cultures were centrifuged at 2000 x g for 10 minutes at 4 °C. Supernatant was filtered through a 0.22 μm PVDF filter (MilliporeSigma, cat. SLGV033NK) and stored at -80 °C until LC or LC-MS analysis.
Titer Determination using Protein A Affinity Chromatography
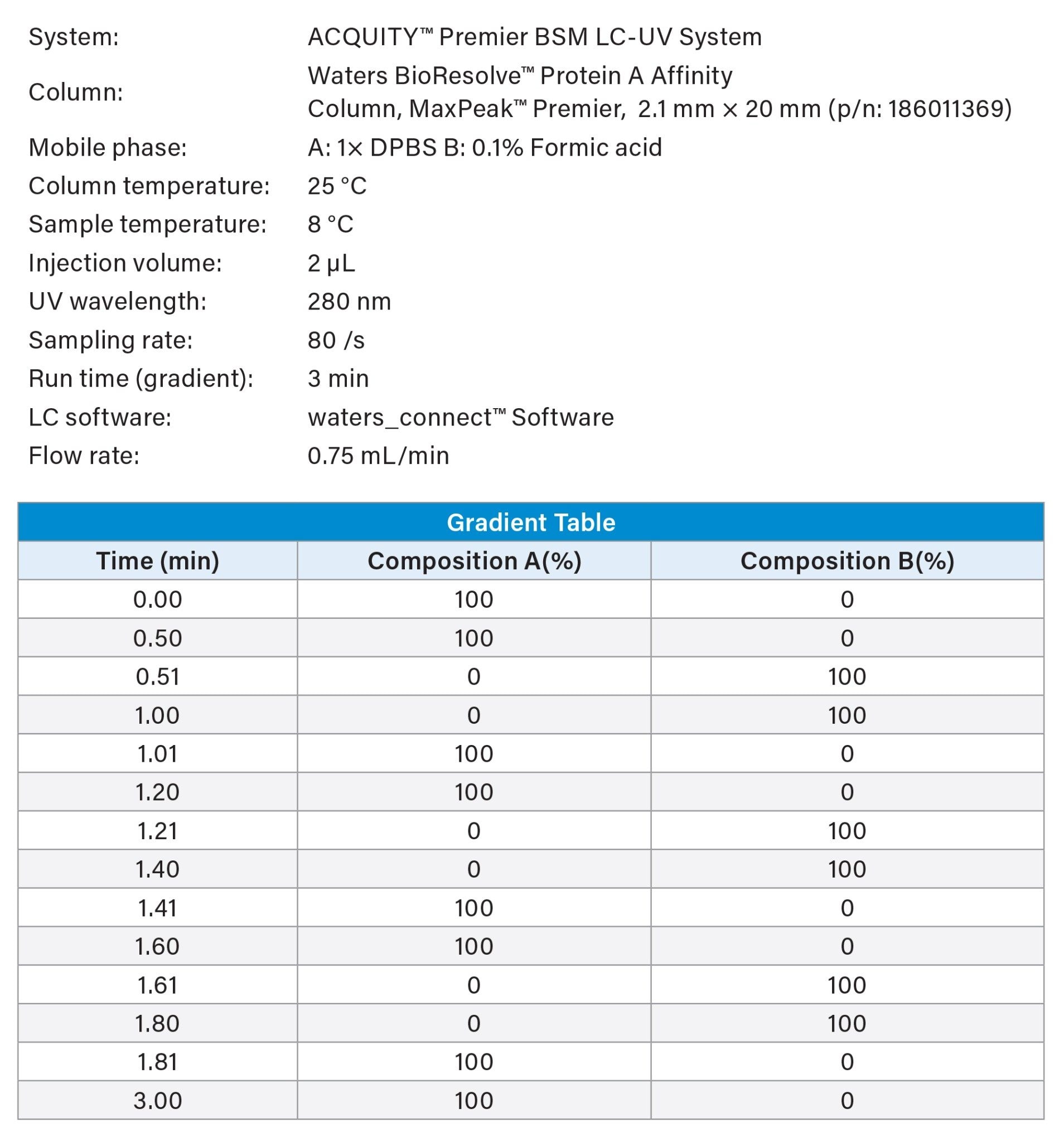
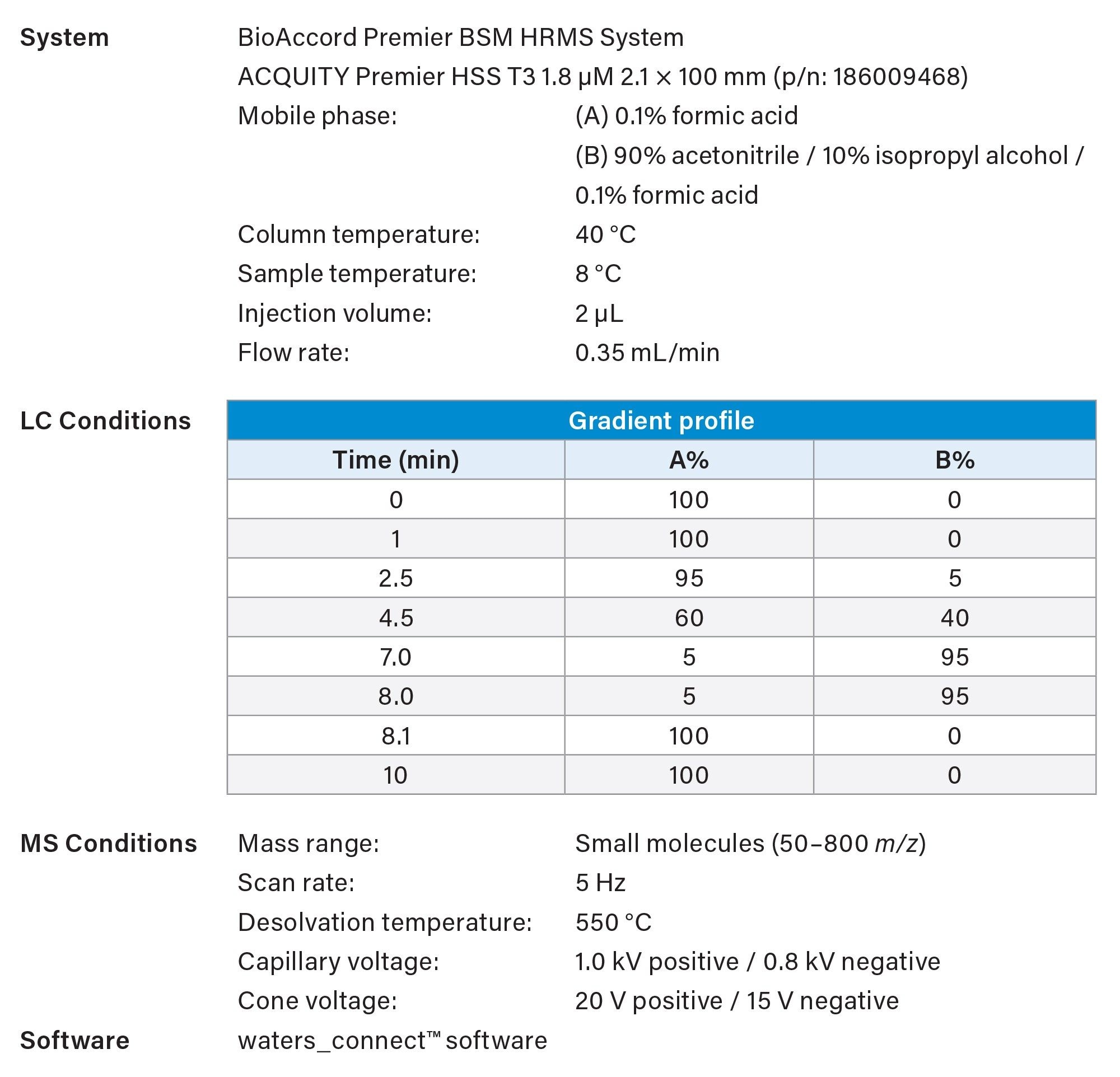
Product titers were measured using UPLC™ Protein A Affinity chromatography.3 Clarified spent media samples were injected directly without further preparation. Key experimental parameters are listed in Table 2.
Cell Culture Media and Metabolites Analysis
Cell culture media components and metabolites were quantified using LC–MS. Clarified spent media samples were diluted 400–2000 × in 0.1% formic acid and injected without further preparation. Key experimental parameters are listed in Table 3.
Results and Discussion
Amino Acid Feed Impact on Biomass and Waste Accumulation
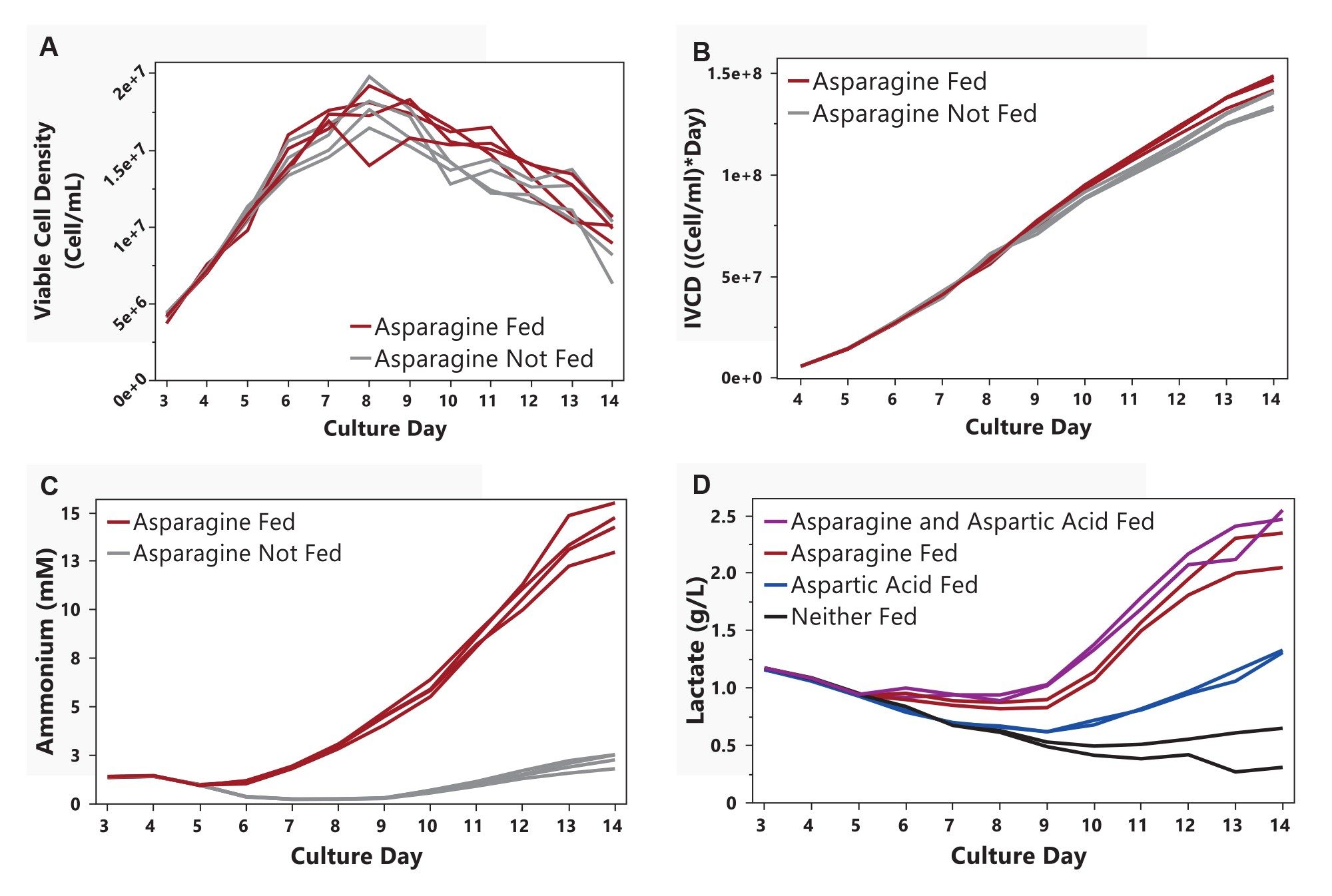
The impact of amino acid supplementation on cell growth, viability, and waste metabolite accumulation was evaluated (Figure 2). As shown in Figure 2A, amino acid supplementation did not increase growth rates or peak viable cell density (VCD). Cell cultures with asparagine supplementation maintained higher VCD values than those without asparagine supplementation during the production and cell death phases (from day 8 onward).
Integrated viable cell density (IVCD) represents cumulative live cell biomass available to produce mAb product. IVCD can be calculated using equation 2 where VCD is the viable cell density and t is the sampling time.
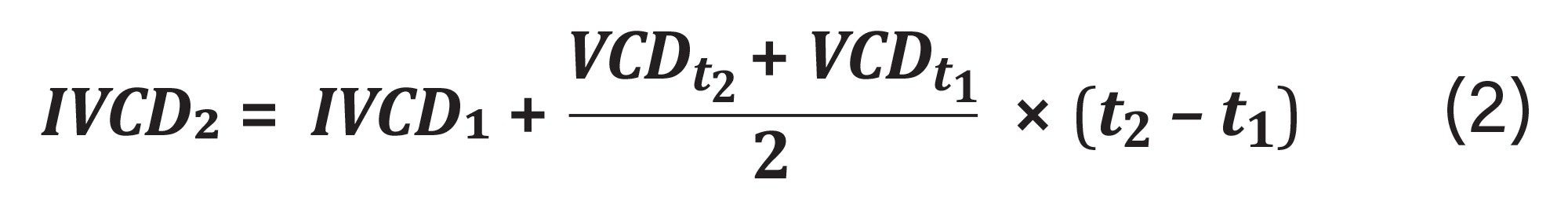
Regardless of feeding strategy, IVCDs were similar throughout the first 8 days of culture (Figure 2B). Conditions with asparagine supplementation diverged around day 9, resulting in increased cumulative biomass availability for potential mAb production. Backward stepwise regression identified asparagine as the only amino acid with a statistically significant effect, increasing IVCD by 7.3% on day 14.
The increased IVCD observed in asparagine-fed cultures was accompanied by substantial waste metabolite accumulation. Ammonium concentrations peaked between 10–15 mM in conditions fed asparagine while all other groups remained below 3 mM (Figure 2C). Similarly, cultures fed asparagine accumulated lactate concentrations exceeding 2 g/L, whereas other conditions did not exceed 1.5 g/L. Valine supplementation did not significantly affect biomass or waste accumulation under the conditions tested (data not shown).
Amino Acid Feed Impact on Product Yield and Specific Productivity
Representative day 14 Protein A Affinity chromatograms are shown in Figure 3A. Titers were similar across all conditions through day 7, after which asparagine-fed cultures diverged. By day 14, asparagine-fed cultures exhibited titers approximately 19.1% lower than the vehicle control (1.52 g/L vs 1.88 g/L) (Figure 3B). In cultures supplemented with aspartic acid, cystine, or both, titers ranged from 1.9% to 9.0% higher (1.91 g/L, 2.01 g/L, and 2.05 g/L, respectively) than the vehicle control (1.88 g/L).
To account for differences in final culture volume between vessels, total yield was calculated on day 14 as the product of titer and culture volume. The results are shown in Figure 3C. Total yield followed a trend similar to that observed for titer, with conditions fed cystine and aspartic acid generating yields above the vehicle control, while conditions fed asparagine yielded less product. In line with reduced yields, final-day ammonium and lactate concentrations showed a strong inverse relationship with yield in the asparagine-fed groups (Figure 3C).

Specific productivity (qP) was calculated using equation (3). Asparagine-fed conditions exhibited lower overall specific productivities, ranging from 9.7–11.08 pg/cell/day, while all other conditions were higher and ranged between 13.58–15.48 pg/cell/day. These results indicate that cells cultured without asparagine supplementation were intrinsically more productive on a per-cell basis.
Accumulation of ammonium above ~5 mM and lactate above ~50–60 mM has previously been associated with reduced growth and productivity in CHO cultures.4-6. In the asparagine-fed conditions evaluated here, ammonium levels reached 12–15 mM, well above this reported inhibitory threshold, and likely contributed to the observed reduction in specific productivity and final yield. Lactate levels reached 2–2.5 g/L (~22–28 mM) by day 14, which is below literature-reported inhibitory ranges but remained substantially higher than in the aspartic acid or cystine-fed groups, consistent with prior reports linking lower lactate accumulation to improved mAb production.6
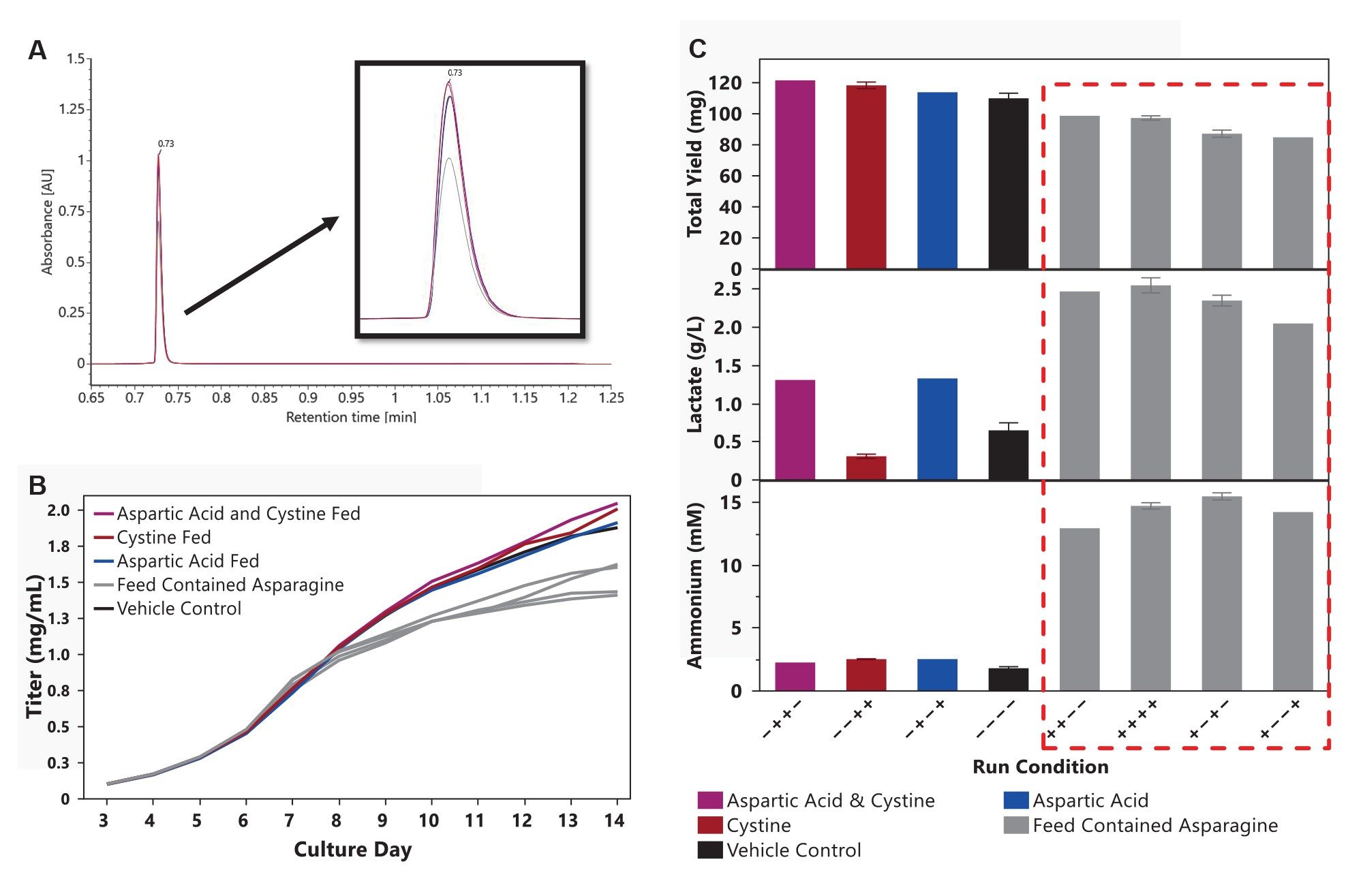
By comparison, aspartic acid fed cultures did not accumulate ammonium, consistent with distinct metabolic entry points into the TCA cycle: asparagine undergoes deamidation to aspartate with ammonium release, whereas aspartic acid directly transaminates to oxaloacetate without ammonium generation7. Given the high energetic demand of protein synthesis and secretion, aspartic acid supplementation may have alleviated TCA cycle bottlenecks, supporting higher ATP availability and improved productivity. Cystine supplementation similarly increased mAb production, consistent with its role in maintaining intracellular redox balance and mitigating endoplasmic reticulum stress, as previously reported8, and did not lead to accumulation of by-products such as lactate or ammonium (Figure 3C).
Overall, these results demonstrate that although asparagine supplementation increased cumulative biomass, the associated accumulation of inhibitory metabolites likely reduced per-cell productivity and final yield, indicating that prolonged asparagine supplementation should be avoided to preserve optimal mAb production.
Model Based Yield Optimization and Feed Interaction Analysis
A regression model was constructed using backward stepwise regression (p-value threshold of 0.05) to relate final yield to amino acid supplementation. As shown in Figure 4A, the main effects of aspartic acid, asparagine, and cystine, as well as multiple interaction terms were retained. Scaled parameter estimates indicate the direction and relative magnitude of yield changes associated with each amino acid and their interactions. Model predictions aligned closely with observed experimental values (Figure 4B) showing high linearity (R2 = 0.986) and close agreement to measured yields (RMSE = 2.138). Residual analysis revealed no systematic patterns, indicating an adequate model fit within the tested design space.
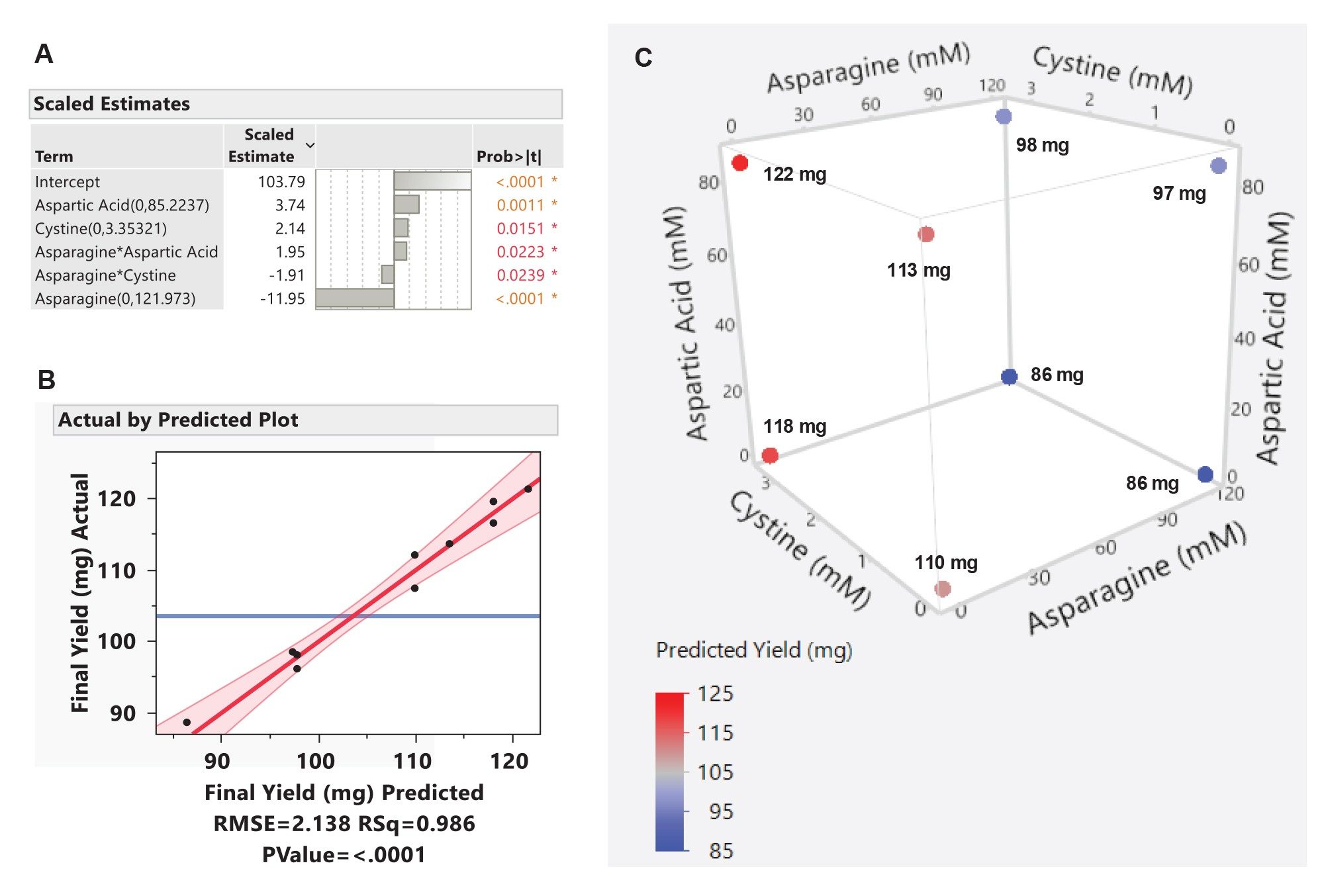
Figure 4C depicts the predicted yields at each vertex of the experimental design space. Feeding cystine and aspartic acid was predicted to increase yield by 7.3% (118 mg vs 110 mg) and 2.7% (113 mg vs 110 mg), respectively, while asparagine feeding was predicted to decrease yield by 21.8% (86 mg vs 110 mg). The largest predicted improvement (10.6%) occurred when cystine and aspartic acid were fed in combination. This synergistic effect on mAb production likely reflects their complementary metabolic roles: aspartic acid supports TCA cycle flux and cellular energy generation without ammonium release, while cystine contributes to maintenance of intracellular redox balance and mitigation of endoplasmic reticulum stress during high protein secretion demand. Collectively, these effects support higher per-cell productivity without accumulation of inhibitory waste metabolites.
Amino Acid Supplementation Impact on Media Depletion
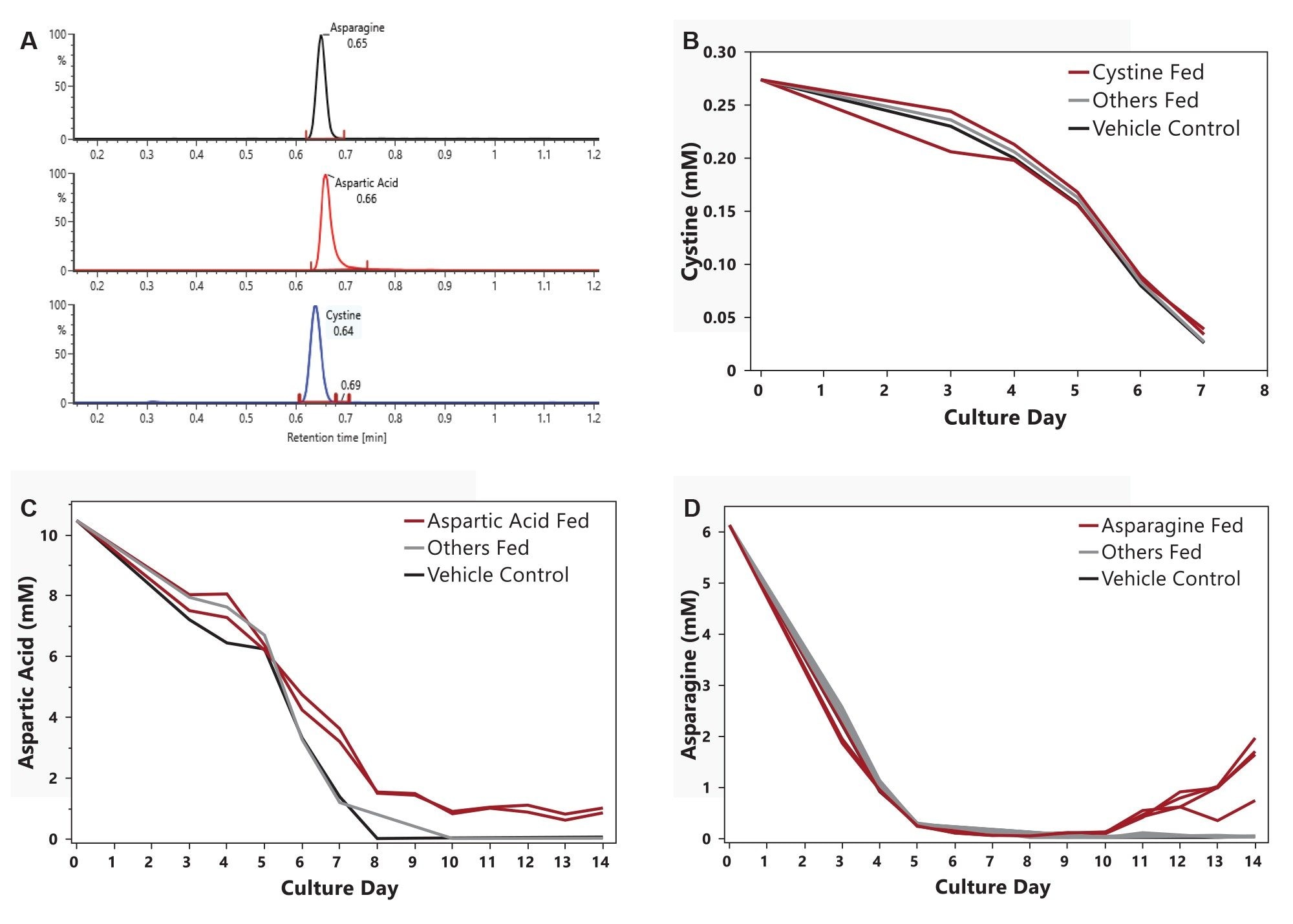
LC–MS analysis of spent media provided quantitative insight into amino acid depletion and accumulation dynamics across feeding conditions. Day 0 extracted ion chromatograms (XICs) of asparagine, aspartic acid and cystine are shown in Figure 5A. Despite daily supplementation that increased cystine concentrations by approximately 0.04–0.08 mM, cystine was rapidly depleted to below the limit of quantification (LOQ) by day 7 and remained below LOQ for the remainder of the culture (Figure 5B). These results indicate that the applied supplementation strategy was insufficient to offset rapid cellular uptake, identifying cystine as a persistent metabolic bottleneck.
Figure 5C shows a different trend for conditions fed aspartic acid. Depletion slowed after day 8, three days following aspartic acid supplementation. Concentrations then stabilized near 1 mM, suggesting that the feeding strategy adequately met cellular demand.
Figure 5D depicts all experimental conditions, including those fed asparagine. Extracellular asparagine concentrations followed a similar trajectory until day 11, when asparagine-fed cultures began to accumulate excess asparagine. It is noted that asparagine-fed cultures already exhibited reduced productivity and lower titers prior to the extracellular accumulation, indicating that reduced productivity was not driven by excess extracellular asparagine but likely by associated metabolic by-products, particularly ammonium. These results demonstrate how LC–MS based spent media analysis enables mechanistic interpretation of nutrient limitations and metabolic stress when integrated with DoE methodologies.
Conclusion
Amino acid metabolism in CHO cell cultures is highly context dependent, necessitating systematic evaluation of feed strategies for each process. By integrating LC-MS based media analysis with DoE methodologies, this study identified an amino acid feed strategy predicted to increase final mAb yield by 10.6% in CHO cultures.
Aspartic acid and cystine supplementation enhanced productivity through complementary mechanisms: aspartic acid supported central carbon metabolism without ammonium generation, while cystine supported redox balance and protein folding. In contrast, asparagine supplementation increased cumulative biomass but reduced specific productivity and final yield likely due to elevated ammonium accumulation. LC–MS derived amino acid profiles enabled quantitative evaluation of nutrient consumption and accumulation, allowing clear discrimination between beneficial and detrimental supplementation strategies.
Finally, these findings extend beyond a single process and contribute to datasets suitable for predictive, model-based process control. Overall, this work illustrates how LC–MS can function as a central analytical pillar for data-driven bioprocess optimization in monoclonal antibody manufacturing.
References
- Comparative analysis of amino acid metabolism and transport in CHO variants with different levels of productivity. Sarantos Kyriakopoulos et al., Journal of Biotechnology 168 (2013) 543–551
- Using Metabolomics to Identify Cell Line-Independent Indicators of Growth Inhibition for Chinese Hamster Ovary Cell-Based Bioprocesses Nicholas Alden et al., Metabolites 2020, 10, 199; doi:10.3390/metabo10050199
- Alelyunas et al., Analytical LC-MS Platform Methodologies to Support Upstream Bioprocess Optimization. Waters Application Note. 720008953. August 2025.
- Effects of Ammonium and Lactate on Growth and Metabolism of a Recombinant Chinese Hamster Ovary Cell Culture. Mio-Sam Lao et al., Biotechnol. Prog. 1997, 13, 688–691
- Identifying Inhibitory Threshold Values of Repressing Metabolites in CHO Cell Culture Using Multivariate Analysis Methods. Zizhuo Xing et al, Biotechnol. Prog. 2008, 24, 675–683
- Decreasing lactate level and increasing antibody production in Chinese Hamster Ovary cells (CHO) by reducing the expression of lactate dehydrogenase and pyruvate dehydrogenase kinases. Meixia Zhou at al., Journal of Biotechnology 153 (2011) 27–34
- Genome-Scale Modeling of CHO Cells Unravel the Critical Role of Asparagine in Cell Culture Feed Media. Kuin Tian Pang et al., Biotechnology Journal, 2024; 19:e202400072
- Multi-Omics Reveals Impact of Cysteine Feed Concentration and Resulting Redox Imbalance on Cellular Energy Metabolism and Specific Productivity in CHO Cell Bioprocessing. Amr S. Ali et al., Biotechnol J. 2020 August ; 15(8): e1900565
Featured Products
720009251, March 2026