For research use only. Not for use in diagnostic procedures.
Here we describe a clinical research method utilizing Ostro phospholipid removal plate technology for the extraction of MMA from serum. Chromatographic separation was performed on an ACQUITY UPLC I-Class System using an ACQUITY UPLC 1.7 μm CSH C18 Column followed by detection on a Waters Xevo TQ-S micro Tandem Quadrupole Mass Spectrometer to enable quantification of very low physiological concentrations of MMA.
Vitamin B12 plays an integral role in critical biological functions, such as DNA synthesis, formation of erythrocytes, and maintenance of the myelin sheath in the nervous system. In its role as a co-factor, vitamin B12 aids the methyl-malonyl-CoA mutase catalyzed conversion of methylmalonyl-CoA to succinyl-CoA, with methylmalonic acid (MMA) being an important by-product in this pathway. MMA concentrations are elevated when there is insufficient vitamin B12 to help catalyze this reaction, meaning that MMA is a candidate biomarker for clinical research investigations into the function of vitamin B12. LC-MS/MS is an ideal platform to perform this analysis. Chromatographic separation is key in separating MMA from isobaric and isomeric species, such as succinic acid, that can cause a significant concentration bias. To help facilitate adoption of this methodology, a simplified workflow is necessary to enable a high throughput of samples.
Here we describe a clinical research method utilizing Ostro phospholipid removal plate technology for the extraction of MMA from serum. Chromatographic separation was performed on an ACQUITY UPLCI-Class System using an ACQUITY UPLC 1.7 μm CSH C18 Column followed by detection on a Waters Xevo TQ-S micro Tandem Quadrupole Mass Spectrometer to enable quantification of very low physiological concentrations of MMA (Figure 1).

|
System: |
ACQUITY UPLC I-Class (FL) with Column Heater (CH) |
|
Needle: |
20 μL |
|
Column: |
ACQUITY UPLC CSH C18 2.1 × 100 mm, 1.7 μm (p/n: 186005297) |
|
Pre-column: |
In-line filter (p/n: 205000343) |
|
Mobile phase A: |
Water with 0.2% formic acid |
|
Mobile phase B: |
Acetonitrile with 0.2% formic acid |
|
Weak wash solvent: |
Mobile phase A, 900 μL |
|
Strong wash solvent: |
Mobile phase B, 600 μL |
|
Column temp.: |
40 °C |
|
Injection volume: |
10 μL |
|
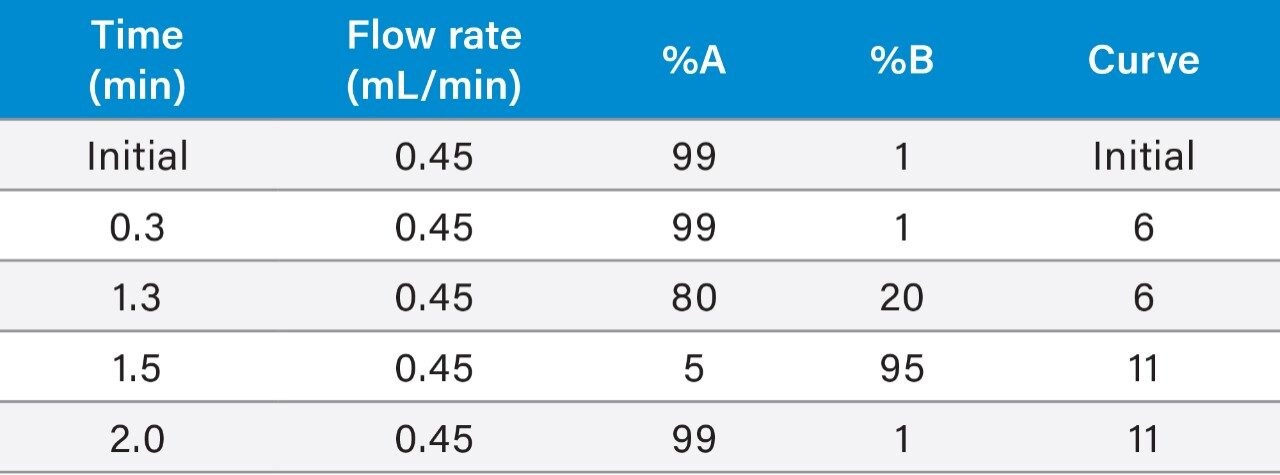
Flow rate: |
See table 1 |
|
Gradient: |
See table 1 |
|
Run time: |
3.3 minutes |
|
System: |
Xevo TQ-S micro |
|
Resolution: |
MS1 (0.75 FWHM) and MS2 (0.75 FWHM) |
|
Acquisition mode: |
Multiple Reaction Monitoring (MRM) (see Table 2 for details) |
|
Polarity: |
ESI- |
|
Capillary: |
0.50 kV |
|
Source temp.: |
150 °C |
|
Desolvation temp.: |
600 °C |
|
Inter-scan delay: |
Automatic |
|
Inter-channel delay: |
Automatic |
MassLynx Software v4.2 with TargetLynx Application Manager
Methylmalonic acid certified reference solutions and methylmalonic acid-13C4 stable labelled internal standard were purchased from Sigma Aldrich (Poole, UK).
Calibrators were prepared in 1% (w/v) bovine serum albumin (BSA) in phosphate buffered saline and quality controls (QCs) were prepared in pooled human plasma (BioIVT, UK). Methylmalonic acid calibrators were prepared over the range of 21–1270 nmol/L. QC concentrations were 152, 364, and 982 nmol/L. Additional MMA QCs (Recipe, Germany) in reconstituted lyophilized serum with concentrations at 264 and 598 nmol/L were also used to evaluate performance of the method. Note: To convert SI units to conventional mass units divide by 8.468 for methylmalonic acid (nmol/L to ng/mL).
Samples were centrifuged at 3000 g for five minutes prior to extraction. 100 μL of sample was added directly to an Ostro phospholipid removal plate (p/n: 186005518). 25 μL of internal standard solution (83 nmol/L methylmalonic acid-13C4) and 400 μL 1% formic acid in acetonitrile were added, mixing vigorously after each reagent addition. Using a vacuum, samples were eluted into a 2-mL collection plate (p/n: 186002482) and then evaporated to dryness using nitrogen at 50 °C. Samples were reconstituted in 60 μL 1% formic acid in water, mixed vigorously, and centrifuged for two minutes at 3000 g prior to injection.

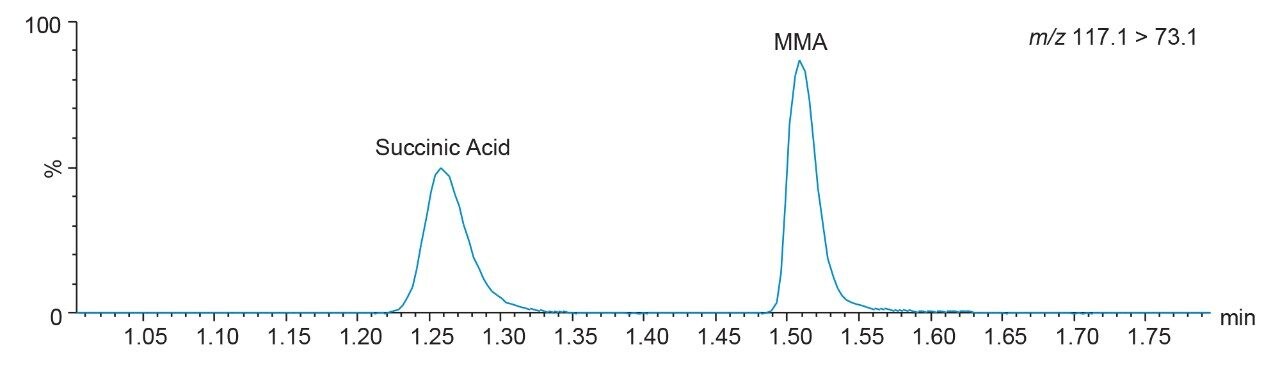
No significant interferences (recovery within ±15% bias) were observed at the retention time of methylmalonic acid, when succinic acid, homocysteine, and homocystine were examined. Chromatographic separation of MMA and succinic acid is demonstrated in Figure 2. No significant interferences (recovery within ±15% bias) were observed when other endogenous compounds were examined (albumin, bilirubin, uric acid, intralipid, triglycerides, and cholesterol).
No significant system carryover (<20% of the lowest calibrator) was observed from high concentration samples into subsequent blank injections. A 1:5 dilution was successfully employed on high concentration samples at 4234 nmol/L, providing a mean accuracy of 103% for methylmalonic acid with an RSD of 3%.
Analytical sensitivity investigations were performed using methylmalonic acid spiked into stripped serum over three occasions across and below the calibration range (n=30 at each concentration). The method would allow for precise quantification (<20% RSD) at 21 nmol/L for methylmalonic acid. The S/N (PtP) was >10 at 21 nmol/L.
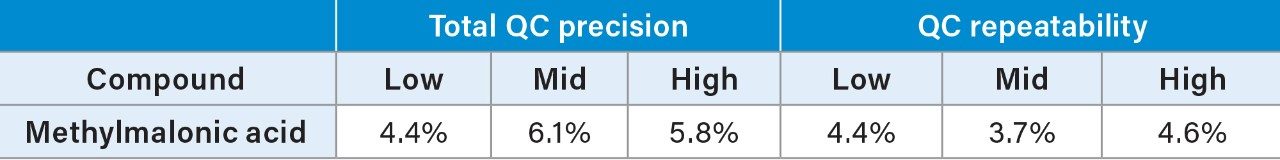
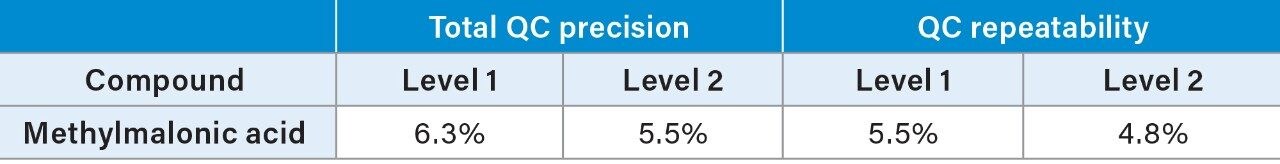
Total precision of MMA was determined in pooled plasma and Recipe serum controls by extracting and quantifying five replicates of the QC material per day over five separate days (n=25). Repeatability was assessed by analyzing five replicates at each QC level. Low, mid, and high concentrations in pooled plasma were 152, 364, and 982 nmol/L (Table 3), and level 1 and 2 concentrations in Recipe controls were 264 and 598 nmol/L (Table 4).
The method was shown to be linear for methylmalonic acid (6.8–1524 nmol/L) when different ratios of high and low concentration pools of the analytes were combined and analyzed. In addition, calibration lines were linear with coefficient of determinations (r2) >0.995 for all analyses.
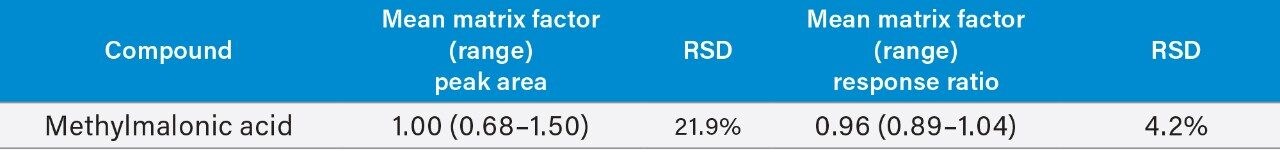
Matrix effect investigations for methylmalonic were performed using individual donor samples (n=6). The matrix factor calculated is shown in Table 5. Normalized matrix factor calculations, based on the analyte:internal standard response ratio, demonstrated that the internal standards compensated for any matrix effects observed.
Stability of extracted MMA samples onboard the autosampler at 8 °C was demonstrated for 67 hours, with mean QC concentrations within 2% of the previously calculated values.
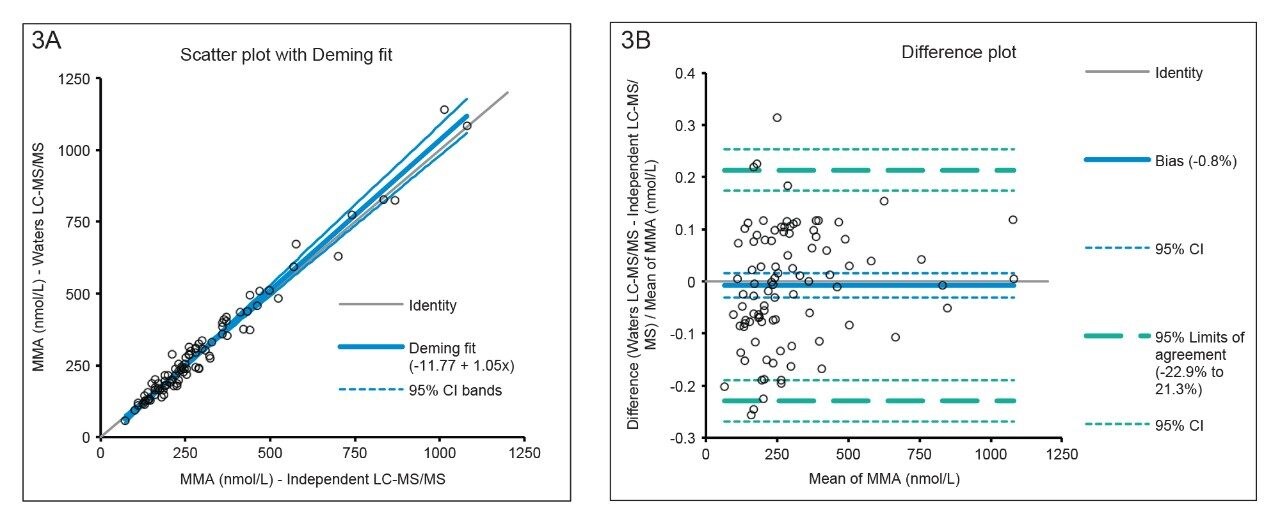
A method comparison to an independent LC-MS/MS method for the analysis of 94 methylmalonic acid serum samples was performed. Statistical analysis using Deming regression demonstrated no statistically significant proportional (p = 0.1950) or constant bias (p = 0.1911) with a fit of y = 1.05x–11.77 (Figure 3A). Bland-Altman agreement for methylmalonic acid demonstrated a mean method bias of -0.8%, demonstrating excellent agreement with the independent LC-MS/MS method for methylmalonic acid (Figure 3B).
An analytically sensitive and selective clinical research method has been developed for the analysis of methylmalonic acid using the Xevo TQ-S micro.
The Xevo TQ-S micro enables the analysis of physiologically low levels down to 21 nmol/L of MMA, while only using 100-μL sample volume. Excellent levels of precision across the calibration range have been demonstrated. Comparison to an independent LC-MS/MS method demonstrated excellent agreement in serum samples. In-well protein precipitation using Ostro plates facilitated a simple sample preparation workflow for high throughput analysis of MMA.
We would like to thank Anne Schmedes and colleagues at Lillebælt Hospital, Denmark for providing anonymized samples for comparison purposes.
720006806, March 2020