UPLC™ Separation of Fifteen Bisphenols Using a Waters Acquity™ Biphenyl RP Column with MaxPeak™ Premier Technology and UV Detection
Kenneth Berthelette, Chris Collins, Euan Ross, Thomas H Walter
Waters Corporation, United States
Published on April 13, 2026
Abstract
Bisphenols are a group of industrial chemicals that have been detected in food and environmental samples. This is concerning because several bisphenols have been shown to exhibit endocrine‑disrupting activity. Here we describe a UPLC method that provides baseline separation of 15 bisphenols in eight minutes using a Waters Acquity Biphenyl RP Column with MaxPeak Premier Technology. This column delivered a superior separation as compared to ACQUITY Premier BEH C18 and ACQUITY Premier BEH Phenyl Columns.
Benefits
- Baseline separation of 15 structurally similar bisphenol compounds using an inert column packed with BEH Biphenyl stationary phase.
- Improved selectivity for aromatic compounds due to enhanced π–π interactions with the biphenyl groups.
- Better resolution of structurally similar bisphenols compared to C18 or phenyl phases.
Introduction
Bisphenols are used to make plastics and epoxy resins for a variety of applications including polycarbonate containers, thermal printer paper, and coatings for food and drink cans.1 Bisphenol A and several other bisphenols have been shown to act as endocrine disruptors, affecting a number of biological functions including metabolic and developmental health.2-3 Due to their potential negative impact on human health, it is important to monitor these compounds not only in foods and consumer products but also in environmental samples. This can be challenging because the structural similarities of some of the compounds make separating them difficult. As shown in Figure 1, bisphenols contain two phenol groups linked by a bridging group at the para position of both hydroxyls. Additional functionality may be attached either to the aryl rings or to the bridging group to modify the properties of the compound. The different bisphenols are identified by one- or two-letter descriptors (e.g. bisphenol A, abbreviated BPA).
As with all method development activities, especially for challenging sample mixtures, it is important to screen different stationary phase chemistries to identify the one that best separates the compounds of interest.4-7 The bonded groups play a significant role in how the analytes interact with the stationary phase in reversed-phase liquid chromatography (RPLC). Typical C18 bonded phases separate and retain analytes primarily through hydrophobic interactions. In the case of bisphenols, additional retention mechanisms may be needed to separate the compounds that have similar hydrophobicity. In such cases, using a phenyl bonded phase may be appropriate since these stationary phases not only separate by hydrophobicity, but also through π–π interactions between the phenyl groups and any analytes possessing π electrons.8 This secondary retention mechanism is strongest when using non-acetonitrile containing mobile phases. Adding additional phenyl groups to the bonded phase can further increase retention and separation for analytes possessing π electrons.9 Biphenyl stationary phases are one example of bonded phases that provide greater π–π interactions than phenyl materials.10
This application brief looks at the separation of 15 bisphenol compounds using three different stationary phases all packed in inert hardware: BEH C18, BEH Phenyl, and BEH Biphenyl. It was found that the column packed with Acquity BEH Biphenyl was the only one that achieved baseline separation of all 15 bisphenol compounds while the other two columns exhibited at least one set of co-eluting peaks.
Experimental
Method Conditions
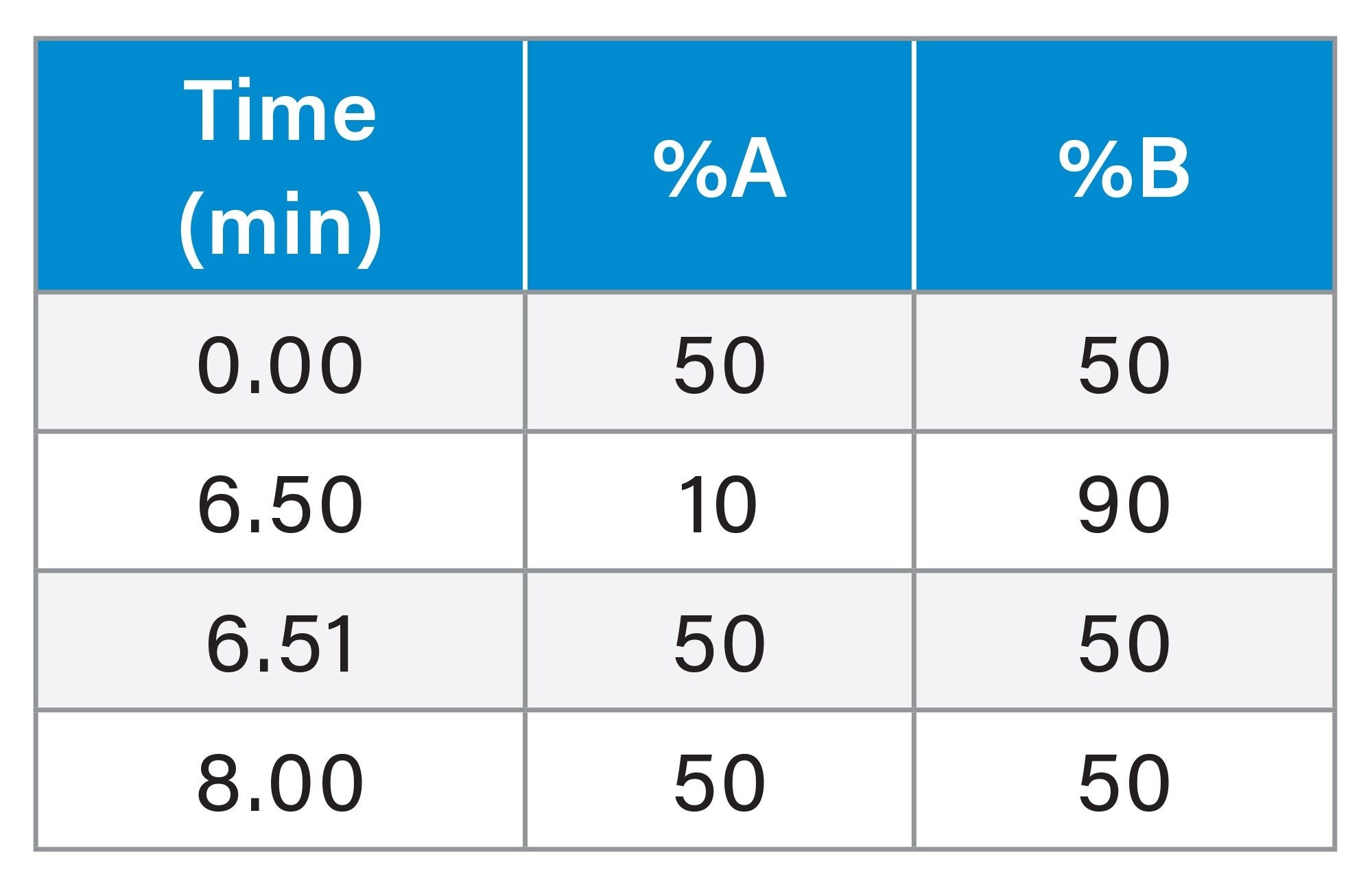
|
System: |
ACQUITY I-Class System with PDA Detector (HPS flowpath) |
|
Columns: |
BEH Phenyl 2.1 x 50 mm 1.7 µm BEH C18, 2.1 x 50 mm, 1.7 µm BEH Biphenyl, 2.1 x 50 mm, 1.7 µm |
|
Mobile phase A: |
Water |
|
Mobile phase B: |
Methanol |
|
Flow rate: |
0.45 mL/min |
|
Gradient: |
See Gradient Table |
|
Column temperature: |
25 °C |
|
Injection volume: |
1.0 µL |
|
Sample: |
15 bisphenols combined at concentrations shown on next slide, using a 75:25 water:methanol diluent |
|
Detection: |
UV @ 220 nm |
Gradient Table
Results and Discussion
A solution containing 15 bisphenol compounds was prepared at a final concentration of 0.1 mg/mL in water and introduced into an Acquity Premier Binary Solvent Manager System for analysis. The structures of the compounds tested are shown in Figure 1.

The system was equipped with a Tunable UV Detector set to 220 nm, as well as a Xevo™ TQ-S micro Mass Spectrometer operated in a scan range of 50–500 m/z in negative ion electrospray ionization mode. A methanol gradient with a linear ramp of 50–90% over 6.5 minutes was used to separate the compounds with water as the weak mobile phase component. The mobile phase contained no additives or pH adjustments of any kind. The column temperature was set to 25 °C and the flow rate was 0.45 mL/min. The columns were all 2.1 x 50 mm and were packed with 1.7 µm particles. A 1.0 µL injection volume was used, and the sample was injected in triplicate on each column to confirm results. The peaks were identified using the mass spectrometry data acquired.
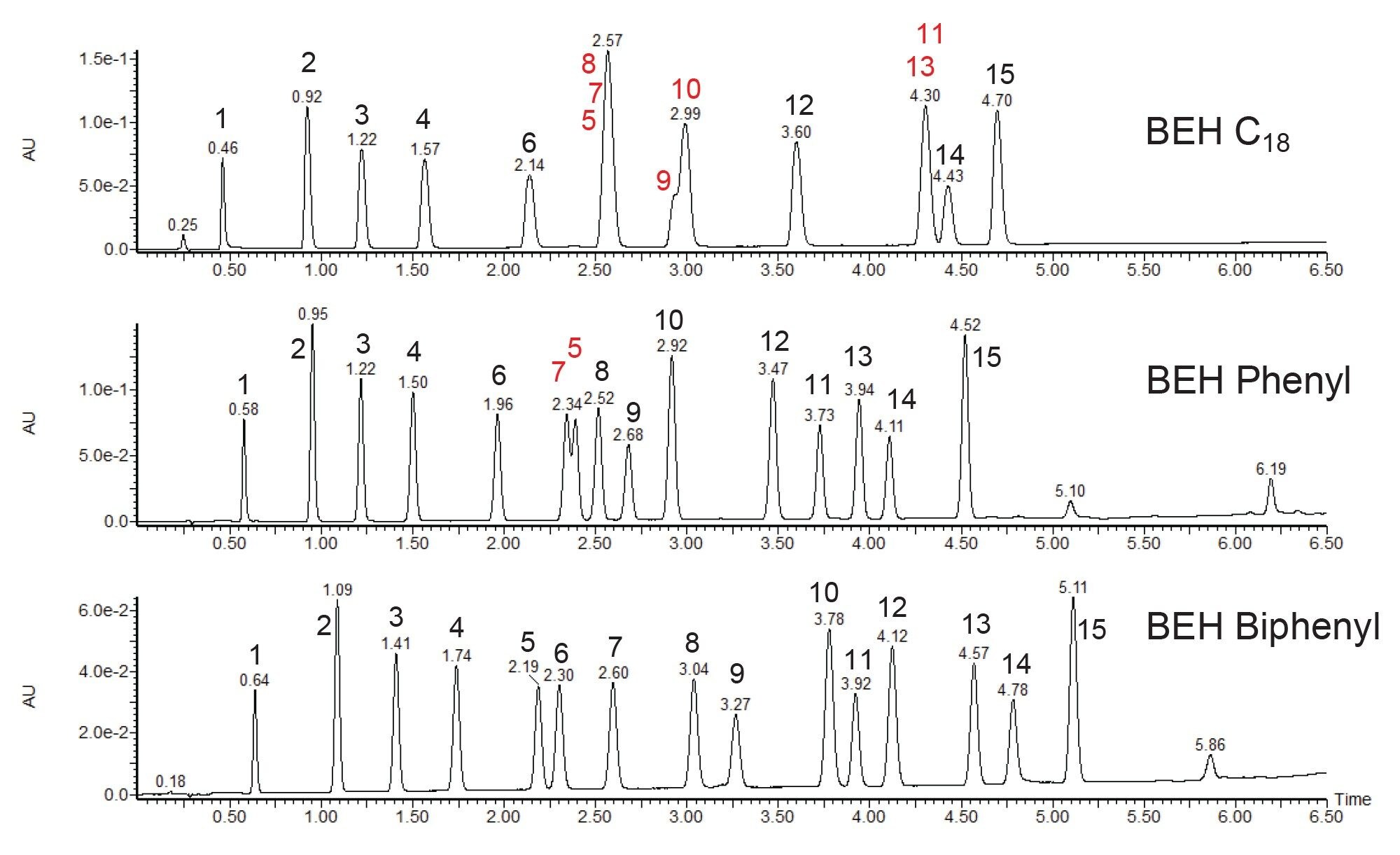
Representative chromatograms for each column are shown in Figure 2 along with peak identifications. The BEH C18 stationary phase didn’t separate all the compounds; in fact, it produced three sets of co-eluting peaks. While some of these compounds may still be distinguished by MS using multiple reaction monitoring (MRM), resolving them chromatographically is preferred as it reduces possible interferences in the MS and simplifies data analysis. The BEH Phenyl stationary phase performed considerably better but still produced one pair of partially co-eluting peaks. This suggests that the π–π interactions that are possible with the phenyl column are beneficial for the separation of these compounds. With only one set of co-eluting peaks, method optimization could be performed to achieve a suitable separation, however that would likely require a longer column and increased analysis time. Instead, the BEH Biphenyl stationary phase was investigated to analyze these compounds.
The additional phenyl group in the biphenyl bonded phase adds interaction sites between the stationary phase and the analytes, leading to increased separation based on the π-electron content of the compounds. The resulting chromatogram shows that the column packed with BEH Biphenyl stationary phase fully separated the bisphenols, allowing for accurate results while also enabling UV detection as an alternative or complement to MS monitoring. The selectivity of the BEH Biphenyl stationary phase provided the best separation without requiring further method optimization or the use of a longer column to improve the resolution of co-eluting peaks. This example highlights the importance of screening multiple columns during method development to optimize selectivity and provide the best separation possible.
Conclusion
The monitoring of bisphenols in food and environmental samples can benefit from a method to rapidly separate and detect as many bisphenol compounds as possible in a single run. When analyzing 15 bisphenol compounds, an inert hardware column packed with BEH Biphenyl stationary phase was shown to separate all compounds while BEH C18 and BEH Phenyl columns did not. The greater π–π interactions provided by the biphenyl column are believed to offer improved selectivity for this group of analytes.
References
- Bisphenols- ECHA Website https://echa.europa.eu/hot-topics/bisphenols Accessed 7-Jan-2026.
- Saal F, Vandenberg L. Update on the Health Effects of Bisphenol A: Overwhelming Evidence of Harm. Endocrinology 162 (2020): 1–25.
- Eladak S, Grisin T, Moison D, et al. A New Chapter in Bisphenol A Story: Bisphenol S and Bisphenol F are not safe alternatives to this compound. Fertil. Steril. 103 (2015): 11–21.
- Hong P, McConville P. A Complete Solution to Perform a Systematic Screening Protocol for LC Method Development. Wates White Paper. 720005268.
- Berthelette K, Aiello M, Collins C, Walter TH. Development of a UPLC Method for a Forced Degradation Study of Radioligand Therapy Precursor PSMA-617. Waters Application Note. 720009042, September 2025.
- Dolan J. Stability Indicating Assays. LCGC North America 20 (4) (2002): 346–349.
- Shah B, Jain S, Prajapati K, Mansuri N. Stability Indicating HPLC Method Development: A Review. Int. J, Pharm, Sci. 3 (2012): 2978–2988.
- Croes K, Steffens A, Marchand DH, Snyder LR. Relevance of π–π and dipole-dipole interactions for retention on cyano and phenyl columns in reversed-phase liquid chromatography. J. Chromatogr. A 1098 (2005) 123–130.
- Sun Y, Sun Z, Wang C, Wei Y. Effect of phenyl numbers in polyphenyl ligand on retention properties of aromatic stationary phases. J. Chromatogr. A 1674 (2022) 463152.
- Taniguchi A, Hirose T, Shimotsuma M. Retention and Selectivity Differences due to Molecular Structure of Phenyl-Type Reversed-Phase HPLC Columns. Chromatography 46 (2025): 55–61.
720009250, April 2026