For research use only. Not for use in diagnostic procedures.
This is an Application Brief and does not contain a detailed Experimental section.
This application brief demonstrates the quantitative analysis of plasma catecholamines and metanephrines using reversed-phase UPLC-MS/MS.
A fast and simple analysis of plasma catecholamines and metanephrines using Oasis WCX μElution plates, reversed-phase UPLC, and the Xevo TQ-S micro Mass Spectrometer.
Clinical researchers are often interested in measuring elevated concentrations of plasma catecholamines and their O-methylated metabolites (metanephrines). However, these compounds can be a challenge to analyze using reversed-phase LC-MS/MS due to their polarity. HILIC LC-MS/MS has been used successfully but can be challenging to implement in some laboratories. We have developed a reversed-phase UPLC-MS/MS method that employs a previously developed mixed-mode SPE procedure and combines it with new reversed-phase chromatography using the Waters ACQUITY UPLC HSS PFP Column.
The use of mixed-mode weak cation exchange solid-phase extraction (SPE) plates, in combination with the unique retention and selectivity of the ACQUITY UPLC HSS PFP Column and the Xevo TQ-S micro Mass Spectrometer, results in a rapid A fast and simple analysis of plasma catecholamines and metanephrines using Oasis WCX μElution plates, reversed-phase UPLC, and the Xevo TQ-S micro Mass Spectrometer. and analytically sensitive method with excellent linearity and precision, and minimal matrix effects. The resulting method provides a simple and fast workflow with no sample transfer after initial loading and pretreatment.
Plasma samples (250 μL) were pre-treated with 250 μL of 50-mM ammonium acetate containing 1 ng/mL internal standard. Samples were loaded directly onto individual wells of an Oasis WCX 96-well μElution Plate (p/n: 186002499) by positive pressure. After loading the samples, wells were washed with 200 μL of 50-mM ammonium acetate followed by 200 μL of methanol. The 96-well plate was dried to remove as much solvent as possible from the sorbent bed. The target compounds were eluted from the plate with 2 x 50-μL aliquots of methanol containing 2% formic acid into a 700-μL 96-well sample collection plate (p/n: 186005837). Samples were evaporated to dryness and reconstituted in 50 μL of 97:2:1 water:acetonitrile:formic acid.
Five microliters (5 μL) of the reconstituted sample was injected on the ACQUITY UPLC I-Class PLUS System with Flow-Through Needle (FTN) utilizing a water/acetonitrile gradient with each containing 0.1% formic acid and an ACQUITY UPLC HSS PFP Column (1.8 μm, 2.1 x 100 mm, p/n: 186005967). The UPLC gradient started at 2% MPB and increased to 30% MPB over 1.5 minutes, followed by an increase to 90% MPB over 0.5 minutes. The flow rate was 0.5 mL/min. Detection was performed using Multiple Reaction Monitoring (MRM) on the Xevo TQ-S micro System in positive ionization mode.
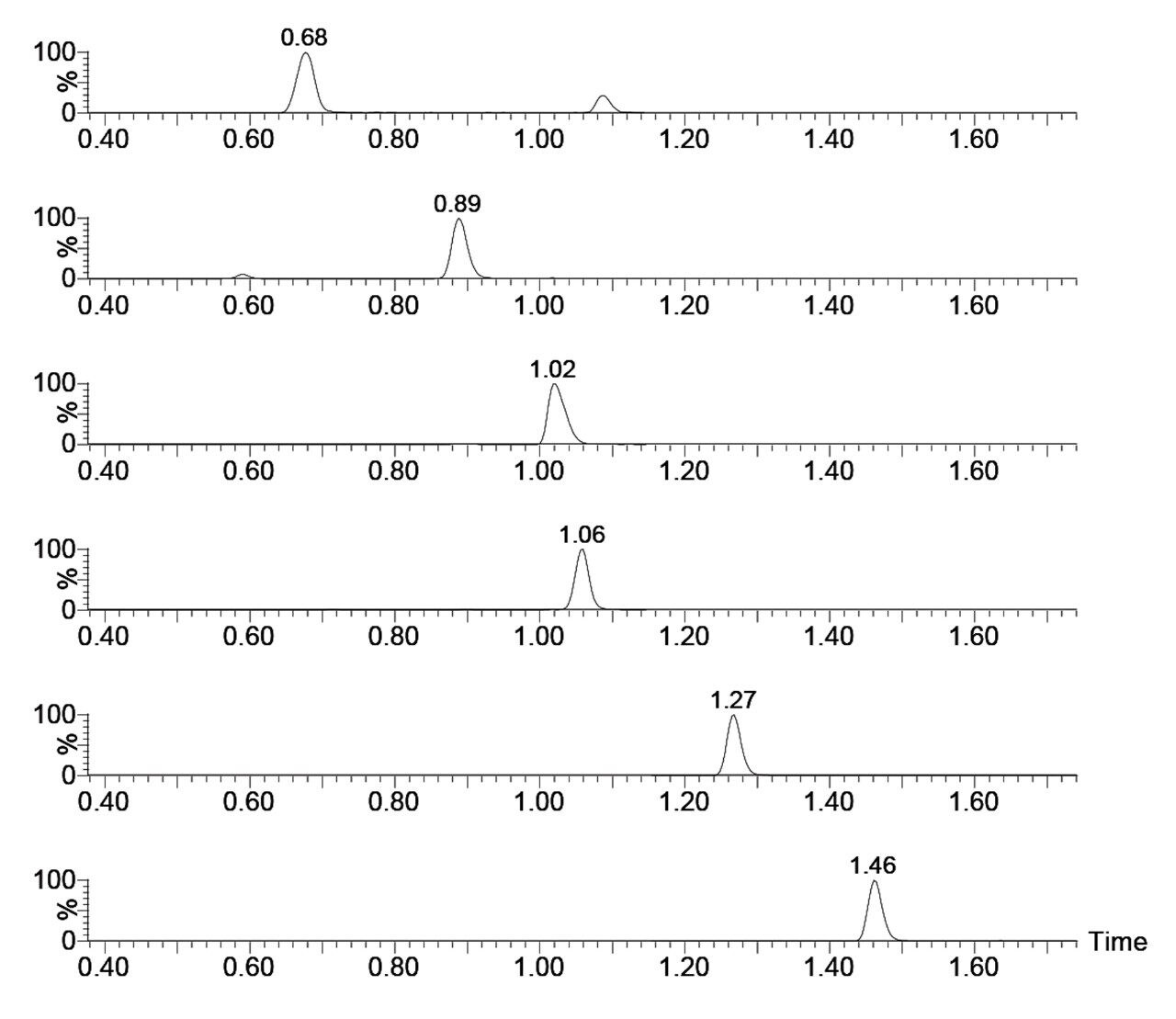
The ACQUITY UPLC HSS PFP Column has a unique selectivity among reversed-phase columns, which enables retention and separation of catecholamines and metanephrines. Figure 1 shows the chromatography of all compounds from a 100-pg/mL extracted calibration standard. The separation is rapid, with all compounds eluting within 1.5 minutes. One crucial feature of this separation is the baseline resolution between epinephrine and normetanephrine. These two compounds have the same molecular formula and can interfere with each other if not adequately separated (for this method an alternative precursor ion was chosen for normetanephrine).
Calibration curves were linear over the entire calibration range, up to 20,000 pg/mL. R² values were >0.999 for all compounds.
LLOQ were established by running three batches with 10 replicates at each low calibrator. LLOQ values were those in which the grand mean (N=30) and the between group mean were both within 20% of the nominal value and had %CVs <20%. These values ranged from 1 pg/mL (3-methoxytyramine) to 10 pg/mL (norepinephrine).
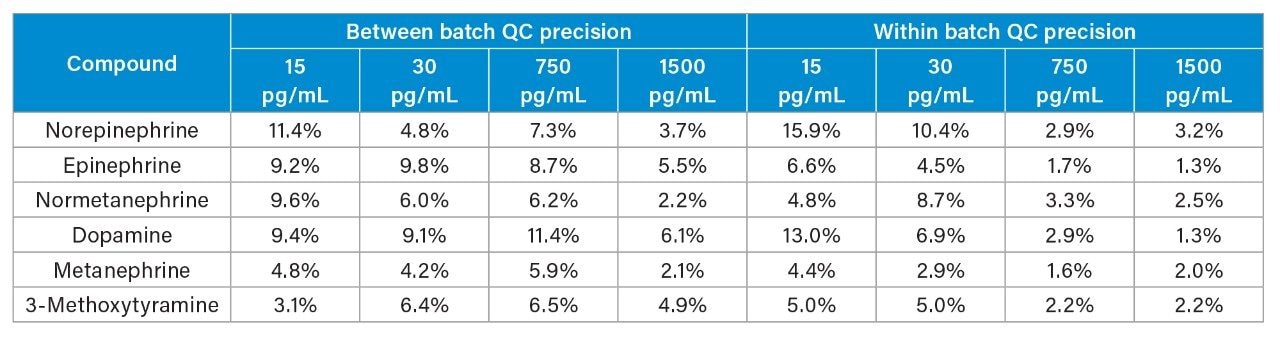
Between and within batch precision for plasma catecholamines and metanephrines is shown in Table 1. Accuracies for within and between batch QCs were all within 10% of target values.
Using the chromatography conditions stated, common interferences, such as metformin, midodrine, and L-DOPA (levodopa), were all chromatographically resolved from the analytes of interest.
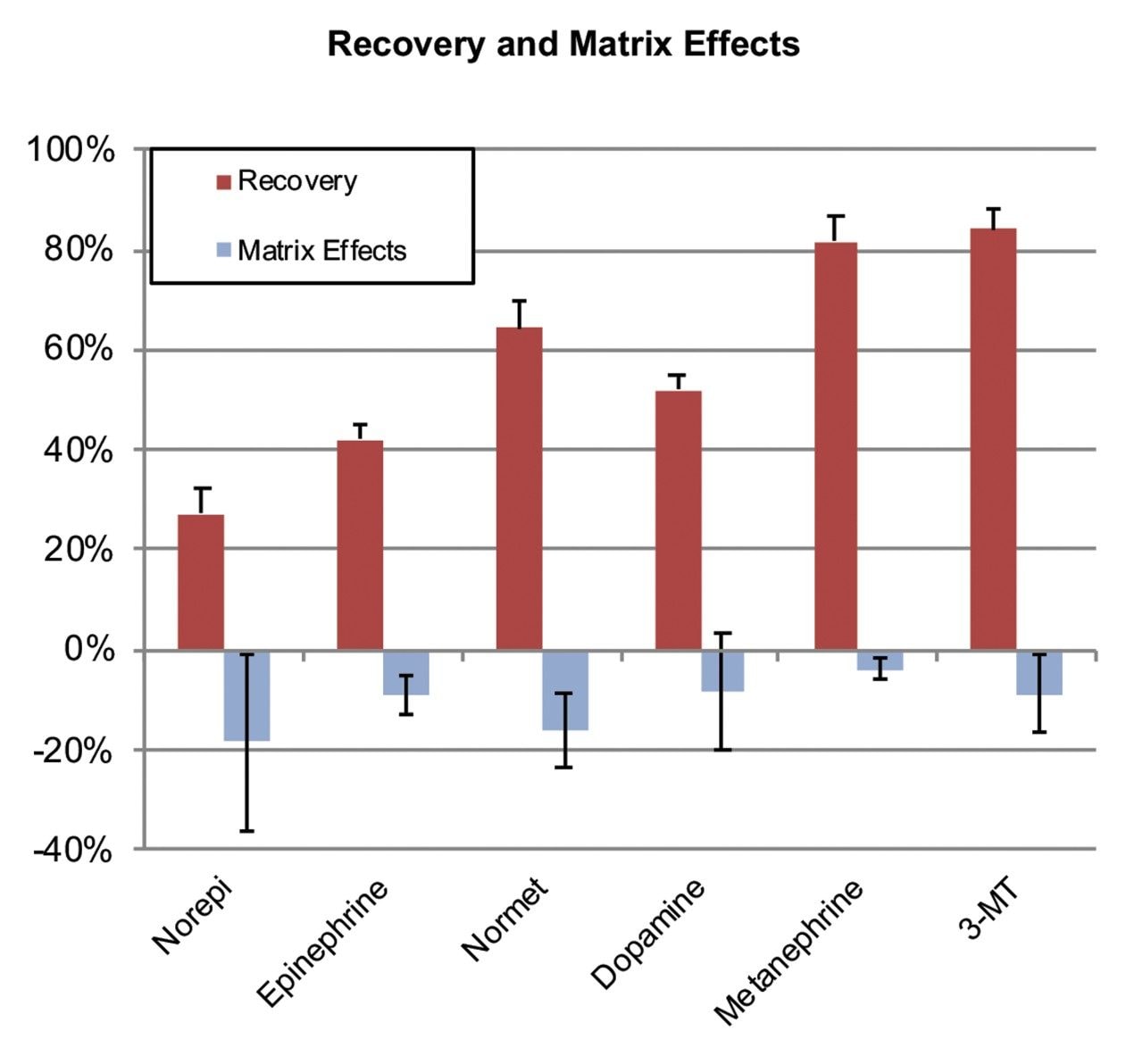
Mean extraction recoveries and matrix effects from five lots of plasma are shown in Figure 2. Despite the somewhat low absolute recoveries for norepinephrine and metanephrine, recoveries were highly consistent, with %CV values <20%, even across multiple lots of matrix. These recoveries were more than adequate for the analytical sensitivity required for this analysis.
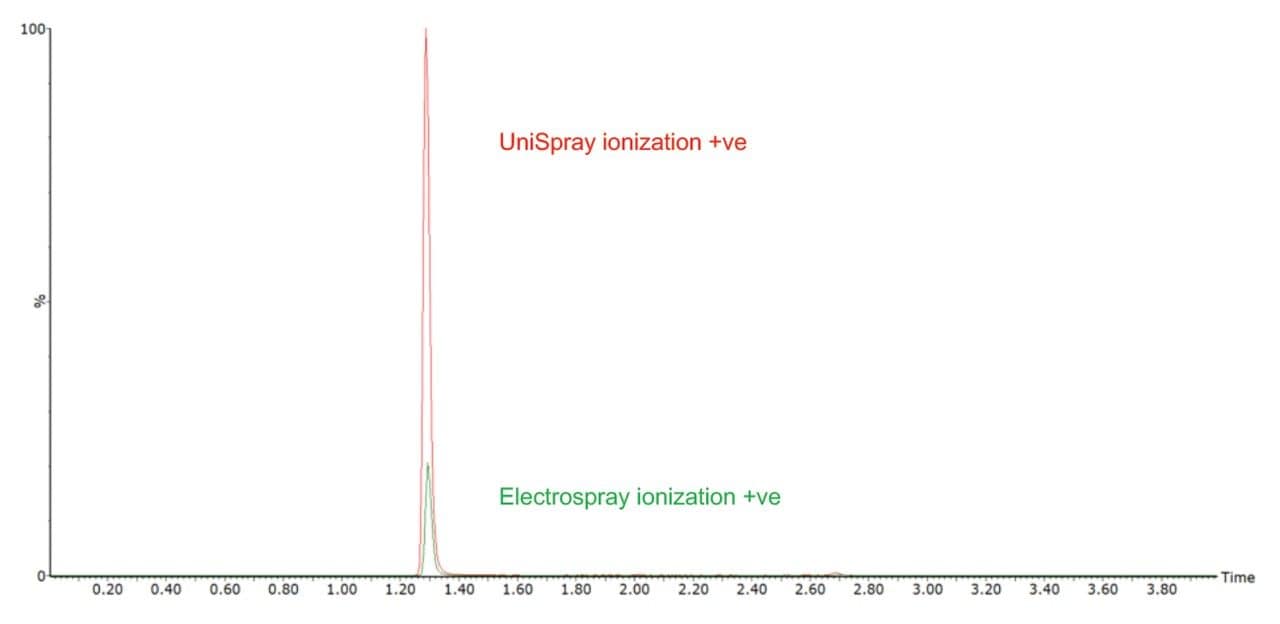
In addition, UniSpray Ion Source ionization was shown to improve the signal intensity for the analysis of catecholamines and metanephrines in plasma by approximately a factor of five. Figure 3 shows the analysis of an extracted metanephrine plasma sample using electrospray (green) and UniSpray (red).
A clinical research reversed-phase UPLC-MS/MS method for the separation and detection of plasma catecholamines and metanephrines has been developed. The benefits of this method include:
720006778, March 2020