Development of a LC-UV Method for Separating 12 Steroids using a Waters Acquity™ Biphenyl RP Column with MaxPeak™ Technology
Kenneth Berthelette, Christopher Collins, Thomas H Walter
Waters Corporation, United States
Published on April 13, 2026
Abstract
In this application note, a suitable LC-UV method was developed for the separation of a mixture of 12 steroids. Six columns were tested using low pH mobile phases along with either acetonitrile or methanol as the strong solvent. The initial scouting showed that the Waters Acquity Biphenyl RP Column with MaxPeak Premier Technology (BEH™ Biphenyl Column), using acetonitrile provided the best overall separation with all 12 compounds resolved. Optimization of the method was performed, both to improve cycle time as well as to separate an impurity peak from a main peak. Finally, a stainless-steel column was packed with the BEH Biphenyl stationary phase and compared to the same material in MaxPeak Premier Column hardware. The MaxPeak Column allowed the detection of betamethasone phosphate, which was not observed when using the conventional stainless-steel column.
Benefits
- Baseline separation of 12 steroids
- Reduction of cycle time through optimization of the gradient
- Excellent recovery of betamethasone phosphate using MaxPeak Premier Column hardware
Introduction
LC method development relied on having a diverse set of columns with which to test. This is especially true for complex mixtures with many structurally similar compounds. Separating such components is not only challenging due to their similar chemical properties, but identification can be an issue as well. If the compounds are isobaric, mass spectrometry can’t be used to distinguish them. Having a diverse panel of columns for method development means selecting them accordingly, which is difficult due to the number of stationary phases available. However, selecting the columns judiciously and employing a structured protocol can streamline method development.
In this application note, 12 steroids, including a steroid phosphate, were analyzed using a modified systematic screening protocol. Of the 12 analytes, there are three sets of isobaric compounds, making it critically important to separate them so that they can be individually quantified. Monitoring multiple steroids is important for diagnosing a range of disorders. The systematic screening protocol has been shown to be highly effective for developing methods quickly.1-3 Coupling the protocol with MaxPeak Premier Columns that incorporate High Performance Surface (HPS) technology greatly improves the process. MaxPeak Premier Column hardware is specifically designed to mitigate interactions between analytes, especially charged acidic compounds, and stainless-steel column hardware.4-8 This means that for initial scouting as a part of method development, the data collected is accurate, allowing for simplified decision-making regarding column and method conditions. The steroids were analyzed using a total of six column chemistries with formic acid modified mobile phases and either acetonitrile or methanol as the strong solvent. Explanations behind the column choice, and selection of the final method conditions are discussed. Method optimization, including a comparison of column hardware was then performed and shows that for the steroid phosphate, the use of MaxPeak Premier Column hardware is critical. Baseline resolution of all compounds was achieved under the final method conditions.
Experimental
Sample Description
Stock solutions of each standard were created or purchased at 1 mg/mL. A mixture was created containing 50 µg/mL of each of the compounds with a final sample composition of approximately 31:69 v/v acetonitrile: water. Figure 1 shows the chemical structures of the compounds tested.
Method Conditions
LC Conditions
|
LC system: |
ACQUITY Premier QSM System with CM and 2 CM Aux, and PDA Detector |
|
Detection: |
UV 275 nm |
|
Column: |
ACQUITY Premier BEH C18 Column, 2.1 x 50 mm, 1.7 µm (p/n: 186009452) ACQUITY Premier CSH Phenyl-Hexyl Column, 2.1 x 50 mm, 1.7 µm (p/n: 186009474) ACQUITY Premier HSS PFP Column, 2.1 x 50 mm, 1.8 µm (p/n: 186010036) Waters Acquity Biphenyl RP Column, 2.1 x 50 mm, 1.7 µm (p/n: 186011744) ACQUITY Premier BEH Phenyl Column, 2.1 x 50 mm, 1.7 µm (p/n: 186010336) CORTECS Premier C8 Column, 2.1 x 50 mm, 1.6 µm (p/n: 186011549) |
|
Column temperature: |
30 °C |
|
Sample temperature: |
10 °C |
|
Injection volume: |
5.0 µL |
|
Flow rate: |
0.4 mL/min |
|
Mobile phase A: |
Water |
|
Mobile phase B: |
Acetonitrile |
|
Mobile phase C: |
Methanol |
|
Mobile phase D: |
2% Formic Acid in Water |
|
Scouting gradient conditions: |
Constant 5% D to maintain formic acid concentration at 0.1%; initial mobile phase conditions 5% organic; linear organic ramp to 95% B in 6.86 minutes; hold for 1.14 minutes; return to initial conditions and re-equilibrate for 2.28 minutes. Total run time 10.3 minutes |
Data Management
|
Chromatography software |
Empower ™ Software |
Results and Discussion
The first step of the systematic screening protocol is to run the sample using both low and high pH modified mobile phases on a suitable column to assess retention of the analytes. For the mixture of steroids in this application note, this step was not performed. At both low and high pH, 11 of the 12 compounds present in the mixture are neutral and are therefore unaffected by the mobile phase pH. The 12 compound, betamethasone phosphate, is considerably more polar at elevated pH as the charge of the phosphate group transitions from -1 to -2. As such, only low pH mobile phases were used, as that provides the best retention for betamethasone phosphate, while the other compounds are unaffected.
Once the mobile phase pH has been determined, a more traditional column screening experiment is performed using either acetonitrile or methanol as the strong mobile phase. The system used for this work allows for up to six columns to be installed and screened, providing the opportunity to compare columns with diverse selectivities. The first chosen was the BEH C18 Column, which employs an ethylene-bridged hybrid organic/inorganic base particle and a full-coverage C18 stationary phase. This column is widely used and regarded as one of the most successful for RPLC method development. Next, a Charged Surface Hybrid (CSH) phenyl-hexyl column was selected as it uses a different base particle and bonded phase. The CSH particle incorporates a slight positive charge, improving the peak shape for basic analytes while also providing anion-exchange functionality. The phenyl-hexyl stationary phase differs from a traditional C18 in that the π electrons of the stationary phase can interact with the π electrons of an analyte, altering the selectivity. Next, a High Strength Silica (HSS) pentafluorophenyl (PFP) column was selected. The HSS particle is not an organic/inorganic hybrid like CSH and BEH, and has a higher concentration of silanols. It also has a smaller pore size and higher surface area, which results in greater retention than BEH particles. The PFP stationary phase allows not only for the π-π interactions mentioned previously but other secondary interactions as well.
The fourth selection was the BEH Biphenyl Column, which is based on the same BEH particle as the BEH C18 Column, but differs in the bonded phase. The biphenyl bonded phase boosts the potential for π-π interactions.9 The fifth column selected was the BEH Phenyl Column, chosen to compare not only the differences a base particle can make when compared to the CSH Phenyl-Hexyl, but also the difference that the biphenyl bonded phase can make. Lastly, a CORTECS C8 Column was selected as both the solid-core silica base particle and the C8 bonded phase are less retentive than the other phases selected. Given that these analytes are hydrophobic, there could be some benefit to eluting them earlier in a gradient run.
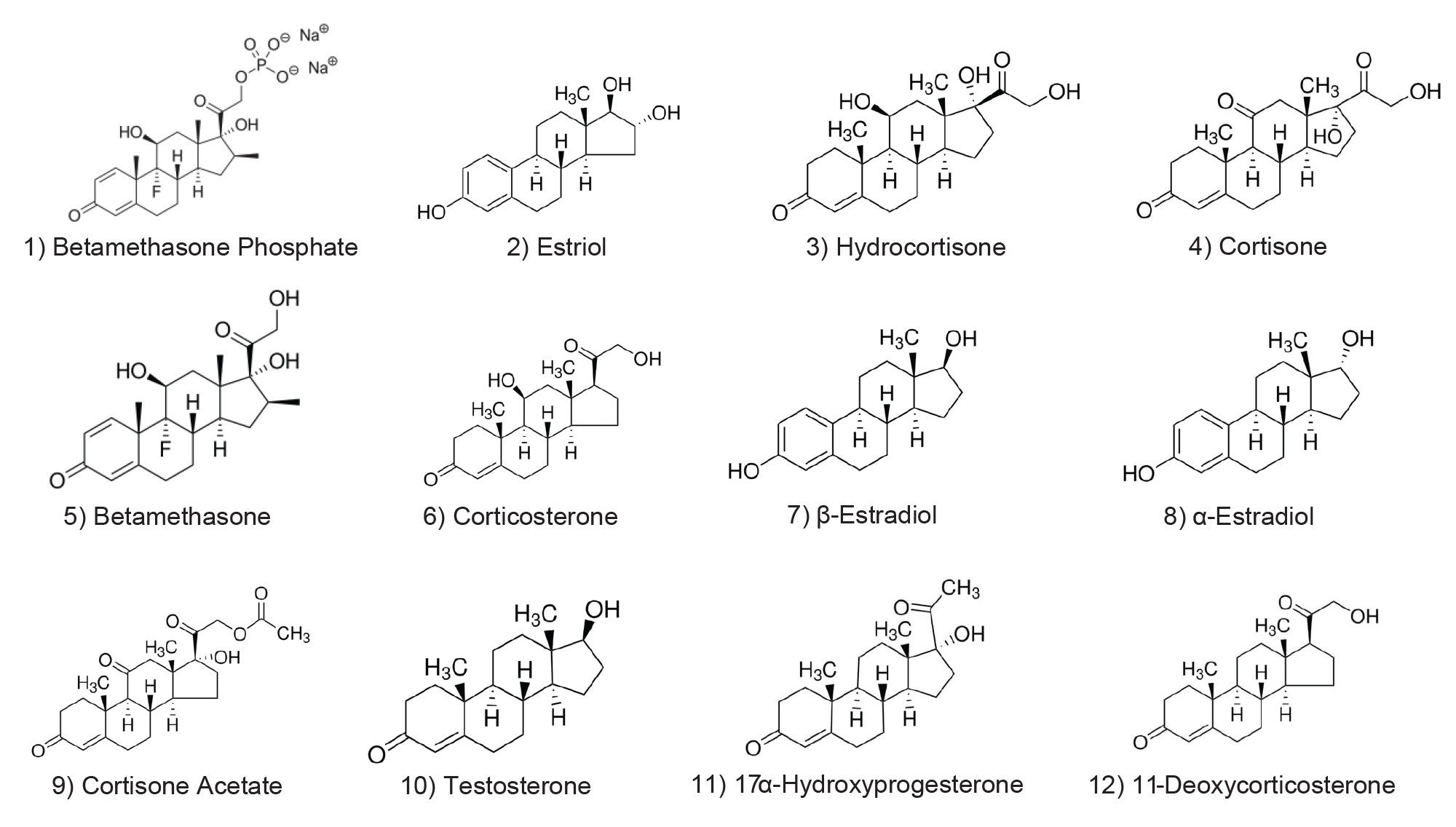
Two strong solvents were selected for this step of method development to increase the likelihood of finding a suitable separation. Acetonitrile and methanol are both popular strong solvents in RPLC, with acetonitrile often preferred due to the lower viscosity and therefore lower pressure associated with its use. Methanol, however, can facilitate π-π interactions between the analytes and the stationary phase, particularly phenyl, PFP, and biphenyl bonded phases. Figure 2 shows the separations of the sample achieved on the six selected columns using methanol, and Figure 3 shows the separations obtained with acetonitrile.
Using methanol, all the analytes were retained beyond 4 minutes, indicative of its weaker elution strength. In terms of selectivity, only the CORTECS C8 Column looks promising as all the peaks can be seen, albeit some with only partial resolution. The separations for the other columns show at least one set of analytes that completely co-elute, making any optimization efforts more challenging. The lack of good separation achieved with one strong solvent is an example of why screening more than one solvent is prudent.
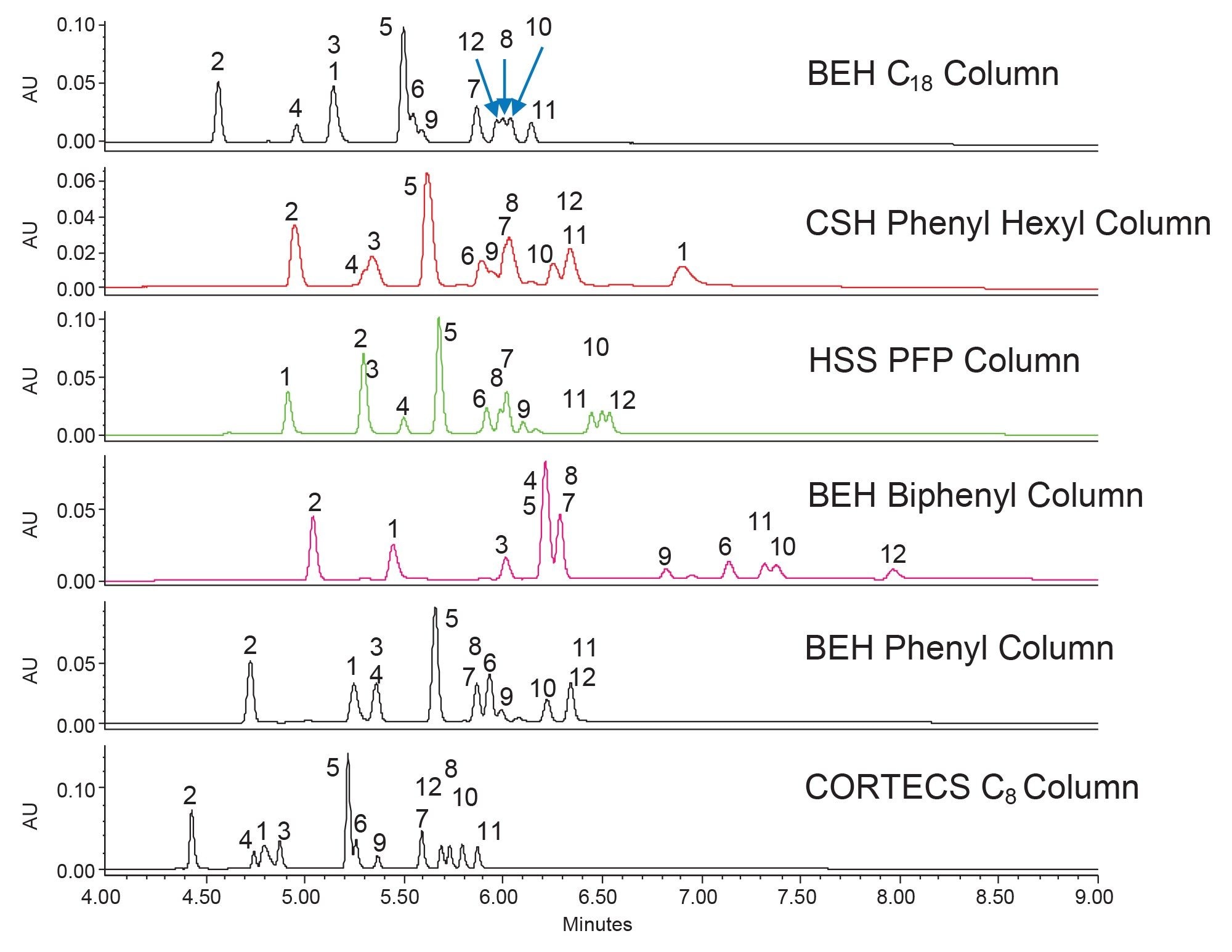
The separations achieved with acetonitrile show not only less retention, allowing for more optimization potential, but also better resolution. Under these conditions, the HSS PFP and BEH Biphenyl Columns separated all 12 compounds with the biphenyl column performing slightly better, especially for components 7, 8, 9, and 10. Using the BEH Biphenyl Column, the three pairs of isobars were all well separated (peaks 2 and 10, 7 and 8, and 11 and 12). The separation needs further modification however, as an unknown impurity peak co-elutes with component 11.
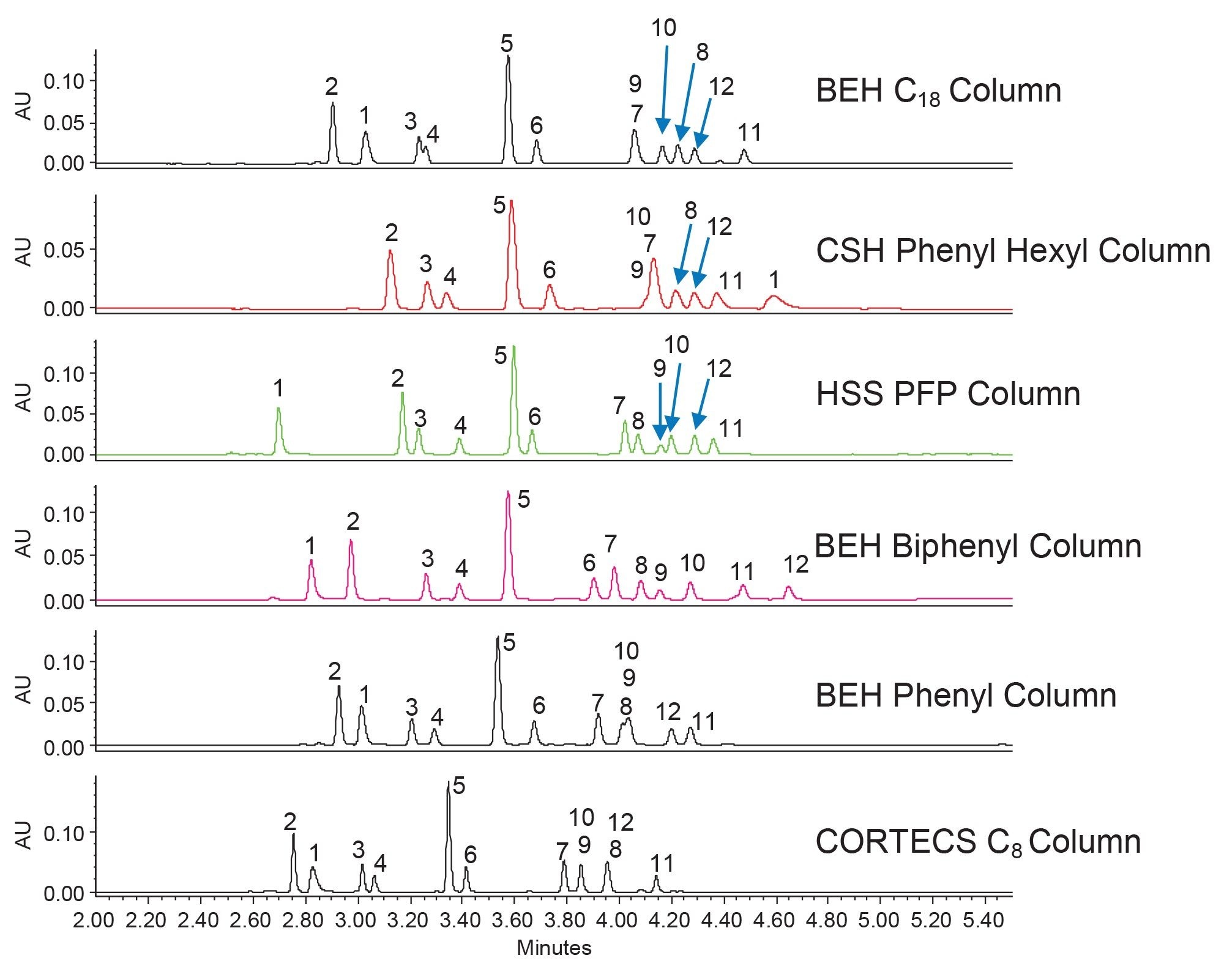
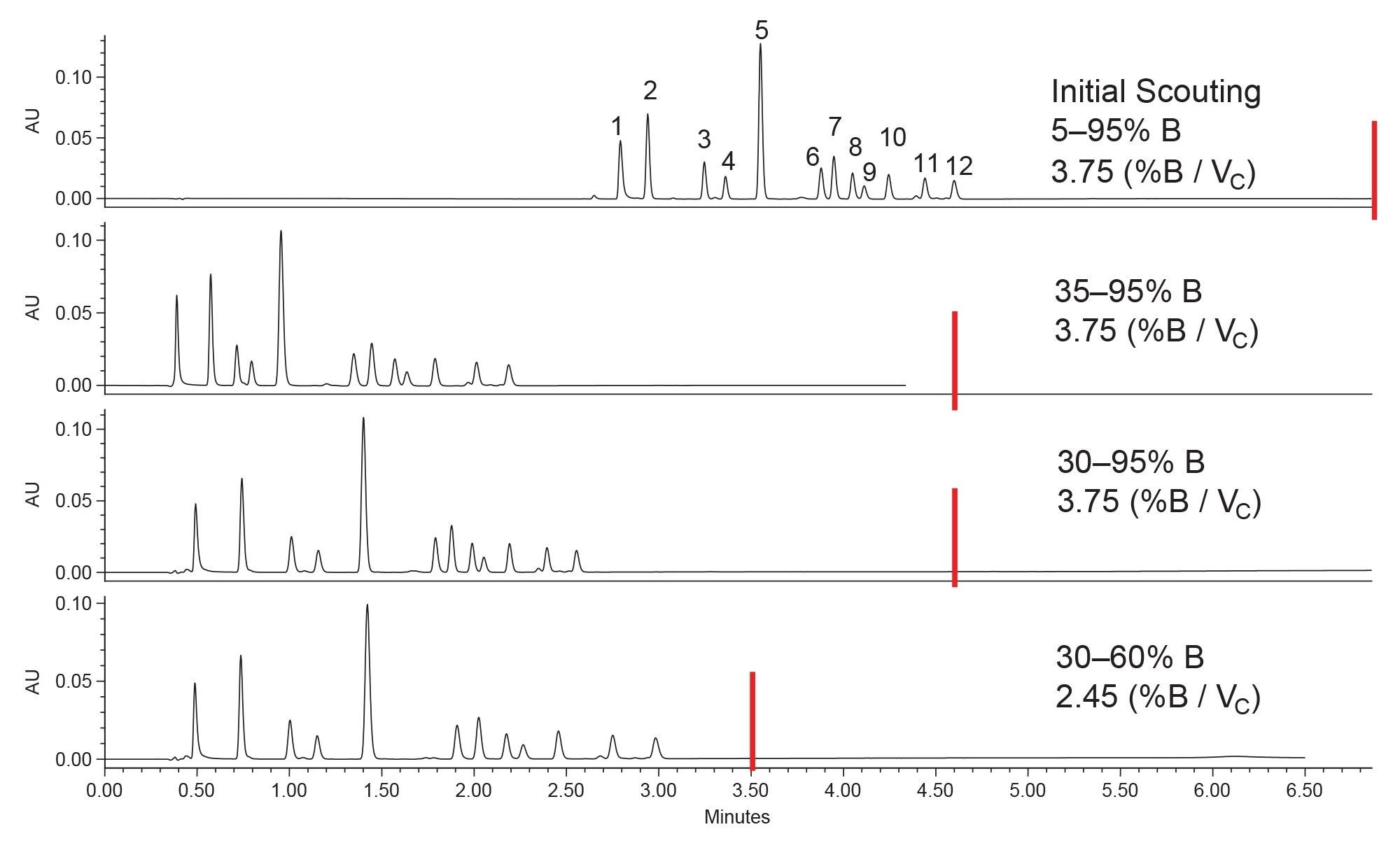
To improve the separation and reduce the analysis time, method optimization was performed. The first priority was to resolve the unknown impurity from component 11. The results of a column temperature optimization step are shown in Figure 4. Higher column temperatures can often be used to sharpen peaks, thereby increasing resolution. Additionally, by increasing the temperature some analytes will elute sooner.
By increasing the column temperature from 30 to 35 °C, the impurity was resolved from component 11. While increasing the temperature to 40 °C further increased the resolution of these two peaks, the improvement was not great enough to warrant the higher temperature. With the impurity resolved, the method could now be optimized to shorten the analysis time. This was accomplished by changing the starting percentage of organic modifier, as well as adjusting the gradient slope to better separate the compounds. The results of the gradient optimization steps are shown in Figure 5. Using the initial scouting conditions, the first compound elutes at ~2.8 minutes. This means there is room to shorten the analysis time by starting the gradient at a higher percent organic to reduce retention times while still achieving the desired separation. The first optimization step was to increase the starting organic content from 5% to 35%, which is the calculated composition needed to elute the first analyte. This caused the first compound to elute near the void time of the column which is not ideal, so the starting percentage was reduced to 30%. It is important to note for these changes in gradient profile, the gradient slope (%B/Vc) was kept constant so that the separation was maintained throughout each experiment. Once the starting percentage was determined, the end point of the gradient could be determined. In this case, the gradient can end at 60% acetonitrile and still elute the most hydrophobic compounds. With the truncated gradient profile, a shallower gradient slope was used to further separate the critical pairs.
Earlier it was mentioned that all columns selected employed MaxPeak Premier Technology to ensure that no interactions between analyte and column hardware would occur. For this sample, it is vital to ensure that no interactions occur, as betamethasone phosphate can adhere to metal surfaces through ionic interactions,8 similar to other phosphorylated compounds.4-7 To determine the benefits of inert hardware for these compounds, the BEH Biphenyl stationary phase was packed into both MaxPeak Premier and stainless steel column hardware. Representative chromatograms obtained using both types of hardware are shown in Figure 6.
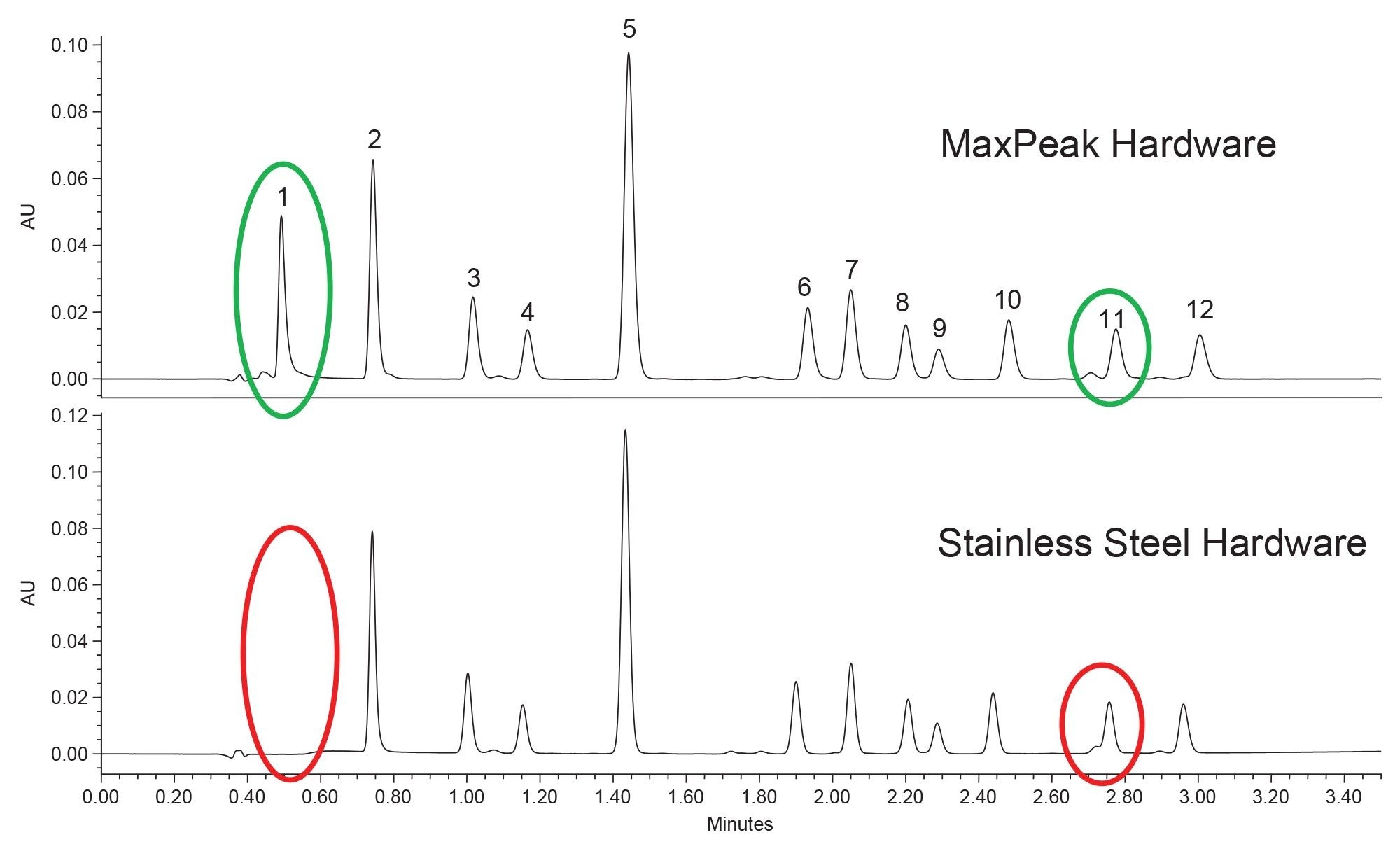
The results show that betamethasone phosphate (1) was not detected when using stainless steel column hardware under these conditions. An unexpected result obtained in this experiment was the slight retention shift of the impurity peak that elutes just before compound 11. During optimization, care was taken to achieve baseline separation of these two components to allow for better quantitation of compound 11. However, when using stainless steel hardware, the two co-eluted once more. This may be due to the impurity peak having an affinity for the stainless steel hardware, causing a slight retention shift. However, without knowing the properties of the impurity, this is purely speculative. What is certain, however, is that using inert hardware is critical for this analysis from the perspective of both analyte recovery and selectivity.
Conclusion
Successful method development hinges on the use of a diverse group of column stationary phases to maximize the selectivity space explored. Using inert column hardware further enhances method development by eliminating interactions between analytes and metal hardware, which can confound results and make optimization or troubleshooting more complex. Finally, using a streamlined method development protocol can simplify the process, making method development accessible for novice users as well as experts. This protocol was successfully employed to separate a mixture of 12 steroids including three sets of isobaric compounds and a steroid phosphate.
References
- Hong P, McConville P. A Complete Solution to Perform a Systematic Screening Protocol for LC Method Development. Waters White Paper. 720005268.
- Maziarz M, McCarthy S, Wrona M. Improving Effectiveness in Method Development by Using a Systematic Screening Protocol. Waters Application note. 720005026.
- Berthelette K, Collins C, Kalwood J, Haynes K. Using the Systematic Screening Protocol and MaxPeak Premier Columns to Separate Seven Janus Kinase Inhibitors. Waters Application note. 720008528.
- DeLano M, Walter TH, Lauber MA, Gilar M, Jung MC, Nguyen JM, Boissel C, Patel AV, Bates-Harrison A, Wyndham KD, Using Hybrid Organic-Inorganic Surface Technology to Mitigate Analyte Interactions with Metal Surfaces in UHPLC, Anal. Chem. 93 (2021) 5773-5781. doi: 10.1021/acs.analchem.0c05203
- Berthelette K, DeLoffi M, Collins C, Kalwood J, Walter TH. Correlation Between the Adsorption of Acidic Analytes on Stainless Steel Columns and Their Ionic Charge. Waters Application note. 720008792.
- Walter TH, Alden BA, Belanger J, Berthelette K, Boissel C, DeLano M, Kizekai L, Nguyen JM, Shiner S. Modifying the Metal Surfaces in HPLC Systems and Columns to Prevent Analyte Adsorption and Other Deleterious Effects. LCGC Supplement (2022) 28-34.
- Berthelette K, Aiello M, Collins C, Walter TH. Development of a UPLC Method for a Forced Degradation Study of Radioligand Therapy Precursor PSMA-617. Waters Application note. 720009042.
- Tanna N, Plumb R, Mullin L. Improvements in Sensitivity for Quantification of Steroid Phosphate Drugs Using ACQUITY Premier System and ACQUITY Premier Columns. Waters Application note. 720007095.
- Zabala G, Alden B, Boissel C, Walter TH, Gu W, Walsh DP, Cook J. A Highly Stable Biphenyl HPLC Stationary Phase Based on Ethylene-Bridged Hybrid Particles. Waters Application note. 720009261.
720009310, April 2026