Enhanced Recovery and Peak Shape of Acidic Peptides with BioResolve™ 1 mm ID Columns with MaxPeak™ Premier Technology
Caitlin M. Hanna, Stephan M. Koza, Balasubrahmanyam Addepalli
Waters Corporation, United States
Published on February 16, 2026
Abstract
Microflow chromatography is increasingly favored in proteomics for its enhanced sensitivity and reduced solvent consumption. However, the recovery and peak shape of acidic peptides remain challenging due to non-specific adsorption to stainless steel column hardware. This application note evaluates the performance of Waters BioResolve 1 mm ID Columns with MaxPeak Premier Technology, which incorporate MaxPeak High Performance Surfaces (HPS) Technology to mitigate unwanted interactions between acidic analytes and column hardware. Using the MassPREP™ Enolase Digest with Phosphopeptides Mix, the BioResolve BEH™ C18 RP MaxPeak Premier 1 mm ID Column was compared to a conventional stainless steel ACQUITY™ Peptide BEH C18 Column. The MaxPeak Premier Column demonstrates superior recovery and peak shape for acidic peptides from the first injection, eliminating the need for extensive column conditioning. Additionally, it maintains comparable resolution and peak capacity, exhibits excellent column-to-column reproducibility, and is compatible with both microflow and analytical UHPLC systems.
Benefits
- Improved recovery and peak shape of acidic analytes including phosphopeptides while maintaining resolution and peak capacity
- Excellent column-to-column reproducibility
- Versatile compatibility with microflow and analytical UHPLC systems
Introduction
Microflow chromatography offers several advantages over traditional analytical scale chromatography in proteomics applications. Microbore columns require less sample load and enable the use of low flow rates (10–100 µL/min). These low flow rates decrease solvent consumption and optimize the interface with MS, increasing MS sensitivity. Waters microbore columns with a 1 mm internal diameter (ID) demonstrate robust separation capabilities and enable sensitive MS detection in proteomics workflows.1,2 However, recovery and peak shape of acidic peptides using microbore columns remains a challenge due to non-specific adsorption to the column hardware.
Waters MaxPeak Premier Columns use MaxPeak HPS Technology to mitigate unwanted interactions between acidic analytes and column hardware. In analytical-scale chromatography, HPS Technology improves recovery of acidic peptides, including phosphopeptides, and reduces the amount of column conditioning required to achieve reproducible chromatography.3,4 This application note describes the benefits of HPS Technology in microflow chromatography using Waters BioResolve Peptide 1 mm ID Columns with MaxPeak Premier Technology. The BioResolve Premier Peptide 1 mm Columns yield improved recovery and peak shape of acidic peptides relative to the stainless steel equivalent while maintaining comparable resolution and peak capacity. Moreover, these columns exhibit excellent column-to-column reproducibility and can be used on both microflow and standard analytical UHPLC systems.
Experimental
Sample Description
Waters MassPREP Enolase Digest with Phosphopeptides Mix (p/n: 186003286) was reconstituted in 100 µL of 0.1% formic acid in water.
Calculations
4σ peak capacities were determined based on the following equation:
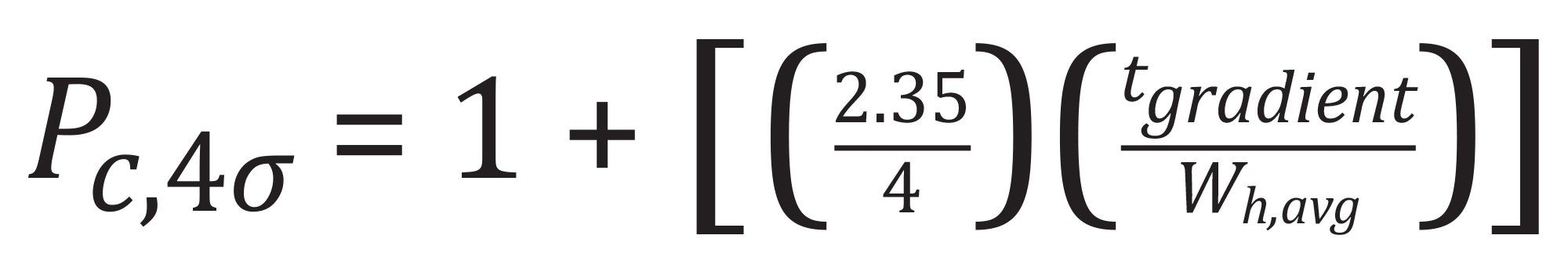
LC Conditions
|
LC system and setup: |
ACQUITY UPLC™ M-Class System: 75 µm ID x 30” ZenFit™ tubing (p/n: 700011513) to connect the micro sample manager (µSM) to the 20 µL mixer (p/n: 289003345), 75 µm ID x 14” ZenFit tubing (p/n: 700011506) to connect the mixer to the micro binary solvent manager (µBSM), 75 µm ID x 26 in ZenFit tubing (p/n: 700011505) to connect µBSM to column. Flow diverted straight from the column to the MS source with 40 µm ID x 30 in tubing (p/n: 700011516). ACQUITY Premier UPLC System: Standard tubing prior to column, 0025” tubing to connect column to the MS divert valve. |
|
Columns: |
Waters BioResolve Peptide C18 RP Column, MaxPeak Premier Technology, 1.7 µm, 300 Å, BEH, 1.0 x 50 mm (p/n: 186011513) ACQUITY Peptide BEH C18 300 Å Column, 1.7 µm, 1.0 x 50 mm (p/n: 186005592) |
|
Column temperature: |
60 °C |
|
Sample temperature: |
6 °C |
|
Injection volume: |
1 µL |
|
Mobile phase A: |
0.1% Formic Acid in H2O |
|
Mobile phase B: |
0.1% Formic Acid in ACN |
|
Sample vials: |
QuanRecovery™ MaxPeak 12 x 32 mm Propylene 300 µL Screw Cap vials (p/n: 186009186) |
Xevo™ G3 Detector Settings
|
ESI probe: |
Low-flow probe (p/n: 186007529) |
|
Mass range: |
50–2000 m/z |
|
Mode: |
ESI+ |
|
Sample rate: |
2 Hz |
|
Cone voltage: |
30 V |
|
Source temperature: |
120 °C |
|
Desolvation temperature: |
250 °C |
|
Capillary voltage: |
2.50 kV |
|
Cone gas: |
50 L/h |
|
Desolvation gas: |
350 L/h |
|
Lockmass: |
LeuEnk (556.27658 m/z) |
|
Informatics: |
MassLynx™ Software (M-class) and waters_connect™ Software (ACQUITY Premier UPLC) |
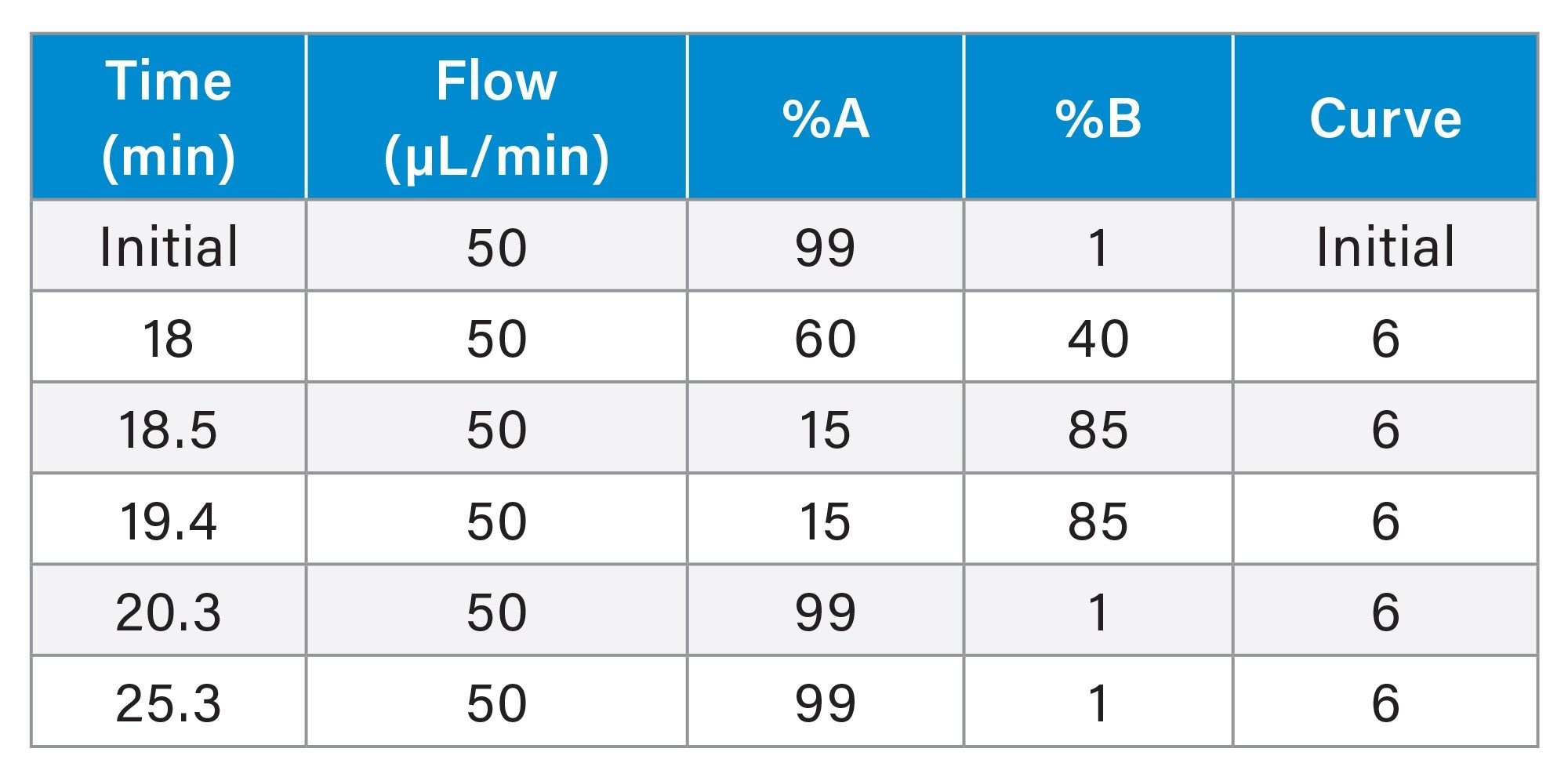
Gradient Table
Results and Discussion
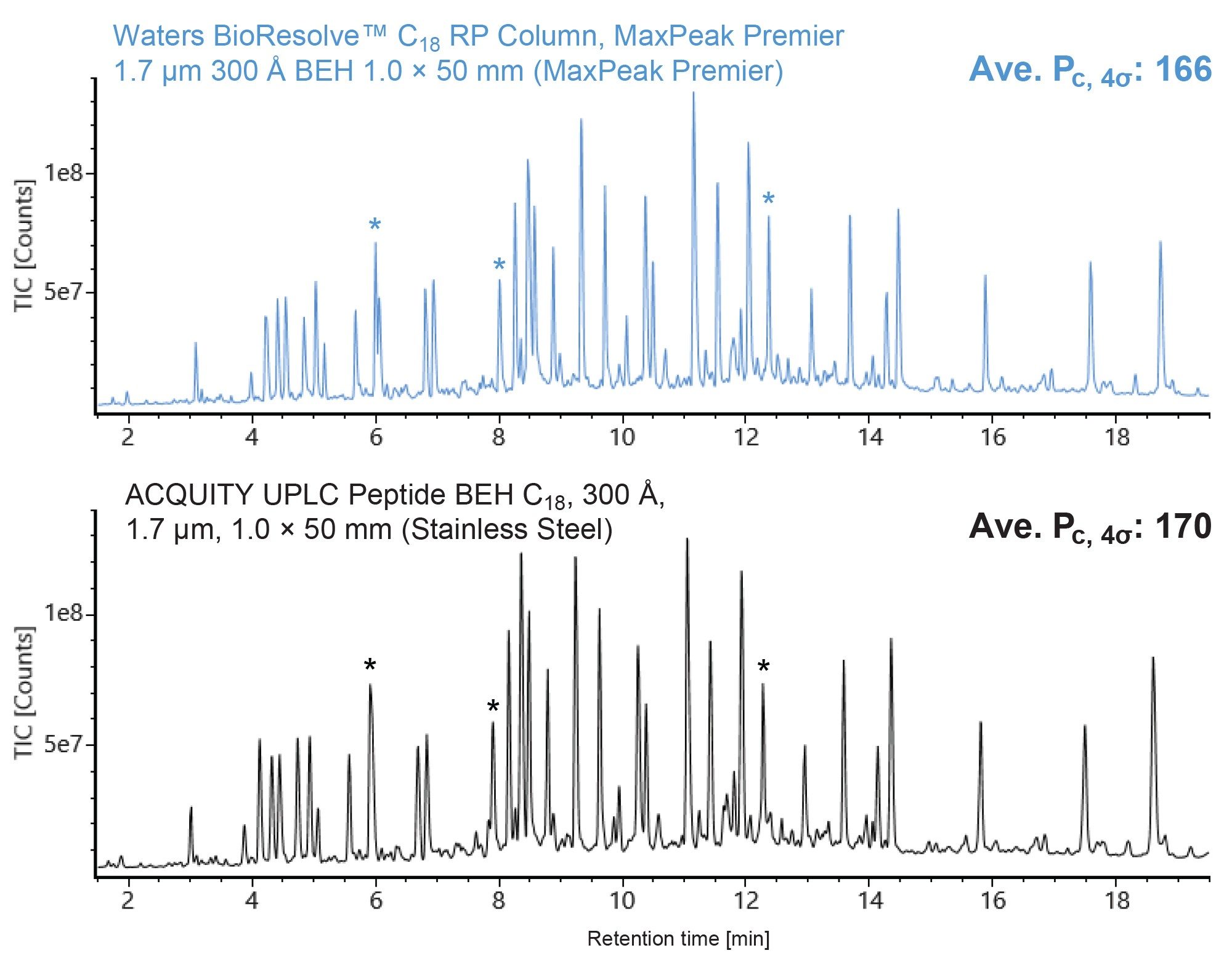
Figure 1 shows the total ion chromatograms (TICs) of the MassPREP Enolase Digest with Phosphopeptides Mix on a BioResolve Peptide C18 RP Column and a stainless steel ACQUITY Peptide BEH C18 Column. The two columns produce similar peak profiles with slight differences in resolution for certain peptides (see peaks denoted with an asterisk). The resolution differences observed are likely a result of differences in selectivity between the two columns. The peak capacity of the MaxPeak Premier Column is comparable to that of the stainless steel column.
HPS hardware mitigates unwanted interaction between acidic analytes and column hardware, resulting in high recovery and improved peak shape relative to stainless steel. Extracted ion chromatograms (XICs) for two acidic peptides on a BioResolve Peptide C18 RP Column and a stainless steel ACQUITY Peptide BEH C18 Column are shown in Figure 2. The T51 peptide possesses three glutamic acid residues in series plus an additional glutamic acid residue and two aspartic acid residues. The second peptide, T19p, is a phosphopeptide. Data is shown for the first and 10th injections on each column. The BioResolve C18 RP Column exhibits high recovery of the T51 peptide on the first injection; consistent with that observed for injection 10. On the stainless steel ACQUITY Peptide BEH C18 Column, low recovery of the T51 peptide is observed on the first injection. With conditioning, recovery increases to levels comparable to the BioResolve Premier Column by the 10th injection. Similar results are observed for peak shape of the T19p phosphopeptide. On the first injection, the T19p peptide exhibits significantly higher tailing on the stainless steel ACQUITY Peptide BEH C18 Column compared to the BioResolve Peptide C18 RP Column. Symmetrical peaks are observed by the 10th injection on both columns. These results highlight the improved recovery and peak shape benefits of HPS technology through microflow separations of acidic peptides.
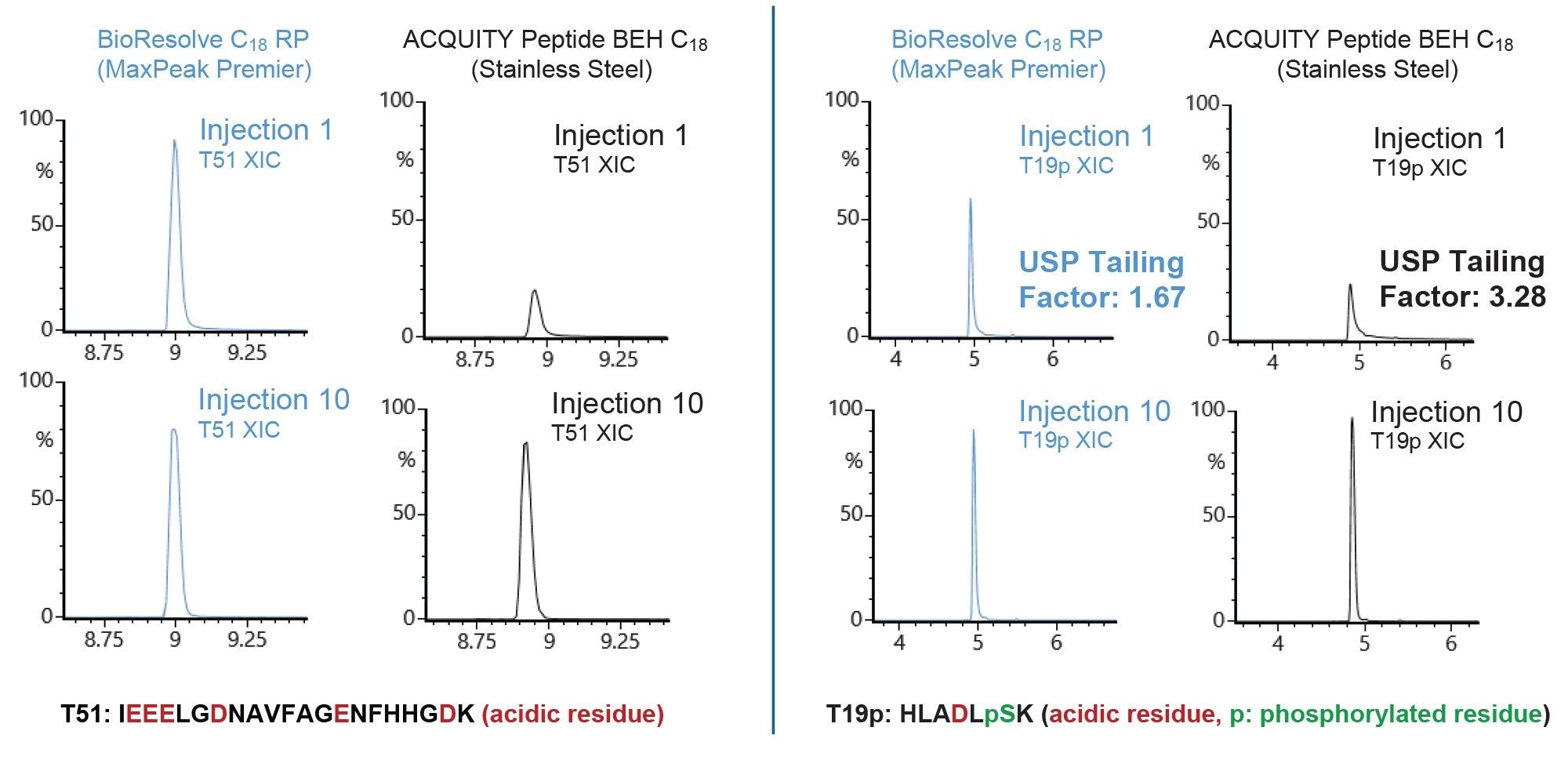
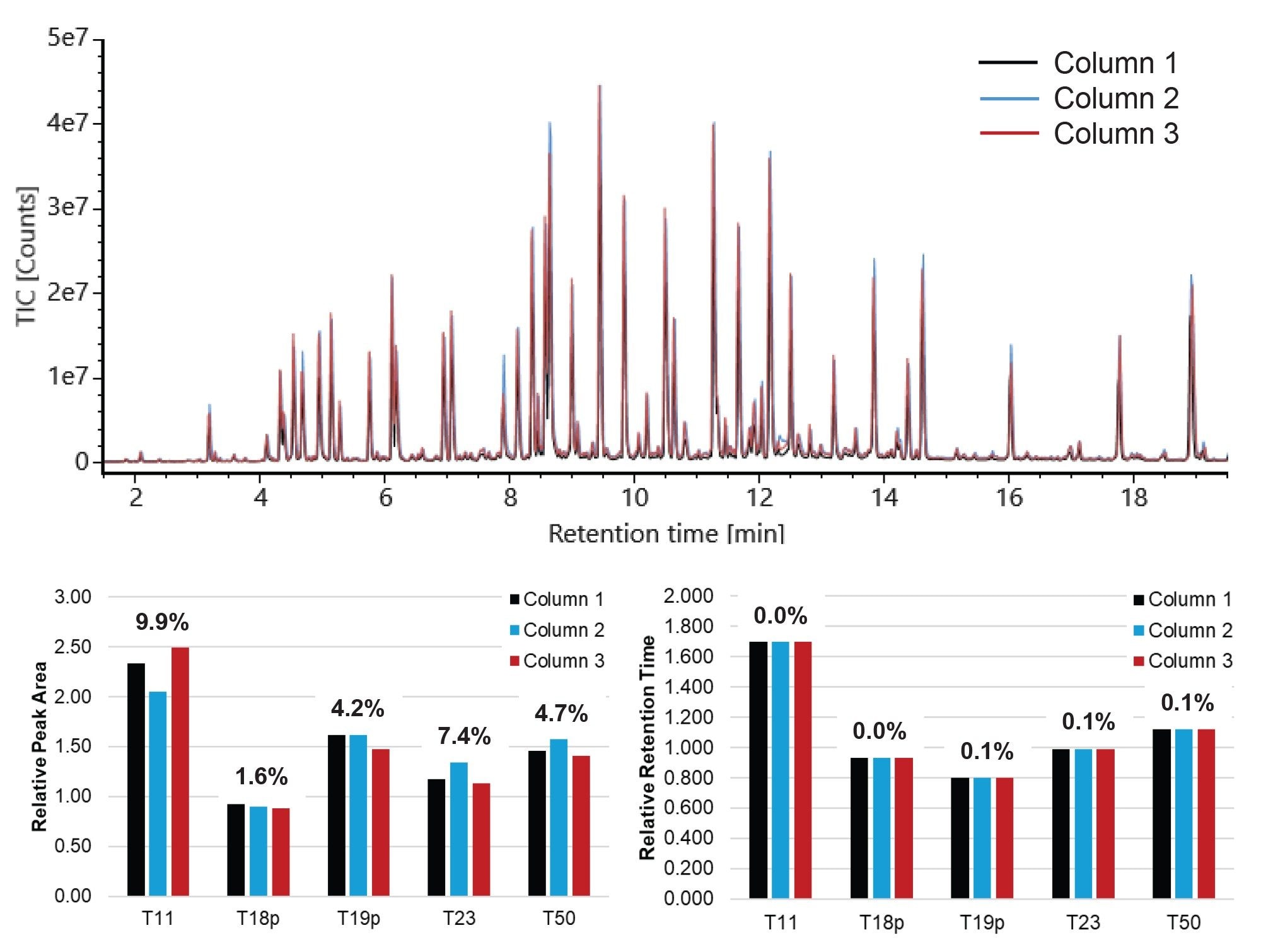
Figure 3 displays overlaid TICs of the MassPREP Enolase Digest with Phosphopeptides Mix on three BioResolve C18 RP 1 mm ID Columns. Excellent reproducibility is observed across the three columns. Using XIC data, relative peak areas and relative retention times were calculated against the T18 peptide. Relative retention time RSDs are less than 1% for each peptide and relative peak area RSDs are less than 10% for each peptide.
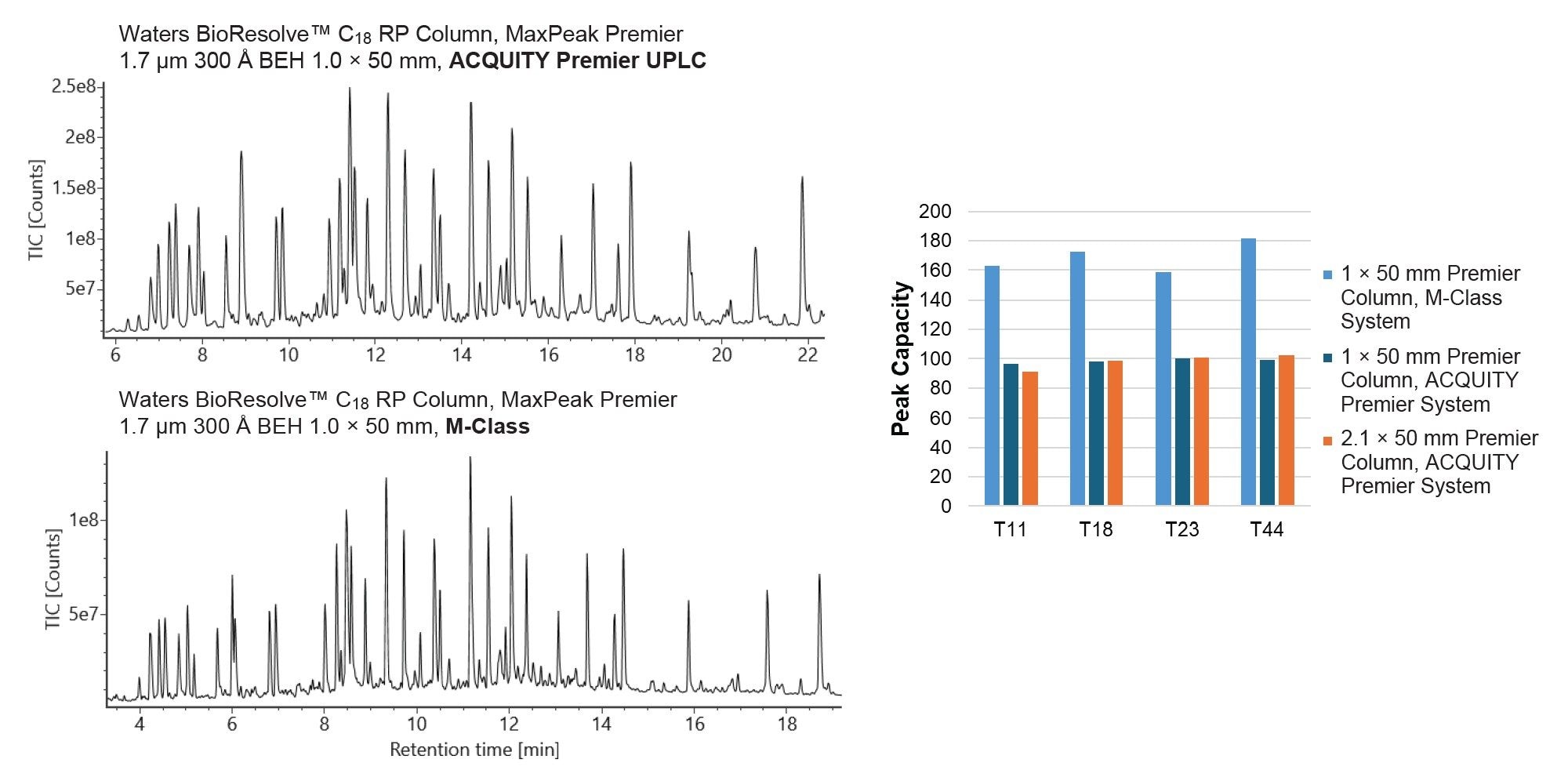
All data presented above were acquired using an ACQUITY UPLC M-Class System. Waters Premier 1 mm Columns can be used on both microflow and standard analytical flow UHPLC instruments. Figure 4 displays the TIC of the MassPREP Enolase Digest with Phosphopeptides Mix on a 1.0 x 50 mm BioResolve C18 RP Column using an ACQUITY Premier UPLC System. Comparable peak profiles are obtained using both the ACQUITY UPLC M-Class System and ACQUITY Premier UPLC System. As expected, peak capacity for the BioResolve C18 RP 1.0 x 50 mm Column decreases on the ACQUITY Premier UPLC System due to increased impact of post-column dispersion but is comparable to Premier 2.1 x 50 mm Columns run on the same system (Figure 4, right).
Conclusion
Waters BioResolve 1 mm ID Columns equipped with MaxPeak Premier Technology significantly enhance the chromatographic separation of acidic and phosphopeptides in microflow LC-MS workflows. Compared to stainless steel hardware, these columns deliver improved peptide recovery and peak shape from the first injection, reducing the need for conditioning. The MaxPeak Premier Columns maintain high resolution and peak capacity, demonstrate excellent reproducibility across columns, and are versatile across both microflow and analytical UHPLC platforms. These attributes make them a robust and reliable choice for proteomics applications requiring sensitive and reproducible analysis of acidic peptides. Additionally, these columns provide a significant solvent savings option compared to 2.1 mm ID analytical columns, lowering operating costs and contributing to greater ecological sustainability.
References
- Gao, Y., Fillmore, T. L., Munoz, N., Bentley, G. J., Johnson, C. W., Kim, J., Meadows, J. A., Zucker, J., Burnet, M. C., Lipton, A. K., Bilbao, A., Orton, D. J., Kim, Y., Moore, R. J., Robinson, E. W., Baker, S. E., Webb-Robertson, B. M., Guss, A. M., Gladden, J. M., Beckham, G. T., Magnuson, J. K., Burnum-Johnson, K. E. High-Throughput Large-Scale Targeted Proteomics Assays for Quantifying Pathway Proteins in Pseudomonas putida KT2440. Front. Bioeng. Biotechnol., 2020, 8, 1–13.
- Lennon, S., Hughes, C. J., Muazzam, A., Townsend, P. A., Gethings, L. A., Wilson, I. D., Plumb, R. S. High-Throughput Microbore Ultrahigh-Performance Liquid Chromatography-Ion Mobility-Enabled-Mass Spectrometry-Based Proteomics Methodology for the Exploratory Analysis of Serum Samples from Large Cohort Studies. J. Proteome Res. 2021, 20, 3, 1705–1715.
- Nguyen, J. M., Rzewuski, S. C., Lauber, M. A. Enhancing Phosphopeptide Quantitation Using ACQUITY Premier Peptide CSH C18 Columns. 2021, Waters App Note 720007211.
- Reed, C., Hong, P., Wong, N., Birdsall, R. E. Improving Peptide Mapping Separations Containing Phosphopeptides Using MaxPeak™ Premier Column Technology on an ACQUITY™ Premier System. 2023, Waters App Note 720008136.
Featured Products
720009231, February 2026